-
The replication and assembly of the Reovirus occurs in distinctive cytoplasmic inclusion bodies in infected cells which are called viral factories. These structures are attribute to viral infection, and are believed to be necessary in virion assembly. While the formation mechanism of this structure is unclear, studies have shown that the reovirus nonstructural protein μNS plays a major role throughout viral replication cycles (2, 24).
μNS, which is present in most species of Reovirus, has a relatively conserved coding sequence. The molecular weight of the protein among different reovirus isolates is around 80kDa. Studies of mammalian orthoreoviruses (MRV) indicate that a single μNS, or even a truncated carboxyl-proximal region (4), is sufficient for forming phase-dense inclusions in transfected cells (3, 7). Interestingly, the two forms of μNS, μNS and μNSC, and a proportion of expression between 1: 1 to 4: 1 can be detected in infected cells (25), indicating that μNSC may be important in for-mation of inclusions and the viron assembly process. In addition, the other viral proteins, λ1, λ2, λ3, μ2, σ2, and the nonstructural protein σNS and viral RNA, are all involved in interacting with μNS during virus assembly (5, 6, 20).
Grass carp reovirus(GCRV), the main pathogen that can cause outbreaks of hemorrhagic disease in aquatic animals, belongs to the genus Aquareovirus in the family Reoviridea (1, 19, 22). It has been recognized that GCRV is the most pathogenic agent among all the isolates of aquareoviruses reported to date (21). Similar to other members of Aquareovirus, GCRV, a multilayered spherically structured particle, contains a genome of 11 segmented dsRNA, which encodes 7 structure proteins (VP1-VP7) and 5 nonstructural proteins (9, 27).
While considerable fundamental and applied research has been carried out on mammalian or human isolates from the genus Orthoreovirus and Rotavirus (8, 10, 15, 17, 23, 26), relatively little progress has been made with members of the genus Aquareovirus, and is mainly focused on the molecular and structural biology of their structural proteins. To understand the assembly of GCRV infection in its host cells, it is necessary to investigate the biological function of non-structural proteins involved in viral replication.
According to the complete genome sequence of GCRV and the analyses of the homology alignment, NS80, the nonstructural protein encoded by GCRV s4, has the highest level sequence similarity with the μNS protein in MRV, which has a function in the formation of the viral factory. As a fusogenic reovirus, GCRV has unique replication characteristics and produces large inclusion matrix like CPE during its infection. To understand the role of the μNS80 protein in virus replication, the expression and immunogenicity identification of the GCRV NS80 protein inclusion forming-related region was investigated in this study.
HTML
-
cDNA synthesis kit and PCR reagents used in the experiment were purchased from Invitrogen and Perkin Elmer Co. respectively. The PCR primers were obtained from Saibaisheng Company (Shanghai, China). DNA gel extraction kit and PCR clean-up kit were purchased from Ⅴ-gene biotechnology limited Company (Hangzhou, China). T7 expression system (pRSET vector) with BL21(DE3) pLysS, and ProBond Resin used for protein purification were the products of Invitrogen (Invitrogen, Carlsbad, USA).
-
CIK(Ctenopharyngodon idellus kidney)cells were used for proliferating Grass carp reovirus(GCRV). GCRV 873 was isolated and stored in author's laboratory. The methods relating to cell culture and virus infection and replication were previously described elsewhere (12, 16, 28).
-
The primer targeting the GCRV Segment 4 nucleic acid 5' terminal from 1 028bp to 3' terminal end of the segment named as S4(1028-2229) containing speci fic restriction enzyme digestion sites was designated based on GenBank sequence (AF403390). the sense primer was: 5'-GAAGGATCCTCCCTCCTTACCCTT C-3', the bases underlined are the enzyme digestion site of BamH Ⅰ; the anti-sense primer was: 5'-CAAGAATTC GACACAGAAACACAGAGC-3', the bases underlined are the enzyme digestion site of EcoR Ⅰ. Thermal cycling parameters conditions were as follows : one cycle of denaturation (94℃, 2 min and 30 sec) followed by 35 cycles of denaturation (94℃, 30 sec), annealing (52℃, 1 min and 30 sec) and extension (72℃, 2 min and 30 sec). Final extension step was at 72℃ for 15 min.
-
The PCR amplified GCRV S4(1028-2229) segment was first purified from agarose gels using a DNA gel extraction kit or PCR clean-up kit. Then the gel recovered S4(1028-2229) fragment was ligated to pRSETA vector, the product was named as pR/GCRVS4(1028-2229). The recombination vector was transformed into E. coli DH5α and BL21 (DE3) PLysS cells by the CaCl2 method respectively, and were confirmed by restriction enzyme digestion and PCR amplification as described (11). The recombinant plasmid was also sequenced by Invitrogen Biotechnology Inc. (Shanghai, China).
