This special issue is dedicated to the recent research progress on human herpesviruses (HHVs). Human herpesviruses are distributed worldwide, and more than 90% of adults are infected by one or multiple HHVs. The HHV family contains three sub-families: the alpha sub-family [herpes simplex virus 1 (HSV-1), HSV-2, and varicella-zoster virus (VZV)], beta sub-family [human cytomegalovirus (HCMV), HHV6, and HHV7)], and gamma sub-family. [Epstein-Barr virus (EBV) and Kaposi’s sarcoma–associated herpesvirus (KSHV)]. All the viruses typically establish latent infection in host, and undergo lytic reactivation in certain pathophysiological conditions. In this issue, we collectively present ten articles focusing on the epidemiology, pathogenesis, and interventions of HSV-1, VZV, HCMV, EBV and KSHV respectively, and these high-quality review and research articles are contributed by experts on those specific viruses.
-
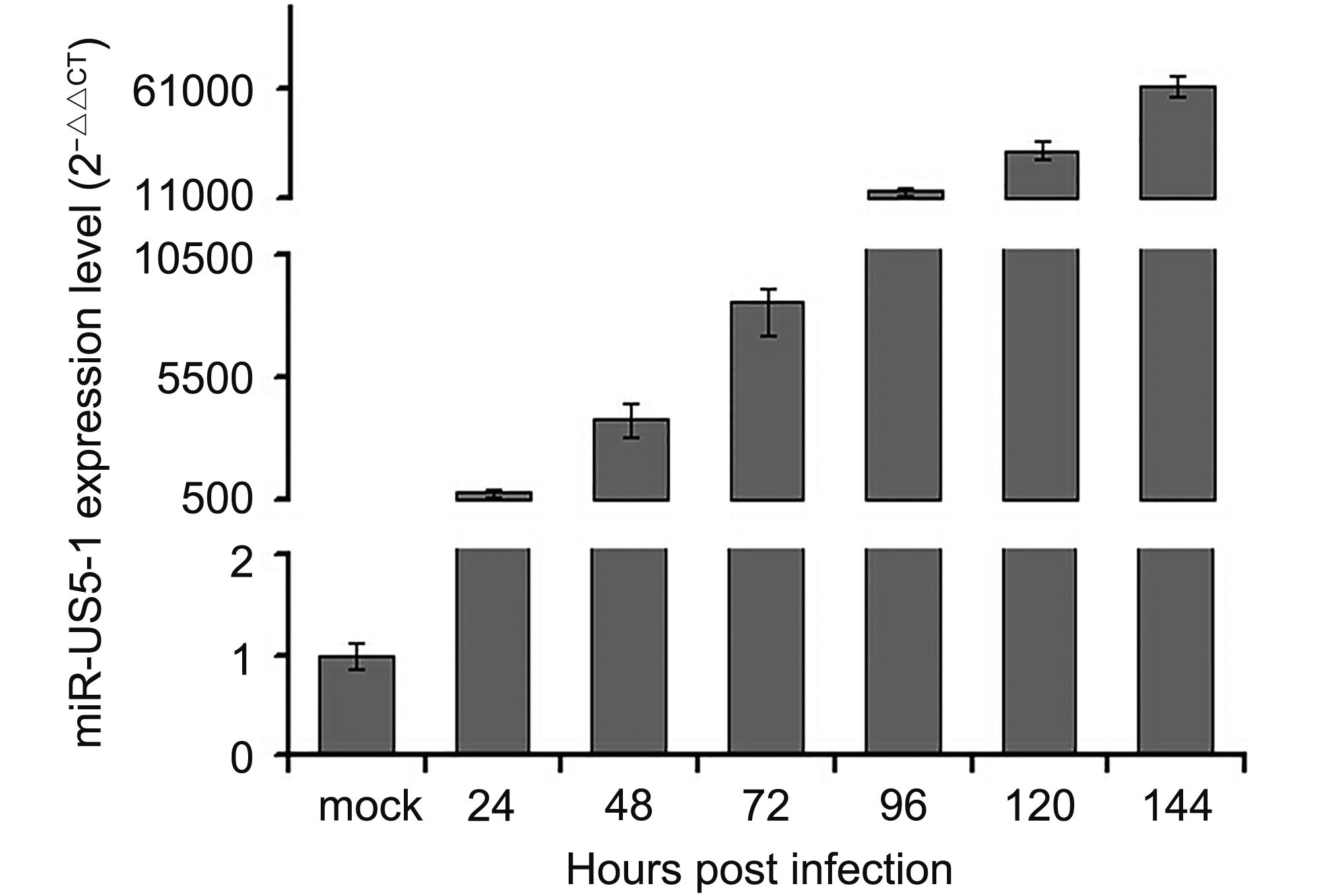
Human cytomegalovirus miR-US5-1 inhibits viral replication by targeting Geminin mRNA
2017, 32(5): 431 doi: 10.1007/s12250-017-4064-x
Received: 14 August 2017 Accepted: 26 October 2017 Published: 31 October 2017Viruses commonly create favorable cellular conditions for their survival through multiple mechanisms. MicroRNAs (miRNAs), which function as post-transcriptional regulators, are utilized by human cytomegalovirus (HCMV) in its infection and pathogenesis. In the present study, the DNA replication inhibitor Geminin (GMNN) was identified to be a direct target of hcmv-miR-US5-1. Overexpression of hcmv-miR-US5-1 could block the accumulation of GMNN during HCMV infection, and the decrease of GMNN expression caused by hcmv-miR-US5-1 or GMNN specific siRNA reduced HCMV DNA copies in U373 cells. Meanwhile, ectopic expression of hcmv-miR-US5-1 and consequent lower expression of GMNN influenced host cell cycle and proliferation. These results imply that hcmv-miR-US5-1 may affect viral replication and host cellular environment by regulating expression kinetics of GMNN during HCMV infection. -
