-

-
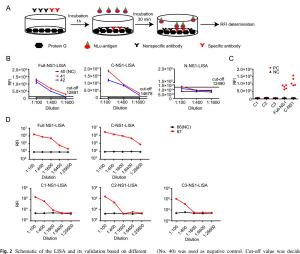
-
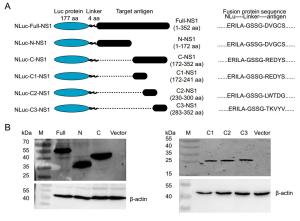
-
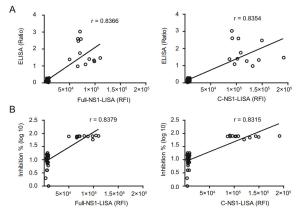
-

-

-
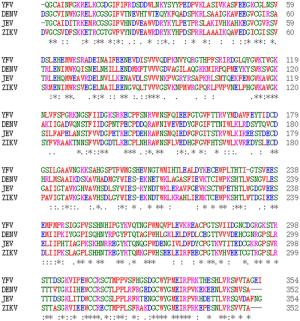
-
Group Reference No. Source ZIKV-RT-PCR MN ELISA LISA-Full-NS1 LISA-C-NS1 Region Notes Control 1-40 China Serum from blood donors - - - - - Samples from ZIKV patients (n=63) 41 Cambodia Serum; 14 days; acute phase + + + + + 42 Cambodia Serum; 285 days; from #41 patient; - + + + + 43 Venezuela Serum; 10 days; acute + + + + + 44 Venezuela Serum; 3 days; acute phase + - - - - 45 Venezuela Serum; 18 days; from #44 patient; + + - + + 46-53 CHs from SA Serum; from 8 confirmed ZIKV patients + + + + + 54 CH from SA Urine; acute phase + - - - - 55 CH from SA Urine; acute phase + - - - - 56 CH from SA Saliva; from #55 patient; acute phase + - - - - Animal serum (n=13) 57 China Monkey w/o ZIKV-infection - - ND - - 58-60 China ZIKV-infected monkey - + ND + + 61 China Mice w/o ZIKV-infection - - ND - - 62-65 China ZIKV-infected mice - + ND + + 66 China Rabbit w/o ZIKV-infection - - ND - - 67-69 China ZIKV-infected rabbit - + ND + + DENV serum (n=47) 70-106 China anti-DENV IgG (+) - - - - - 107 China anti-DENV IgG (+) - - + + + 108 China anti-DENV IgG (+) - - + + + 109 China anti-DENV IgG (+) - - + + + 110-115 Africa anti-DENV IgG (+) - - - - - 116 Africa anti-DENV IgG (+) - - + + + JEV serum (n=6) 136-141 China anti JEV IgG (+) - - - - - HCV serum (n=10) 142-151 China anti HCV IgG (+) - - - - - +, positive; -, negative; CH, Chinese case; SA, South American; ND, not detected. Table 1. Summary of sample detection in this study.
-
Virus ZIKV-NS1 ZIKV-NS1-C(aa: 172-352) ZIKV-NS1 100 100 DENV-NS1 54 61 JEV-NS1 56 61 WNV-NS1 56 60 YFV-NS1 47 51 HCV-NS1 44 42 NCBI accession numbers for the different viruses are: ZIKV (BeH819015), DENV (NP_059433.1), JEV (AAA81554.1), WNV (ADZ13193.1), YFV (NP_041726.1), and HCV (BAJ07247.1). Table 2. Comparison of amino acid homology between ZIKV NS1 and corresponding segments of different Flaviviridae members.
Figure 7 个
Table 2 个