-
BL21(DE3) PLysS cells with recombinant vector were grown in SOB medium while shaking at 225 r/min at 37℃ for 4 h. Then the culture medium was induced by IPTG in either 37℃ or 28℃ for time course culture. Bacteria were collected at different time of induction and lysed. Supernatant or sediment of cell lysate was re-suspended and boiled for 5 min. The samples were then loaded on a SDS-PAGE gel for electrophoresing as described elsewhere (13). Proteins were visualized by Coomassie brilliant blue R-250 (Sigma, USA).
-
To gain purified recombinant NS80(335-742) protein, the identified recombinant vector in BL21(DE3) PLysS cells induced by IPTG were cultured at 28℃ for 4 hrs. The cultured cells were collected and lysed with guanidinium lysis buffer. As the recombinant proteins contain a His-tag region in its N-terminal, ProBond resin was used to purify the protein following the ProBond Purification System Kit Instructions.
-
Antiserum against expressed recombinant NS80(335-742) Protein was prepared by subcutaneously injecting New Zealand white rabbits with a mixed emulsion of 200 μL of purified stock virus solution (0.5μg/μL) and equal volume of Freund's complete aduvant (FCA), followed by an intramuscular booster injection with an equal volume of a mixture of purified virus and incomplete Freund's adjuvant two weeks later. After another week, a final booster injection consisting of purified virus mixture was administered intramuscularly. Three days later, the rabbits were bled, and sera were separated and stored at –30℃. The titer of antibody was determined using the ELISA.
-
His-tag antibodies (mouse) and rabbit anti-NS80(335-742) serum were used to detect expression of recombinant protein, which contain a his-tag region in the N-terminal, by using western blot analysis. Briefly, BL21(DE3) PLysS cells with recombinant vector or infected CIK cell were lysed and subjected to 10% SDS-PAGE. Semi-dry transfer cell was used following the instrument's instruction. His-tag antibodies or rabbit anti-NS(335-7427) serum were used (mouse) as first antibody and goat anti-rabbit or mouse IgG as second antibody (1: 2 000 concentration). The result was observed by developing with AP substrate solution (NBT/BCIP).
Reagents
Cell and virus
Gene amplification
Construction and identification of recombinant plasmid
Expression and analysis of recombinant protein by SDS-PAGE
Purification of recombinant protein NS80(335-742)
Preparation of anti-NS80(335-742) serum
Western blotting analysis
-
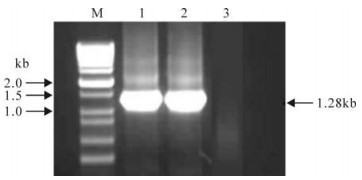
In order to obtain the gene of interest for the GCRV segment4 carboxyl terminal, named as S4(1028-2229), RT-PCR was performed by using the purified GCRV whole genome as template and the selected primer pairs as indicated in the Material and Methods section. As shown in Fig. 1, the amplified products of interest are around 1.3 kb, which corresponds to the predicted value.
-
We chose the pRSET-A vector for constructing a recombinant plasmid with replication in DH5α and expression in BL21(DE3) pLysS since the vector contains a T7 promoter and the recombinant protein would thus obtain a polyhistidine at the N-terminal to allow further purification. For molecular cloning, the amplified S4(1028-2229) fragment and pRSET-A vector were digested with BamH Ⅰ and EcoR Ⅰ respectively, and then ligated by T4 DNA ligase. We named the recombinant as pR/GCRV-S4(1028-2229). The positive plasmid was first identified by both restriction enzyme digestion and PCR amplification (Fig. 2), the values correspond well with their predicted size, which is also confirmed by sequence analysis (data not shown).