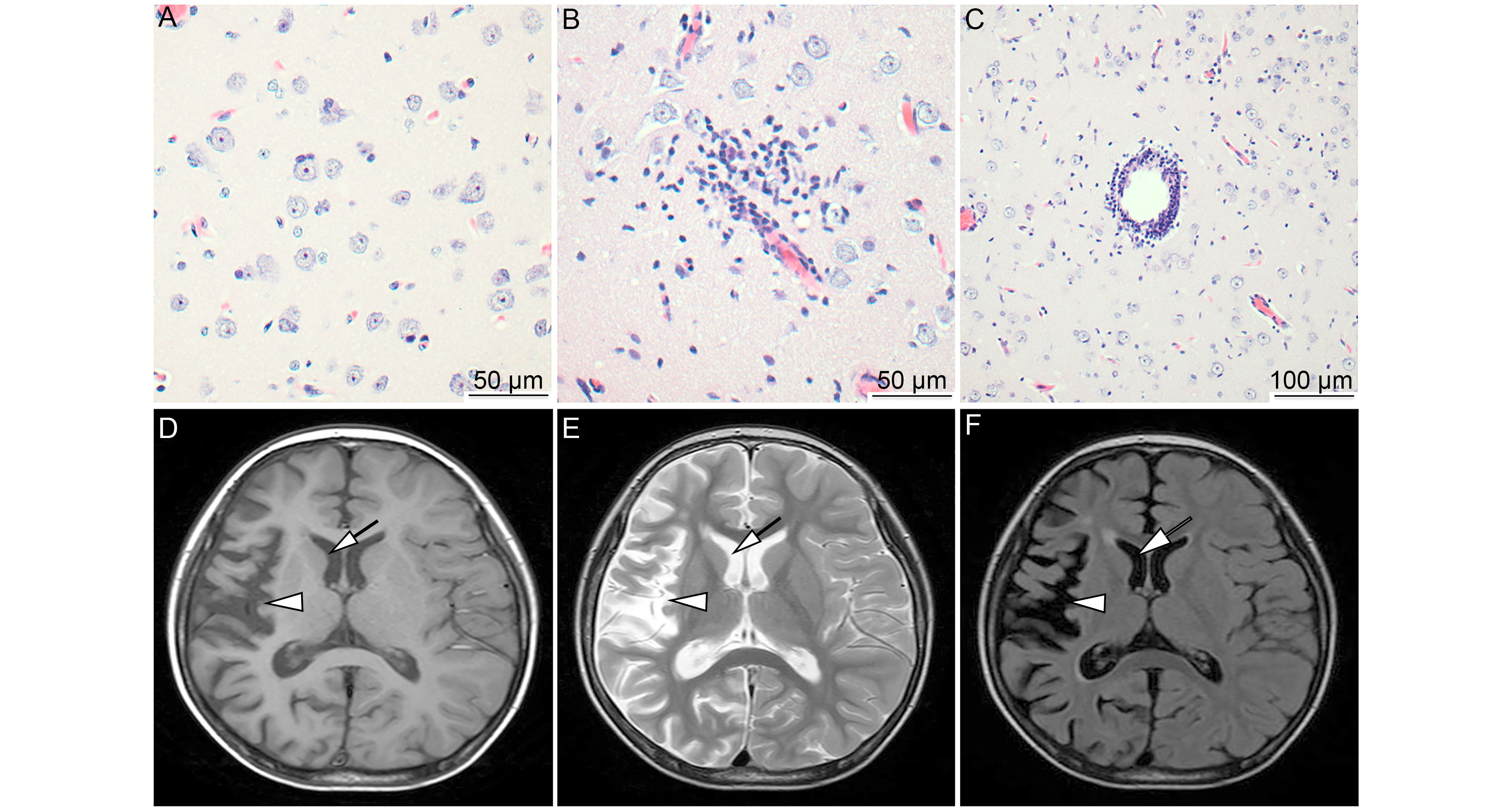
Elevated expression of EBV and TLRs in the brain is associated with Rasmussen’s encephalitis
2017, 32(5): 423 doi: 10.1007/s12250-017-4058-8
Received: 26 July 2017 Accepted: 30 September 2017 Published: 30 October 2017Rasmussen’s encephalitis (RE) is a rare pediatric neurological disorder, the etiology of which remains unclear. It has been speculated that the immunopathogenesis of RE involves damage to neurons, which eventually leads to the occurrence of RE. Viral infection may be a critical factor in triggering RE immunopathogenesis. In this study, we analyzed the expression of Epstein-Barr virus (EBV) antigens as well as of Toll-like receptor 3 (TLR3), TLR9, and downstream adapter TIRdomain-containing adapter-inducing interferon-β (TRIF) in the brain tissues of 26 patients with RE and 16 control individuals using immunohistochemistry (IHC). In the RE group, EBV antigens were detected in 53% of individuals at various expression levels. In contrast, there was no detectable EBV antigen expression in control brain tissues. Moreover, we found marked increases in the expression of TLR3, TLR9, and TRIF in the brain tissues of RE patients compared with levels in the control group. Furthermore, among RE cases, EBV expression and high TLR3 expression were associated with more severe brain atrophy. Our results suggest that the elevated expression of EBV and TLRs may be involved in RE occurrence through the activation of downstream molecules. -
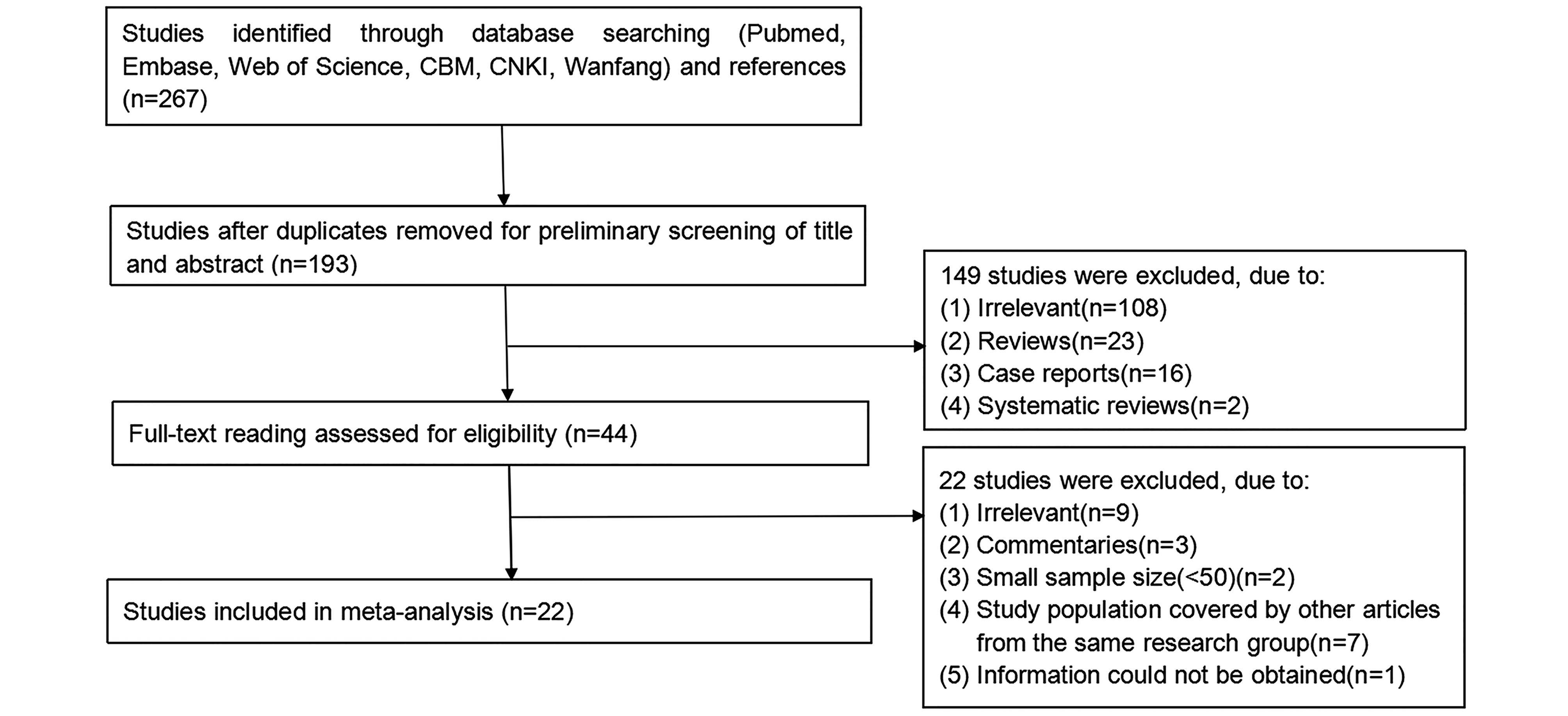
Prevalence of Kaposi’s sarcoma-associated herpesvirus among intravenous drug users: a systematic review and meta-analysis
2017, 32(5): 415 doi: 10.1007/s12250-017-4051-2
Received: 30 July 2017 Accepted: 18 September 2017 Published: 13 October 2017Intravenous drug users (IDUs) have been demonstrated to be highly vulnerable to HIV/AIDS. Nevertheless, the prevalence of Kaposi’s sarcoma associated herpesvirus (KSHV), an important co-infected agent with HIV, among this population remained obscure. We conducted a systematic review on the epidemiological features of KSHV among IDUs worldwide. Eligible studies were retrieved from 6 electronic databases (PubMed, EMBASE, Web of Science, CBM, CNKI and Wanfang). We calculated the pooled prevalence and 95% confidence interval (CI) overall and among subgroups using either random-effects model or fixed-effects model depending on between-study heterogeneity. The potential publication bias was assessed by the Egger’s test. A meta-regression analysis was performed to explore the sources of heterogeneity. Finally, twenty-two studies with a total sample of 7881 IDUs were included in the analysis. The pooled prevalence of KSHV was 14.71% (95% CI 11.12%–19.46%) among IDUs. Specifically, KSHV prevalence was 10.86% (95% CI 6.95%–16.96%) in HIV-negative IDUs, and 13.56% (95% CI 10.57%–17.38%) in HIV-positive IDUs. Moreover, prevalence among IDUs from the three continents involved in the current study was similar: 16.10% (95%CI 7.73%–33.54%) in Asia; 14.22% (95%CI 8.96%–22.57%) in Europe and 14.06% (95%CI 11.38%–17.37%) in America. Globally, IDUs are at higher risk of the KSHV infection when compared with the general population, regardless of geographical region or HIV-infection status. -

Attenuated phenotypes and analysis of a herpes simplex virus 1 strain with partial deletion of the UL7, UL41 and LAT genes
2017, 32(5): 404 doi: 10.1007/s12250-017-3947-1
Received: 19 January 2017 Accepted: 17 August 2017 Published: 29 September 2017We previously constructed a herpes simplex virus 1 (HSV-1) UL7 mutant virus (M1) and showed that a partial deletion mutation of the UL7 gene led to a lower proliferative rate and an attenuated phenotype. Using the M1 mutant, we further modified the UL41 gene, which encodes another tegument protein, and the latency-associated transcript (LAT) gene. Observations of the resulting mutants with modified UL7 and UL41 (M2) or UL7, UL41 and LAT (M3) genes indicated attenuated phenotypes, with lower proliferative ratios in various cells, non-lethal infections in mice and lower viral loads in nervous tissues compared with the wild-type strain. Furthermore, no LAT stable intron could be detected in the trigeminal ganglion of M3-infected animals. The results obtained with the three HSV-1 mutants indicate that the M3 mutant is an attenuated strain with low pathogenicity during both acute and latent infections. Together, the results support the use of the M3 mutant as a candidate for the development of an HSV-1 vaccine. -
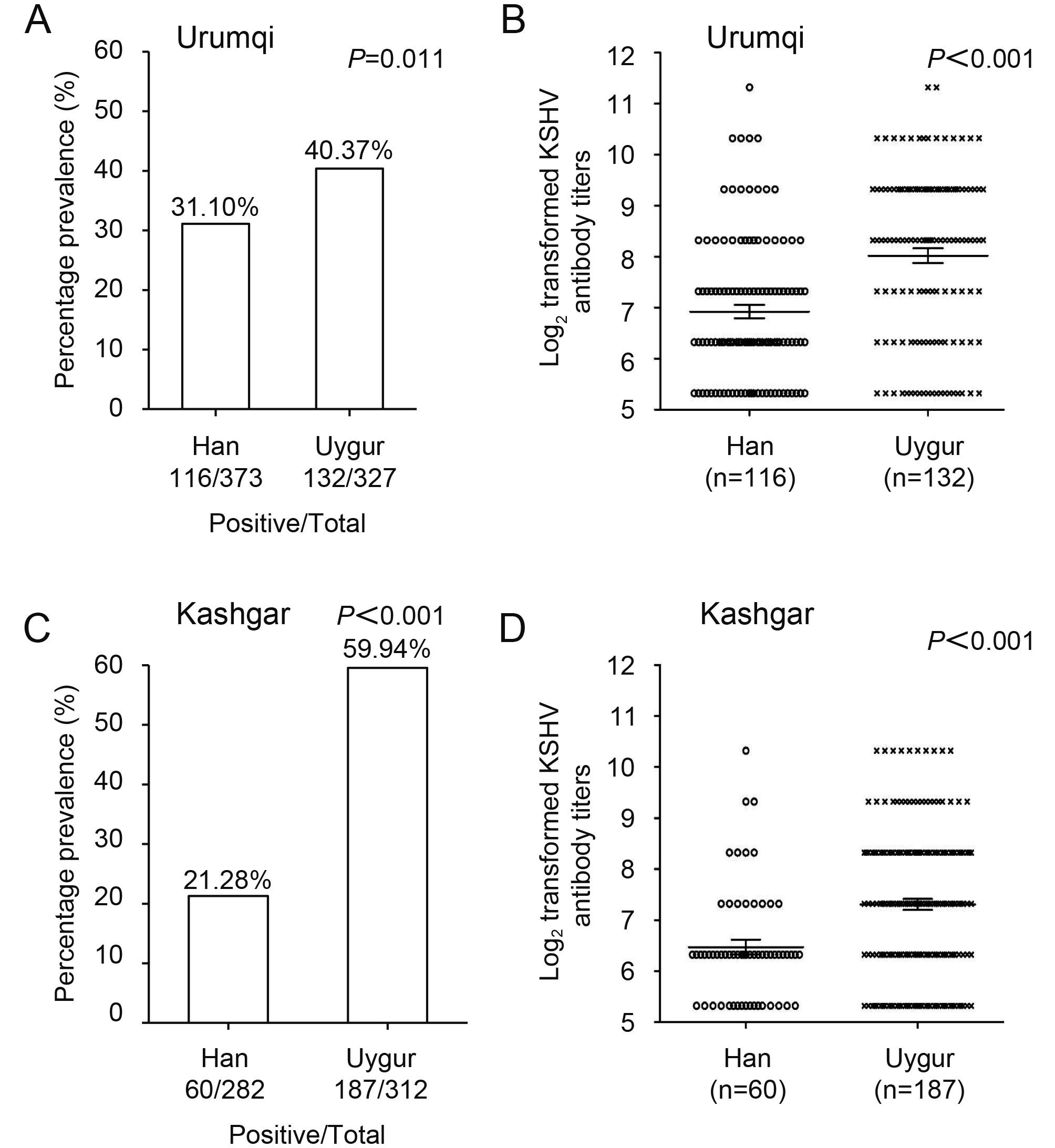
Prevalence of Kaposi’s sarcoma-associated herpesvirus in Uygur and Han populations from the Urumqi and Kashgar regions of Xinjiang, China
2017, 32(5): 396 doi: 10.1007/s12250-017-4049-9
Received: 29 July 2017 Accepted: 18 September 2017 Published: 25 October 2017Kaposi’s sarcoma-associated herpesvirus (KSHV) is the infectious etiologic agent associated with Kaposi’s sarcoma (KS), primary effusion lymphoma, and multicentric Castleman disease. It has been shown that high KSHV prevalence and high incidence of both classic KS and AIDS associated KS are found mostly among people of Uygur ethnicity in Xinjiang, while people of Han ethnicity in Xinjiang have a higher KSHV seroprevalence than those of other Han populations in mainland China. However, it is still unclear why there is such geographical and population variation in KSHV distribution in China. In this work, we focused on the populations in the Kashgar region and Urumqi area, where a total of 1294 research subjects were randomly selected to investigate the potential correlation between KSHV prevalence and different ethnicities in endemic areas of Xinjiang, and to determine risk factors that may affect KSHV infection rates or KS incidence. We identified a high seroprevalence of KSHV and high peripheral blood DNA infection in the general Uygur and Han populations in both Urumqi and Kashgar regions of Xinjiang, and determined that advancing age, low education level, and stationary population status affect KSHV infection rates. Further, KSHV-positive Uygur participants were shown to have higher prevalence of neutralizing antibodies and neutralizing antibody titers than KSHV-positive Han participants. -
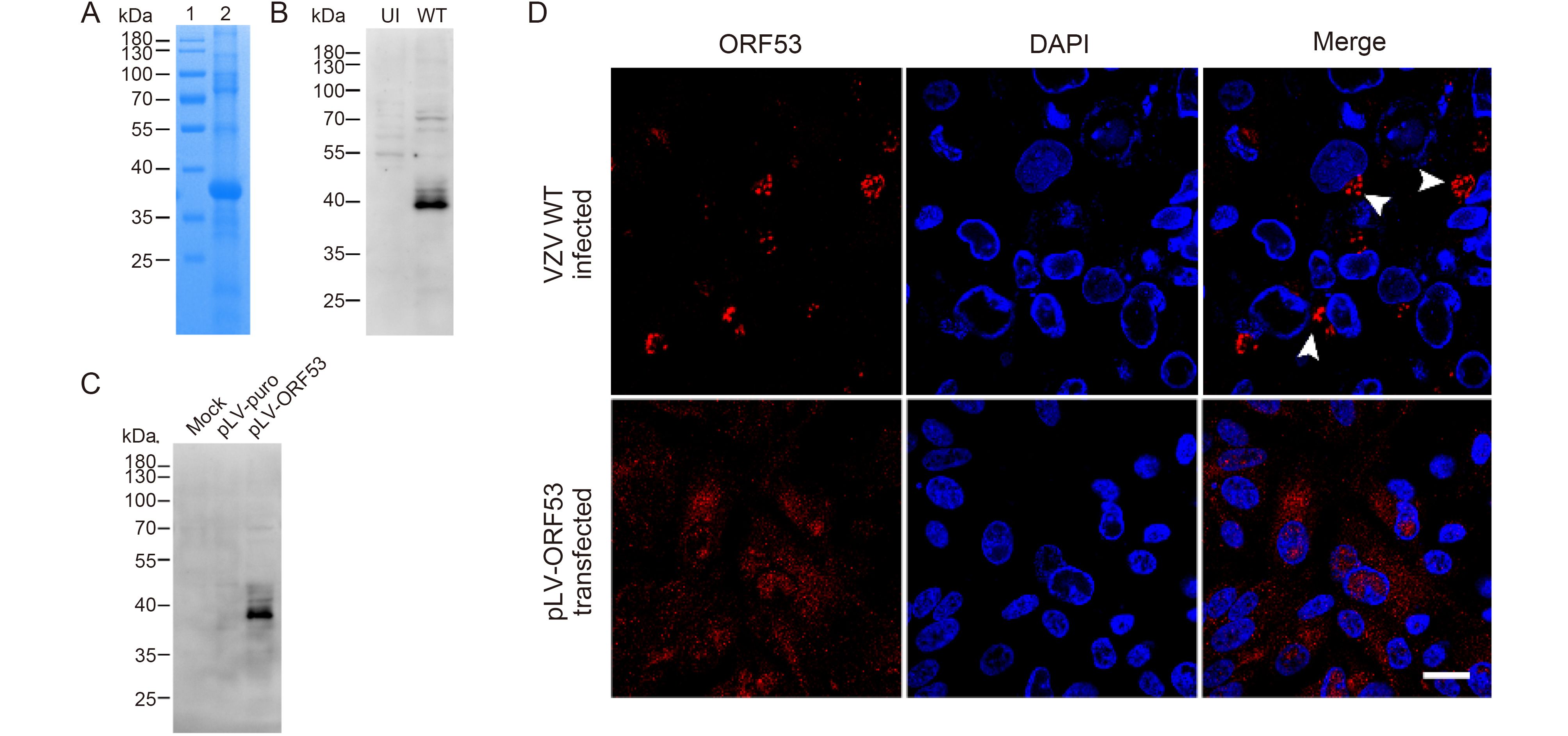
Varicella-zoster virus ORF7 interacts with ORF53 and plays a role in its trans-Golgi network localization
2017, 32(5): 387 doi: 10.1007/s12250-017-4048-x
Received: 28 July 2017 Accepted: 29 September 2017 Published: 30 October 2017Varicella-zoster virus (VZV) is a neurotropic alphaherpesvirus that causes chickenpox and shingles. ORF7 is an important virulence determinant of VZV in both human skin and nerve tissues, however, its specific function and involved molecular mechanism in VZV pathogenesis remain largely elusive. Previous yeast two-hybrid studies on intraviral protein-protein interaction network in herpesviruses have revealed that VZV ORF7 may interact with ORF53, which is a virtually unstudied but essential viral protein. The aim of this study is to identify and characterize VZV ORF53, and to investigate its relationship with ORF7. For this purpose, we prepared monoclonal antibodies against ORF53 and, for the first time, characterized it as a ~40 kDa viral protein predominantly localizing to the trans-Golgi network of the infected host cell. Next, we further confirmed the interaction between ORF7 and ORF53 by co-immunoprecipitation and co-localization studies in both plasmid-transfected and VZV-infected cells. Moreover, interestingly, we found that ORF53 lost its trans-Golgi network localization and became dispersed in the cytoplasm of host cells infected with an ORF7-deleted recombinant VZV, and thus ORF7 seems to play a role in normal subcellular localization of ORF53. Collectively, these results suggested that ORF7 and ORF53 may function as a complex during infection, which may be implicated in VZV pathogenesis. -
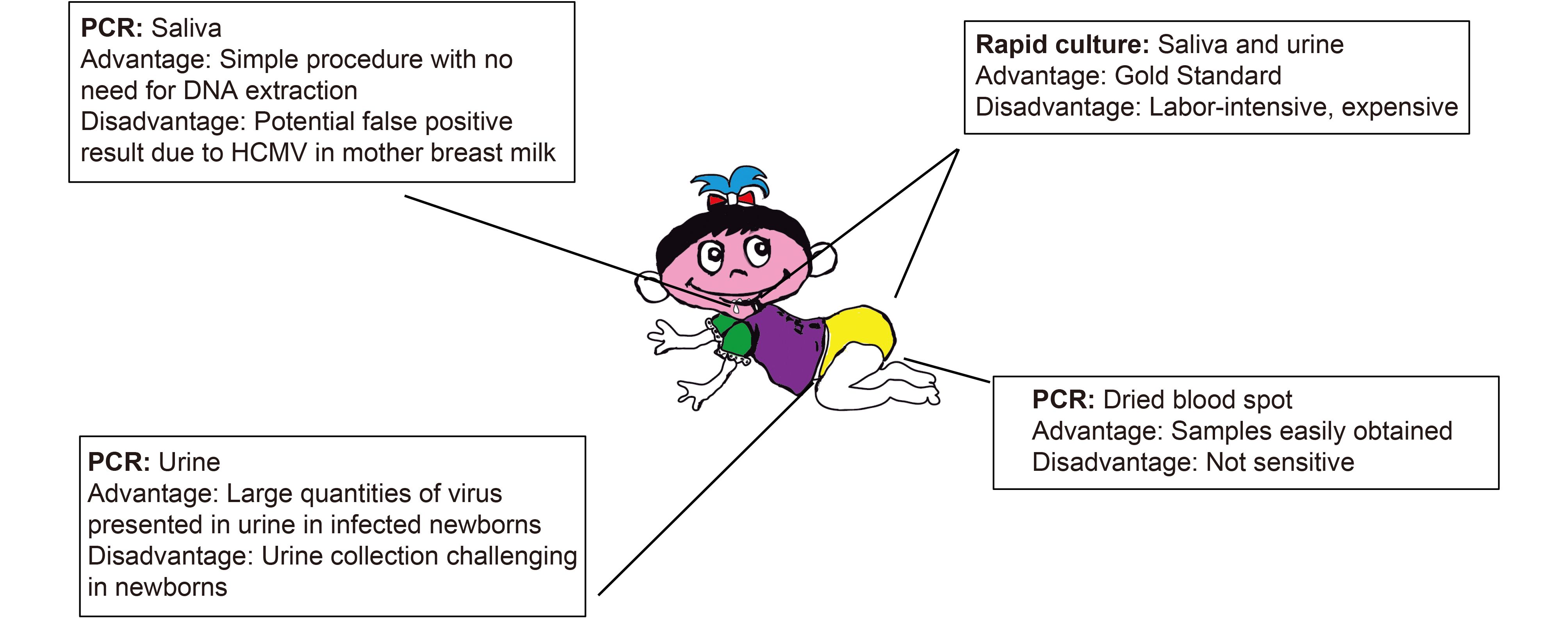
Detection of congenital cytomegalovirus in newborns using nucleic acid amplification techniques and its public health implications
2017, 32(5): 376 doi: 10.1007/s12250-017-4055-y
Received: 02 August 2017 Accepted: 23 October 2017 Published: 30 October 2017Human cytomegalovirus (HCMV), a herpesvirus, is an important human pathogen that causes asymptomatic infections in healthy or immunocompetent individuals but can lead to severe and potentially life-threatening complications in immune-immature individuals such as neonates or immune-compromised patients such as organ-transplant recipients and HIV-positive individuals. Congenital HCMV infection represents a significant public health issue and poses substantial healthcare and economic burden to society. This virus causes the most common viral congenital infection worldwide, and is the leading non-genetic cause of sensorineural hearing loss in children in developed countries. Congenital HCMV infection is believed to fulfill the criteria of the American College of Medical Genetics to be considered as a condition targeted for a newborn screening program. This is because congenital HCMV infection can be identified during a time (within 2 days after birth) at which it would not ordinarily be detected clinically, and there are demonstrated benefits of early detection, timely intervention, and efficacious treatment of the condition. Recent progresses in developing polymerase chain reaction-based approaches to detect HCMV in samples obtained from newborns have generated much excitement in the field. In this review, we highlight the recent progress in diagnostic techniques that could potentially be used for the detection of HCMV infection in neonates and its direct implications in public health settings for diagnosing congenital HCMV infection. -

Lipids, lipid metabolism and Kaposi’s sarcoma-associated herpesvirus pathogenesis
2017, 32(5): 369 doi: 10.1007/s12250-017-4027-2
Received: 02 June 2017 Accepted: 05 September 2017 Published: 10 October 2017Lipids are essential for mammalian cells to maintain many physiological functions. Emerging evidence has shown that cancer cells can develop specific alterations in lipid biosynthesis and metabolism to facilitate their survival and various malignant behaviors. To date, the precise role of cellular lipids and lipid metabolism in viral oncogenesis is still largely unclear with only a handful of literature covering this topic to implicate lipid metabolism in oncogenic virus associated pathogenesis. In this review, we focus on the role of lipid biosynthesis and metabolism in the pathogenesis of the Kaposi’s sarcoma-associated herpesvirus, a common causative factor for cancers arising in the immunocompromised settings. -
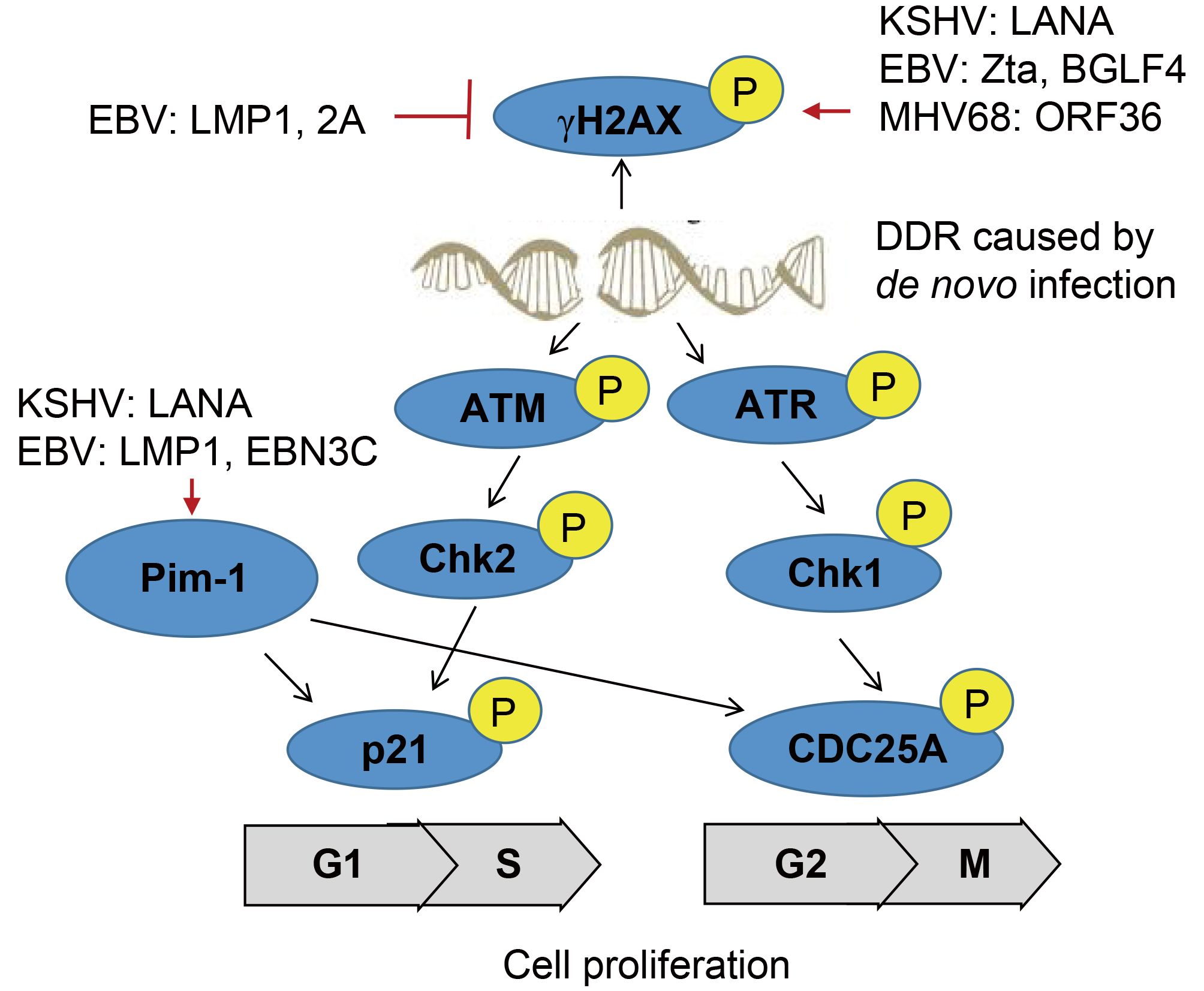
The regulatory role of protein phosphorylation in human gammaherpesvirus associated cancers
2017, 32(5): 357 doi: 10.1007/s12250-017-4081-9
Received: 11 September 2017 Accepted: 23 October 2017 Published: 30 October 2017Activation of specific sets of protein kinases by intracellular signal molecules has become more and more apparent in the past decade. Phosphorylation, one of key posttranslational modification events, is activated by kinase or regulatory protein and is vital for controlling many physiological functions of eukaryotic cells such as cell proliferation, differentiation, malignant transformation, and signal transduction mediated by external stimuli. Moreovers, the reversible modification of phosphorylation and dephosphorylation can result in different features of the target substrate molecules including DNA binding, protein-protein interaction, subcellular location and enzymatic activity, and is often hijacked by viral infection. Epstein-Barr virus (EBV) and Kaposi’s sarcomaassociated herpesvirus (KSHV), two human oncogenic gamma-herpesviruses, are shown to tightly associate with many malignancies. In this review, we summarize the recent progresses on understanding of molecular properties and regulatory modes of cellular and viral proteins phosphorylation influenced by these two tumor viruses, and highlight the potential therapeutic targets and strategies against their related cancers. -
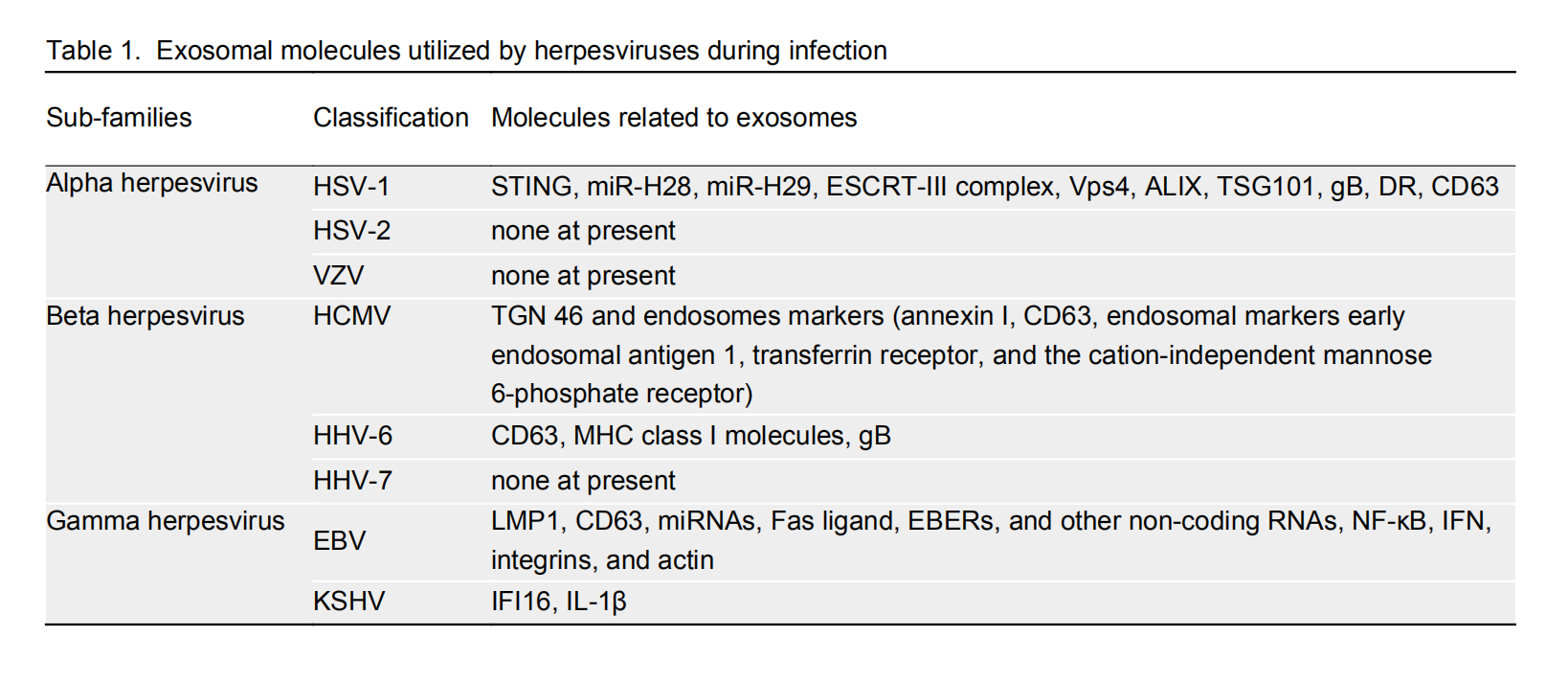
Extracellular vesicles: novel vehicles in herpesvirus infection
2017, 32(5): 349 doi: 10.1007/s12250-017-4073-9
Received: 23 August 2017 Accepted: 09 October 2017 Published: 30 October 2017Herpesviruses are remarkable pathogens that have evolved multiple mechanisms to evade host immunity, ensuring their proliferation and egress. Among these mechanisms, herpesviruses utilize elaborate extracellular vesicles, including exosomes, for the intricate interplay between infected host and recipient cells. Herpesviruses incorporate genome expression products and direct cellular products into exosomal cargoes. These components alter the content and function of exosomes released from donor cells, thus affecting the downstream signalings of recipient cells. In this way, herpesviruses hijack exosomal pathways to ensure their survival and persistence, and exosomes are emerging as critical mediators for virus infection-associated intercellular communication and microenvironment alteration. In this review, the function and effects of exosomes in herpesvirus infection will be discussed, so that we will have a better understanding about the pathogenesis of herpesviruses. -
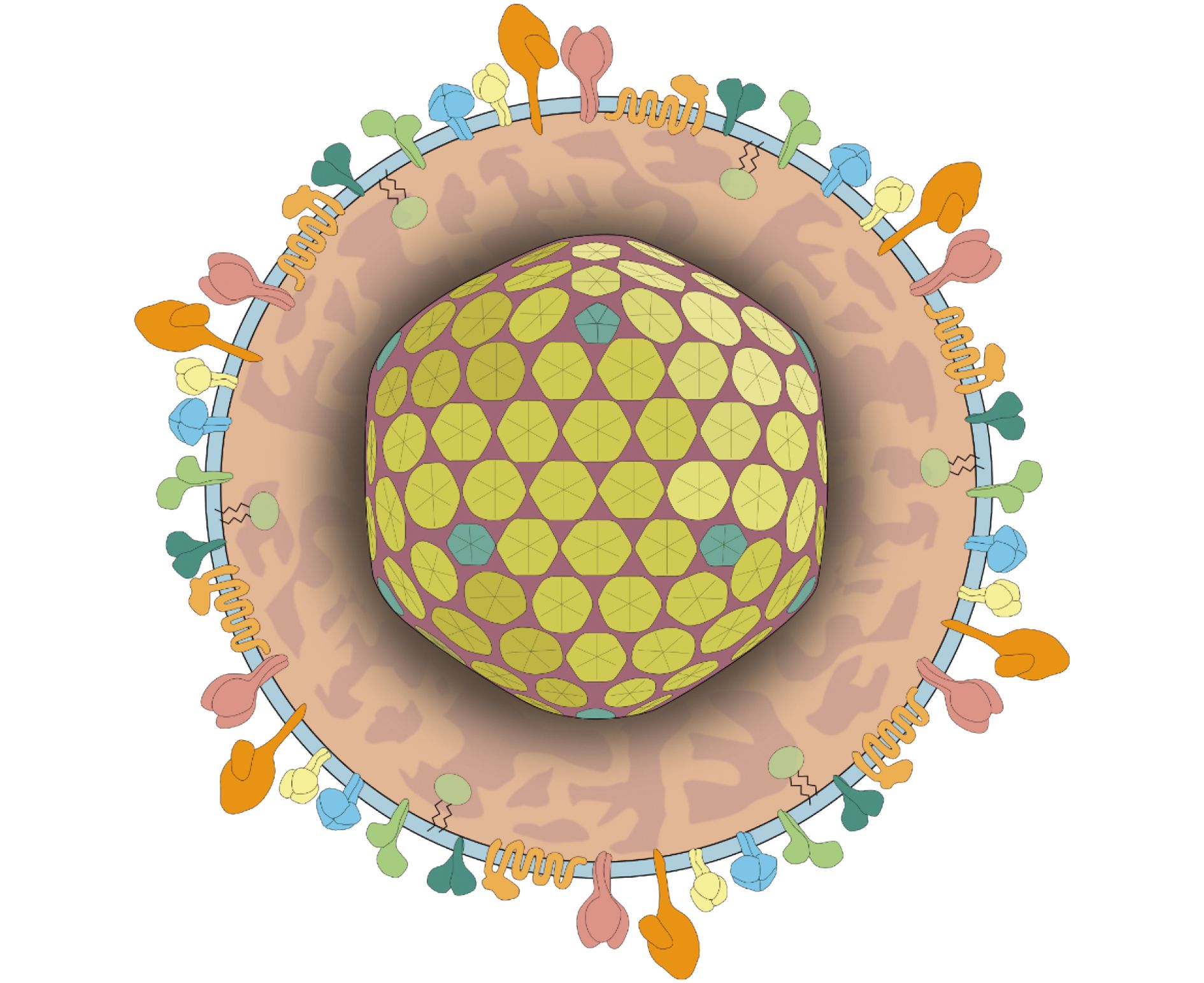
Herpesviruses: epidemiology, pathogenesis, and interventions
2017, 32(5): 347 doi: 10.1007/s12250-017-4108-2
Published: 31 October 2017In this issue, we collectively present ten articles focusing on the epidemiology, pathogenesis, and interventions of HSV-1, VZV, HCMV, EBV and KSHV respectively, and these high-quality review and research articles are contributed by experts on those specific viruses.