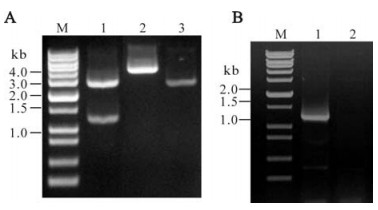
Figure 2. Identification of recombinant plasmid. A: Analysis of recombinant plasmid by enzyme digestion, M, 1 kb plus DNA ladder marker; 1, Double enzyme digested recombinant plasmid with BamH Ⅰ+EcoR Ⅰ; 2, Single enzyme digested recombinant plasmid with EcoR Ⅰ.; 3, Single enzyme digested pRSET A plasmid with EcoR Ⅰ. B: Analysis of recombinant plasmid by PCR amplification, M, 1 kb plus DNA ladder marker; 1, Positive amplification of PCR by using as template; 2, Negative control
-
To investigate the in frame expression of the constructed recombinant, the identified positive pR/GCRV-S4(1028-2229) plasmid was transformed into E. coli BL21(DE3) pLysS cells for further analysis. Since the protein of interest is a nonstructural protein and related to aquareovirus replication and assembly, we set up both 37℃ and 28 ℃ conditions for growing the recombinant bacteria. The recombinant protein was expressed in E. coli and induced by 1 nmol/L IPTG for 3 h as shown in Fig. 3, and the expression level induced by 1 nmol/L IPTG was found to be much higher at 28℃ than at 37℃. It appeared there is over-expression presented in cell lysate pellet, when conducted at 28℃ cultivation. The molecular weight of the fusion protein of interest is about 50 kDa, which is consistent with the predicted value. In addition, there is no obvious band appearing in the cell lysate supernatant samples at both 37℃ and 28℃ culture condition, suggesting the over-expressed protein is in an insoluble form.
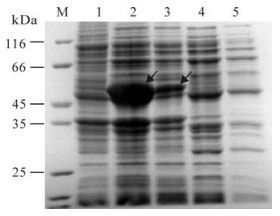
Figure 3. Expression of GCRV NS80(335-742)at different temperatures. M, Standard protein marker; 1, pRSET empty vector as negative control induced for 3h by IPTG; 2 and 4, Recombinant NS80(335-742) protein expression cell lysate pellet and supernatant induced for 3h by IPTG at 28℃; 3 and 5 Recombinant NS80(335-742) protein expression cell lysate pellet and supernatant induced 3h by IPTG at 37℃. Diagonal arrows indicate induced fusion protein products.
-
To further optimize expression condition, a time course expression of recombinant fusion protein was performed. As observed above, the expression yield is much lower at 37℃ conditions, so the time course expression of recombinant fusion protein was conducted at 28℃, and induced by using 1 nmol/L IPTG at 0, 1, 2, 3, 4 and 5 h, respectively. SDS-PAGE analysis revealed the level of expression increased as time progressed, peaking at about 4 h cultivation and appeared in a precipitated form in the cell lysate, suggesting the over-expressed protein accumulated as inclusion bodies in E.coli.
To provide further confirmation the identity of the expressed protein was the his-tag fusion protein, the identity of the expressed NS80(335-742) protein was checked by Western blotting analysis. It was found that throughout the entire time course experiment the expressed protein from the cell lysate pellet was able to bind immunologically to monoclonal mouse anti-Histag serum, with no visible cross reaction band observed with cell lysate supernatant sample (Fig. 4B). This suggests that the over-expressed fusion protein was in the precipitated form, and the recombinant protein was the his-tag fusion protein as expected.

Figure 4. SDS-PAGE analysis of time course expression of NS80(335-742) and Western blotting identification. A: SDS-PAGE analysis of time course expression. M, Standard protein marker; Lane 1, pRSET empty vector as negative control induced 3h by IPTG; 2, Recombinant expression NS80(335-742) protein cell lysate supernatant induced 3h by IPTG; 3-8, Recombinant expression NS80(335-742) protein cell lysate pellets induced by IPTG in 0, 1, 2, 3, 4, 5 h respectively. B: Western blot analysis of time course expression corresponding to above protein samples (1-8) with His-Tag monoclonal antibody.
-
To carry out functional assays, it was necessary to get the purified protein of interest. As indicated above there are 6 tandem histidine residues at the N-terminal peptide in pRSET vector, which have a high affinity for probondresin, so we selected probondresin for protein purification. We tried both native and denaturing conditions for the expressed protein purification, but found that the yield of purified protein was much higher by using the denaturing condition (shown in Fig. 5A.), which was is consistent with the observation that the expressed protein is in an inclusion body formation.
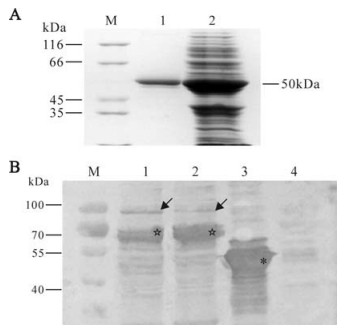
Figure 5. Purification and western blot analysis of NS80(335-742) expression. A: SDS-PAGE. M, Standard protein marker; 1, NS80(335-742)protein after purification; 2, Unpurified expression lysate of protein NS80(335-742). B: Western Blot. M, Prestained standard protein marker; 1 and 2, Cytoplastic lysate of CIK cell infected with GCRV at 16 h and 24h p.i.; 3, Expressed NS80(335-742) protein; 4, Cytoplastic lysate of uninfected CIK cell Diagonal arrows indicate NS80 protein. *50kDa expressed protein as positive control. ☆: Possible NS80C, a another predicted form of NS80 in GCRV infected cells.
To further verify whether prepared rabbit antiserum is immunologically specific to expressed NS80(335-742), and able to bind to nonstructural protein produced during GCRV replication in infected cell, we analysed cytoplasmic lysate of CIK cells infected with GCRV at 16h and 24 h respectively. Western blotting analysis indicate the prepared antibody is specific to expressed protein NS80(335-742). Additionally, nonstructural NS80 protein expression could be recognized in immunoblots by polyclonal rabbit antiserum that we raised against NS80(335-742). Furthermore, it appeared there was not any immune reaction across the normal cell lysate as shown in Fig. 5B, suggesting that expressed NS80(335-742) possesses good antigenicity, and could be used for detecting NS80 expression in GCRV infected cells.
Generation of GCRV S4 (1028-2229) fragment
Construction of recombinant plasmid
Expression of recombinant fusion protein in E.coli
Time course expression and western blot analysis
Purification and specificity analysis of expressed protein NS80(335-742)
-
To date, many studies have shown that nonstructural proteins of reovirus play an important role in viral replication. The viral nonstructural protein uNS of MRV, encoded by genome segment M3, is expressed to high levels in infected cells and is concentrated in the infected cell factory matrices called viral inclusions. Though the homologous protein of μNS in MRV and biological function of μNS have been well studied, the nonstructural protein μNS80 in GCRV has not been characterized. Our initial study of the expression and identification of the inclusion forming-related region of the NS80 nonstructural protein of GCRV in Prokaryotic cells provides a basis for further studies on understanding the mechanism of this protein as well as formation of viral factory.
It should be mentioned that the expression of nonstructural protein NS80 could be detected with a visible band in GCRV infected cell lysate at 16 h and 24 h by polyclonal rabbit anti-NS80(335-742) serum(as shown in Fig. 5B), and the molecular weight in virus infected cell lysate appears to be a little bit larger than the 80kDa observed in the immunoblotting map. But since it corresponds to the migrated size of uNS in MRV infected cytoplamic lysate (6), we suppose the difference may be caused by mobility issues with DSD-PAGE electrophoresis, due to formation of a large uNS-core or uNS-RNA complex since it is reported that uNS of MRV in infected cells have extensive interactions with core particles or RNA genomes during virus genome transcription or particle assembly (6).
Another form of μNS exists in MRV, which lacks about 5 kDa from its N terminus and is called μNSC, could also be detected in infected cells (25). Previous experiments indicate that the protein is supposed to be a translation product of the M3 segment from the second AUG at Met41 of the μNS sequence, but is not a digested product of μNS (23, 25). The role of μNSC has not been determined because there are reports that indicate expression of μNSC is neither necessary nor sufficient to support MRV growth in infected cells (4, 18). In this study, we detected an obvious immunoblotting band with a size around 70 kDa in the GCRV infected cell lysate, which might be a μNSC form a third-AUG transcriptional product based on the value of molecular weight, or it might just be the degraded form of NS80 after viral infection. It is worth noting that the proportion of μNS and μNSC is between 1: 1 and 4: 1 in MRV infected cells, however, it seems in reverse as the detail showed in Fig. 5B. The phenomenon might be caused by μNS degradation after cell lysis, or perhaps it is a unique characteristic of the GCRV infection process which warrants further attention. Further experiments involving detailed time course infection with the virus should provide a greater insight.
In summary, we have expressed the GCRV NS80(335-742) protein in prokaryotic cells for the first time. The recombinant fusion protein is over-expressed, and appears in the formation of an inclusion body, and is immunologically related to the NS80 protein expressed in GCRV infected cells. Generally, the nonstructural protein is not a component of the mature virion, only playing a role in virus genome replication and virion assembly. Interestingly, we obtained over expressed protein at 28℃ in E.coli rather than at 37℃, which is consistent with the report that GCRV replicates well in the CIK (Ctenopharyngodon idellus kidney) cell line at 25 -30℃ (12, 28). To further investigate the role of NS80 played in virus replication and assembly, our present results will provide an experimental foundation and method for construction of a series of recombinant NS80 proteins which can be used for detailed analysis of the mechanisms.













 DownLoad:
DownLoad: