HTML
-
Viruses have evolved a variety of strategies to avoid the host innate immune response, including by blocking the production of interferons (IFNs) and IFN-stimulated genes. IFNs are mainly divided into three types. Type Ⅰ IFN including IFN-α and IFN-β is involved in viral clearance through the induction of an immune response and plays a protective role against acute viral infection. Type Ⅱ IFN, primarily produced by activated NK cells and T cells, plays a key role in both innate and adaptive immunity (Goodbourn et al. 2000). IFN-λ (type Ⅲ IFN) is related to the antiviral immune response on the epithelial surface and the receptor for IFN-λ is most abundantly expressed on epithelial-derived cells (Sommereyns et al. 2008; Zhou et al. 2018).
Several lines of evidence demonstrate that type Ⅰ IFN also exerts immunomodulatory effects, including regulating the activities of other cytokines (Biron 2001), enhancing the maturation and activation of dendritic cells (Radvanyi et al. 1999; Santini et al. 2000), promoting the Th1-type response (McBride et al. 2006; Yoo et al. 2010), and activating B cells to promote an antibody response (Le Bon et al. 2001; Braun et al. 2002). The pleiotropic effects of type Ⅰ IFN allow it to act as an essential component of such responses, linking the innate and adaptive immune systems. Further, IFN-α enhances chemokine production by influenza A virus-infected cells (Veckman et al. 2006). The expression of chemokines and cytokines can also amplify the initial response to the virus, leading to the recruitment of immune cells that induce adaptive immunity. The chemokine (C–C motif) ligands (CCLs) CCL25 and CCL28 play essential roles in mucosal immunity and mediate the migration of immune cells to the intestinal tract (Zaballos et al. 1999; Gosling et al. 2000; Lazarus et al. 2003). In addition, CCL25 recruits CCR9+ T cells and CCL28 recruits CCR10+ IgA plasmablasts to the gastrointestinal tract.
The influenza A virus (IAV) can cause respiratory tract disease characterized by respiratory symptoms such as fever and dyspnea in pigs, and is highly prevalent in pig herds. Even if the mortality rate is lower, it still causes considerable economic losses of the pig industry worldwide due to the weight loss in infected pigs and abortion in sows (Myers et al. 2007). In addition, it is called ''mixing vessel'' of IAV and are considered to be important intermediates for gene reassortment and cross-species transmission, which may cause the emergence of human influenza pandemic strains (Scholtissek et al. 1985). IAV is mainly recognized by the RIG-I-like receptor of host cells upon infection, resulting in the stimulation of type Ⅰ IFN expression, triggering an antiviral immune response cascade in the host (Eisfeld et al. 2015). Because IAV invades through the respiratory tract of the host, intranasal administration of vaccines can establish an immune defense at the invasion site of the respiratory mucosa, thus effectively inducing both the secretion of mucosal IgA and a systemic immune response (Chiou et al. 2009).
Type Ⅰ IFN can be used in combination with influenza vaccines to immunize mice via intranasal administration, and this acts as a powerful adjuvant to induce an effective humoral response, preventing both influenza virus infection and weight loss of the host (Bracci et al. 2005). All currently available commercial swine influenza vaccines are based on inactivated viruses; upon intramuscular injection, these can stimulate the production of antibodies, but are associated with little mucosal antibody production (Rajao et al. 2014).
Here, porcine IFN α (PoIFNα) was cloned, expressed, purified, and subsequently combined with inactivated influenza virus to immunize 6-week-old pigs via intranasal administration. By further studies, we explored the possibility that PoIFNα regulates the development of an immune response to the vaccine/IFN combination, by monitoring clinical characteristics of the pigs, production of antibodies, and gene expression at the transcriptional level.
-
MDCK, MDBK, PK15, and WISH cell lines were grown in Dulbecco's modified Eagle's medium (GIBCO) supplemented with 10% fetal bovine serum (GIBCO) at 37 ℃ in an atmosphere of air containing 5% CO2. The New Jersey serotype of VSV was purchased from the Harbin Veterinary Research Institute, Chinese Academy of Agricultural Sciences. The A/JILIN/CAS01/2009 (H1N1) (CAS01) strains were stored in our laboratory and propagated in 10-day-old specific pathogen-free (SPF) chicken embryonated eggs. All viruses were grown in MDCK cells for 72 h at 37 ℃. Viral titers were determined based on plaque assays.
-
Pig anti-H1N1-positive and anti-H1N1-negative serum samples were kind gifts from Professor Jinhua Liu (College of Animal Medicine, China Agricultural University, Beijing, China). HRP-conjugated anti-swine IgG was purchased from Solarbio and TRITC-conjugated anti-swine IgG was purchased from Jackson ImmunoResearch.
-
Protein expression and purification was performed as previously described with minor modifications (Meng et al. 2011). E. coli strain Rosetta (DE3) was transformed with the recombinant expression plasmid pET-His-PoIFNα and a single colony was cultured in LB medium at 37 ℃ until the OD600 reached 0.5. Protein production was then induced with 1 mmol/L IPTG for 5 h at 37 ℃. The cells were collected by centrifugation and precipitations were resuspended in PBS for sonication, and then inclusion bodies were isolated by centrifugation at 4 ℃ and 10, 000 rpm for 10 min, and recombinant PoIFNα purified by gel filtration. Purity was assessed by SDS-PAGE and Western blotting and concentrations of the recombinant protein were determined using a BCA protein assay kit (CW Bio) according to the manufacturer's instructions. The recombinant PoIFNα was treated to remove LPS using a ToxinEraserTM Endotoxin Removal Kit (GenScript), following the manufacturer's directions; it was then diluted and filtered through a 0.22-μm filter and stored at 4 ℃.
-
The antiviral activity of PoIFNα was determined based on the cytopathic effect (CPE) inhibition method, using the VSV/MDCK, VSV/MDBK, VSV/PK15, and VSV/WISH systems, according to previously described protocols (Vogel et al. 2001). In brief, cells were seeded in 96-well plates at a density of 104 cells per well and cultured at 37 ℃ in humid air containing 5% CO2 for 6 h, until they reached confluence. The cells were then stimulated with 100 μL of tenfold serially-diluted PoIFNα for 12 h and then washed and challenged with 100 TCID50 virions per well. The positive control contained virus without PoIFNα, whereas the negative control lacked virus and PoIFNα. All cells were cultured until the CPE of the positive control was apparent. Cultures were stained with crystal violet and the optical density at 570 nm (OD570) was recorded using a Microplate Reader (Thermo Scientific). PoIFNα titers (U/mg) are expressed as the reciprocal of the dilutions that led to 50% virus-induced cell lysis, as determined by the Reed–Muench method.
-
Twenty 6-week-old pigs were obtained from the Beijing Centre for SPF Swine Breeding and Management. All animals tested negative for anti-IAV antibodies, as determined by a commercial ELISA kit (Influenza A Ab Test, IDEXX). The pigs also tested negative for porcine reproductive and respiratory syndrome virus, classical swine fever virus, porcine circovirus type 2, pseudorabies virus, and IAV by PCR and/or RT-PCR. Specific primers for the detection are described in previous study (Ogawa et al. 2009; Lung et al. 2012; Hu et al. 2015). Pigs were randomly separated into five groups (four pigs per group) and housed at Sinder Technology Co., Beijing. Pigs were immunized via intranasal administration according to Supplementary Table S1. EDTA-stabilized blood samples were collected for the isolation of peripheral blood mononuclear cells (PBMCs), which were then used to determine cytokine mRNA expression profiles at 0, 4, 12 and 24 h post-immunization (hpi). Sera separated from the blood at 0, 7, 14 and 21 days post-injection (dpi) were analyzed by ELISA to assess the HA-specific IgG response (as indicated in a subsequent section).
The vaccinated pigs were intranasally inoculated with 106 TCID50 CAS01 at 21 dpi. Rectal temperatures and clinical signs were recorded daily during the experiment for each pig. Additionally, the severity of the clinical signs was evaluated as previously described (Chen et al. 2013), based on a point-scale that independently considers attitude (0–4 points), respiratory rate (0–2 points), respiratory distress (0–3 points), and coughing (0–1 point).
-
From each pig, 5 mL of EDTA-stabilized blood was collected. PBMCs were isolated using a Pig Peripheral Blood Lymphocyte Separation Kit (Solarbio) according to the manufacturer's instructions. Total RNA was extracted from PBMCs and reverse-transcribed into cDNA as previously described (Qu et al. 2013). qRT-PCR was performed using an Applied Biosystems 7500 Real-Time PCR System and TB Green Advantage qPCR Premix (Takara) following the manufacturer's qPCR Protocol. Three independent experiments were conducted in duplicate and mRNA levels were calculated using the 2-△△Ct method. cDNA from PBS group cells was used as a calibrator (set to 1) to evaluate the mRNA levels of genes encoding CC chemokine receptors (CCRs) and cytokines; β-actin was used as an internal control. The primers for qRT-PCR are listed in Supplementary Table S2 (Meurens et al. 2006; Dawson et al. 2005; Lee and Lee 2012; Duvigneau et al. 2005).
-
The recombinant plasmid pET32a-HA was constructed based on the sequence of H1N1 HA (GenBank Accession No. EU516079.1). H1N1 HA-specific antibody responses were determined using an indirect ELISA with purified recombinant HA proteins. Ninety-six-well ELISA plates were coated with 0.4 μg of recombinant HA protein/well diluted in 100 μL of 50 mmol/L bicarbonate buffer (pH 9.6) overnight at 4 ℃ after incubation for 1 h at 37 ℃. The plates were washed three times with PBS containing 0.05% tween-20 (PBST) and blocked with 3% bovine serum albumin in PBS for 1 h at 37 ℃. After washing three times, serum samples were diluted 1:50 in dilution buffer (1% BSA in PBST), and the plates were loaded with 100 μL of diluted samples and negative and positive controls (in triplicate). Plates were incubated for 1 h at 37 ℃ and then rinsed five times before the addition of 100 μL of horse radish peroxidase-conjugated goat anti-swine IgG antibody diluted 1:4000 in dilution buffer, after which samples were incubated for 1 h at 37 ℃. Plates were then rinsed five times and 100 μL of TMB substrate solution (Tiangen) was added for 10 min at room temperature (25 ℃) in the dark. Color development was stopped by the addition of 2 mol/L H2SO4, and the optical density at 450 nm (OD450) was recorded using a Microplate Reader (Thermo Scientific). Serum IgG responses are represented as sample-to-positive ratios calculated based on the following formula: S/P ratio = (sample OD - negative control mean)/(positive control mean - negative control mean).
-
PK15 cells grown on coverslips in 24-well cell culture plates at a concentration of 104 per well were infected with H1N1 (MOI = 0.001) for 12 h. After washing with PBS, the cells were fixed with 4% paraformaldehyde overnight at 4 ℃ and then permeabilized with PBST (0.5% triton X-100 in PBS), which was followed by incubation with blocking buffer (4% BSA in PBST) for 1 h at 37 ℃. The cells were then incubated with pig antisera from the four groups at 21 dpi diluted 1:100 in blocking buffer for 1 h at 37 ℃. After washing with PBST for 50 min, the cells were incubated with TRITC-conjugated anti-mouse IgG antibody diluted 1:200 in blocking buffer for 1 h at 37 ℃ and washed with PBST for 50 min. Cell nuclei were stained with DAPI for 20 min and observed under a confocal laser scanning fluorescence microscope (Olympus LSCMFV500).
-
Statistical analyses were performed using SPSS Statistics v17.0 (SPSS Inc., Chicago, IL). Comparisons among different groups were conducted using the Duncan's multiple range tests and P-values < 0.05 were considered statistically significant.
Virus and Cells
Sera and Antibodies
Expression and Purification of Recombinant PoIFNα
In vitro PoIFNα Antiviral Activity Assay
Pig Immunization and IAV Challenge
Quantitative Real-Time PCR (qRT-PCR) Assay
Serum HA-Specific Antibody Indirect ELISAs
Indirect Immunofluorescence Assay (IFA)
Statistical Analysis
-
The PoIFNα gene was amplified from the cDNA of porcine PBMCs, as previously described (Meng et al. 2011). The protein was highly expressed in the form of inclusion bodies, and SDS-PAGE revealed a major band with an apparent molecular weight of 25 kDa (Fig. 1A) that could be detected by Western blotting using an anti-His antibody (Fig. 1B). The recombinant protein was purified by gel filtration and identified by SDS-PAGE (Fig. 1C). The purified inclusion bodies were then renatured to obtain the biologically active protein.
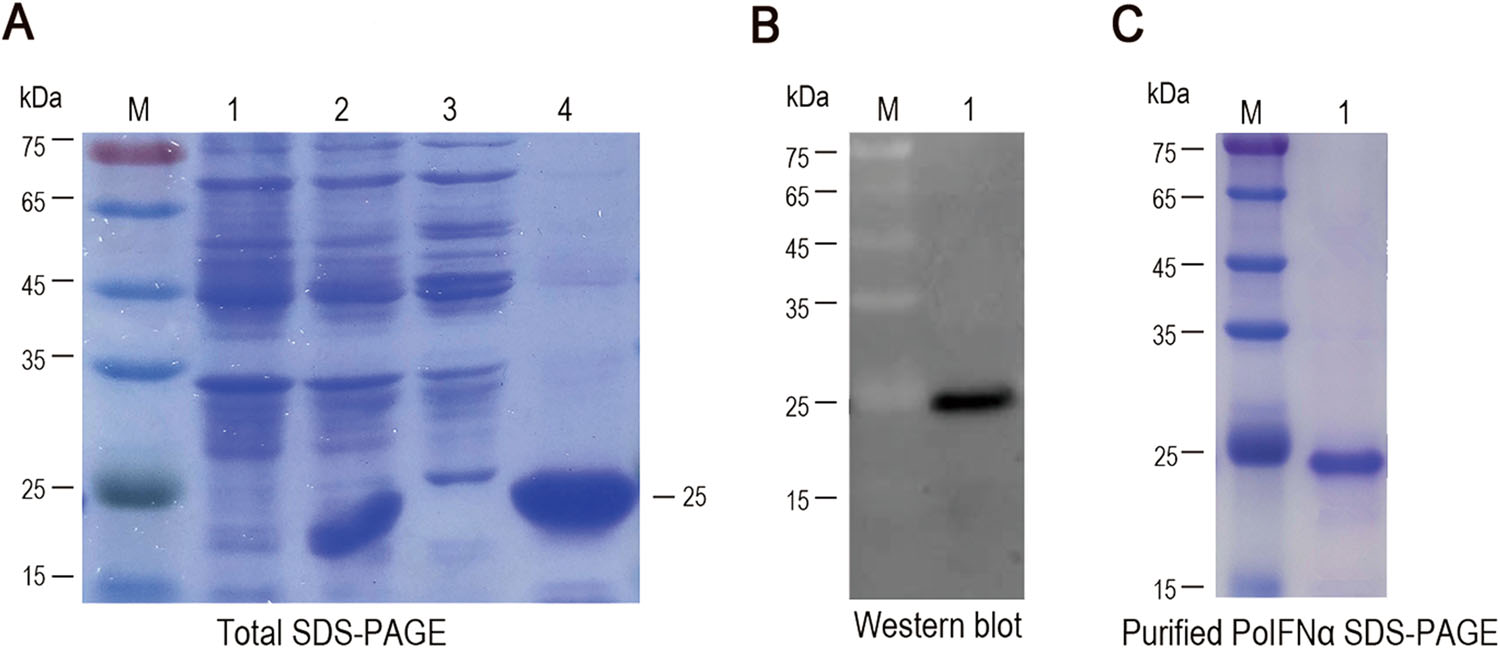
Figure 1. Analysis of recombinant porcine interferon α (PoIFNα). A SDS-PAGE analysis of PoIFNα. Lane M, molecular weight marker; lanes 1–2, total protein of E. coli Rosetta (DE3) cells containing pET-His before and after IPTG induction, respectively; lanes 3–4, supernatant and pellet from induced E. coli lysate, respectively, after centrifugation. B PoIFNα was analyzed by Western blotting. C SDS-PAGE analysis of purified PoIFNα.
-
To identify the effect of purified recombinant PoIFNα, anti-vesicular stomatitis virus (VSV) activity was assessed based on CPE inhibition assays using MDCK, MDBK, PK15 and WISH cells. PoIFNα displayed antiviral activity in all cell lines and the data were analyzed using the Reed– Muench method. When assayed in parallel, the anti-VSV titer of PoIFNα reached 2.9 × 109 U/mg in MDBK cells, for which this treatment was the most effective (Supplementary Table S3).
-
IFNα enhances the transcription of genes encoding cytokines and chemokines in vivo (Mamber et al. 2016). To confirm the role of IFN in the immune response induced by intranasal immunization, qRT-PCR was performed for selected genes. The mRNA levels of the CCRs CCR9 and CCR10 and the immune regulatory cytokines IL2, IL-18, IFN-γ, IL-6, and IL-10 were measured in peripheral blood lymphocytes. cDNA from PBS group cells was used for calibration (set as 1) to evaluate the mRNA levels of CCRs and cytokines; β-actin was used as an internal control.
The secretion of IgA is closely related to the homing of specific B lymphocytes, and the chemokine receptors CCR9 and CCR10 are homing signal molecules unique to IgA-secreting B lymphocytes (Feng et al. 2006). Compared to those in the PBS group, CCR mRNA levels peaked at 12 hpi and decreased slightly at 24 hpi, whereas no differences were found at 4 hpi. Compared to those in the PoIFNα and H1N1 groups, CCR9 and CCR10 mRNA levels were significantly up-regulated at 12 and 24 hpi in the low-dose PoIFNα/H1N1 (LIH) group (P < 0.001). CCR9 mRNA in the LIH group increased by 190-fold at 12 hpi, whereas CCR10 increased by nearly 18-fold (Fig. 2A).

Figure 2. Gene expression in peripheral blood mononuclear cells (PBMCs) from the PoIFNα- and/or H1N1 influenza virus-immunized pigs. PBMCs were harvested from PoIFNα- and/or H1N1 influenza virus-immunized pigs. The expression of genes encoding C–C chemokine receptor (A), cytokine (B), was assayed by qRT-PCR. β-actin was used as an internal control. Fold-differences were determined using the 2-△△Ct method. Data are the mean ± SEM (n = 3). Data were analyzed by one-way ANOVA, and comparisons of means were conducted using the Duncan's multiple range test. Statistically significant differences are indicated (*P < 0.05; **P < 0.001). Groups included PBS, recombinant porcine interferon α (PoIFNα), H1N1 influenza virus-immunized (H1N1), and low-dose PoIFNα and H1N1 (LIH).
Cytokines such as IL2, IL-18, IFN-γ, IL-6, and IL-10 play an important role in the development of innate responses and the induction of adaptive responses (Striz et al. 2014). As shown in Fig. 2B, the expression of IL-2, IL-18, and IFN-γ genes in the LIH group increased significantly as compared to that in the other groups at 12 hpi (P < 0.001) and IFN-γ was up-regulated by approximately 25-fold. Similar to that observed for CCR expression, IL-6 and IL-10 levels increased significantly in the LIH group compared to those in the other groups at 12 and 24 hpi (P < 0.001 or P < 0.05). These results indicate that lowdose PoIFNα and H1N1 can enhance the expression of immunoregulatory cytokines.
-
To determine the optimal dosage of IFN, we further evaluated mRNA levels of cytokines and CCRs in the LIH and high-dose PoIFNα/H1N1 (HIH) groups at 12 hpi. As shown in Fig. 3, these levels were up-regulated in both groups. Interestingly, CCR and cytokine mRNA levels in the LIH group were significantly increased compared to those in the HIH group (P < 0.05). In addition, the increase in CCR9 levels was significantly higher (~10-fold) compared to that for CCR10 (Fig. 3A). With respect to cytokine expression, IFN-γ was the most up-regulated, followed by IL-6 and IL-10, whereas the up-regulation of IL2 and IL-18 was relatively weak (Fig. 3B).
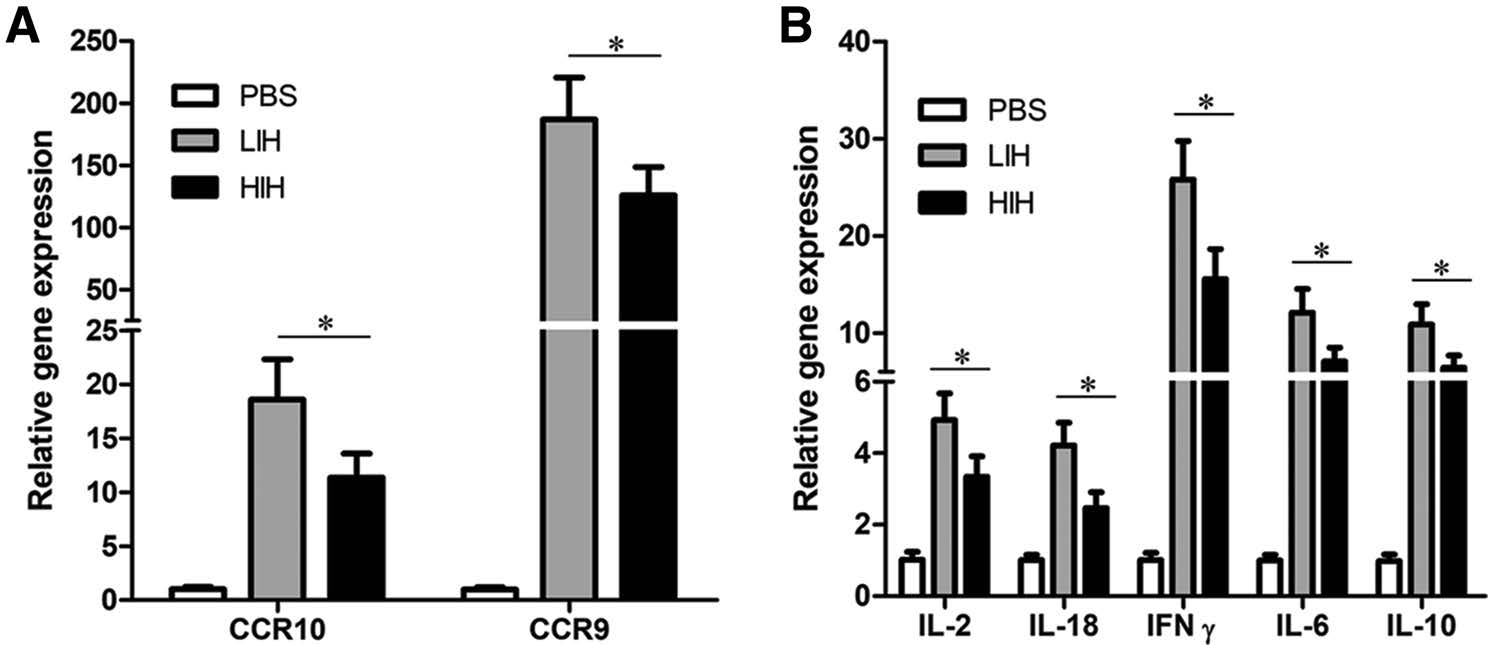
Figure 3. PoIFNα dose-dependent induction of CCR and cytokine mRNA expression. Peripheral blood mononuclear cells (PBMCs) were harvested 12 h post-inoculation for RNA extraction and cDNA preparation. The RNA levels of CCR9 and CCR10 (A), as well as IL2, IL18, IFN-γ, IL6, and IL10 (B) were assayed by real-time PCR. Data are the mean ± SEM (n = 3). Data were analyzed by one-way ANOVA, and comparisons of means were conducted using the Duncan's multiple range test. Statistically significant differences are indicated (*P < 0.05). β-actin was used as an internal control. Groups included PBS, low-dose PoIFNα and H1N1 (LIH), and high-dose PoIFNα and H1N1 (HIH).
-
An IFA was used to determine whether PoIFNα affects the production of specific antibodies against the influenza virus. Sera collected from the H1N1, LIH, and HIH groups at 21 dpi were analyzed and the fluorescence intensity levels in the LIH and HIH groups were higher than those in the group not treated with PoIFNα (Fig. 4A).
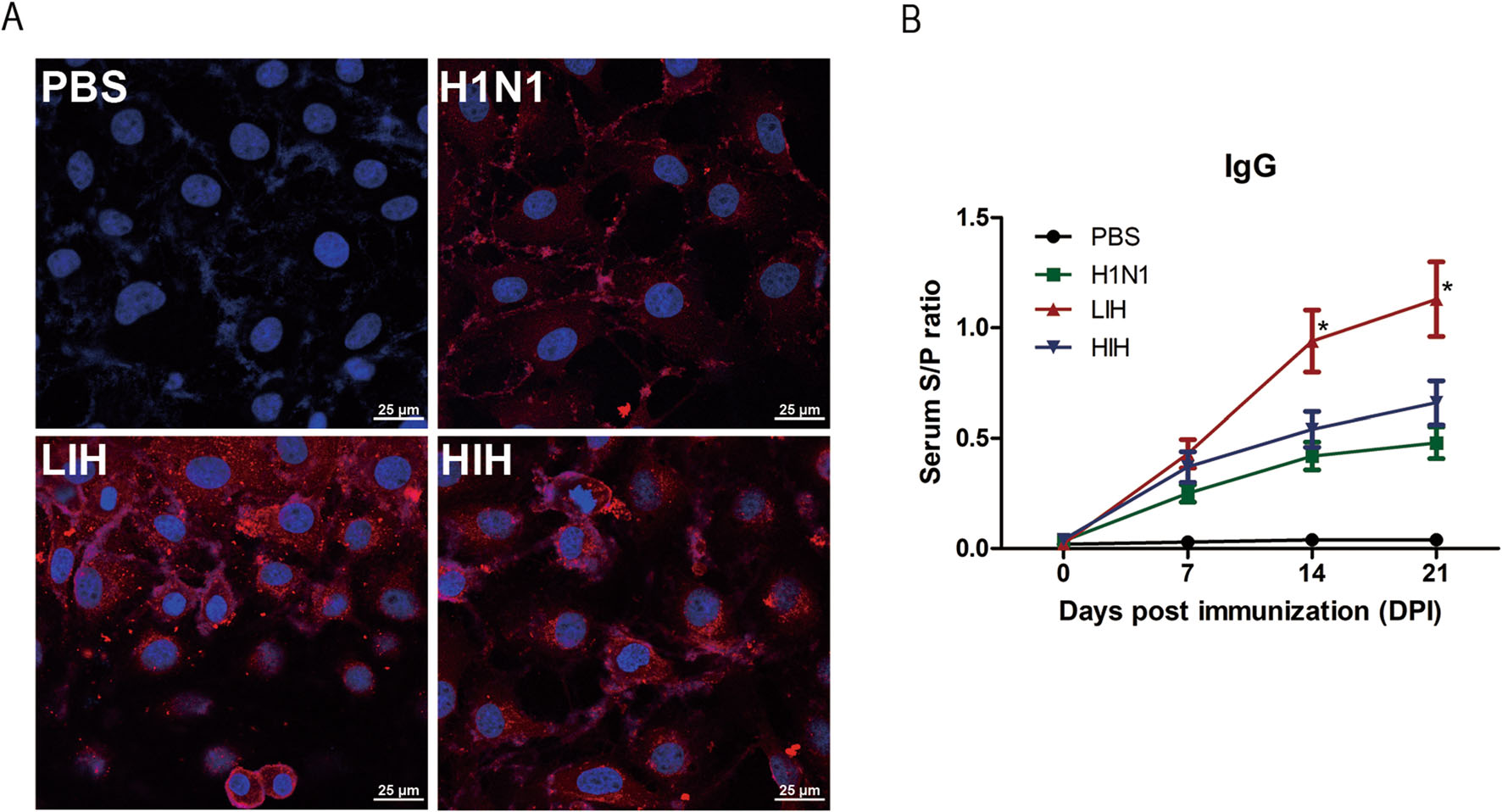
Figure 4. Humoral immune responses in vaccinated pigs induced by PoIFNα. A An immunofluorescence assay (IFA) comparing H1N1 influenza virus and sera from vaccinated pigs was developed to determine binding affinity. PK15 cells infected with CAS01 (MOI = 0.001) were fixed and then incubated with pig sera (1:100) from each group. The cells were incubated with TRITC-conjugated anti-swine IgG antibody (1:100; red), followed by nuclear staining with DAPI (merge; blue), and examined by confocal microscopy. Scale bars, 25 μm. B ELISA was performed to detect the HA-specific antibody responses in sera collected from immunized pigs at 0, 7, 14, and 21 days post-infection (dpi). Antibody responses are expressed as the mean sample-to-positive ratios and error bars represent standard deviations. Statistically significant differences are indicated (*P < 0.05). Group included PBS, recombinant porcine interferon α (PoIFNα), low-dose PoIFNα and H1N1 (LIH), and high-dose PoIFNα and H1N1 (HIH).
Next, ELISAs were performed to quantify serum HA-specific antibodies (Fig. 4B). Compared to that in the PBS group, the ratio of IgG increased in the experimental groups post-immunization. However, there were no significant differences in IgG responses among the experimental groups at 7 dpi, based on a one-way analysis of variance (ANOVA). Further, the IgG ratio in the LIH group was significantly higher than that in the other control groups at 14 and 21 dpi (P < 0.05).
-
After challenge with the influenza virus (strain H1N1-CAS01) at 21 dpi, rectal temperatures were measured, and clinical signs were recorded daily (Fig. 5). Pigs in the PBS and H1N1 groups developed high fevers (≥ 40.5 ℃) 2 days post-challenge (dpc), whereas animals in the PoIFNα group exhibited only a slight fever (39.5 ℃–40.4 ℃) 3 dpc, after which, body temperatures gradually recovered (Fig. 5A). Pigs in the PBS group began to lose their appetites from 1 dpc and clinical signs (e.g., depression, dyspnea, severe coughing, and mucus discharge from the eyes and nose) began to appear at 2 dpc. Similar low-level clinical signs were observed in the H1N1 and PoIFNα groups (Fig. 5B). Pigs in the LIH group developed slight fevers within 7 days (Fig. 5A), but no other signs were apparent. Further, the clinical scores of the LIH group were significantly lower than those in the other groups 4 and 5 dpc (P < 0.05; Fig. 5B).
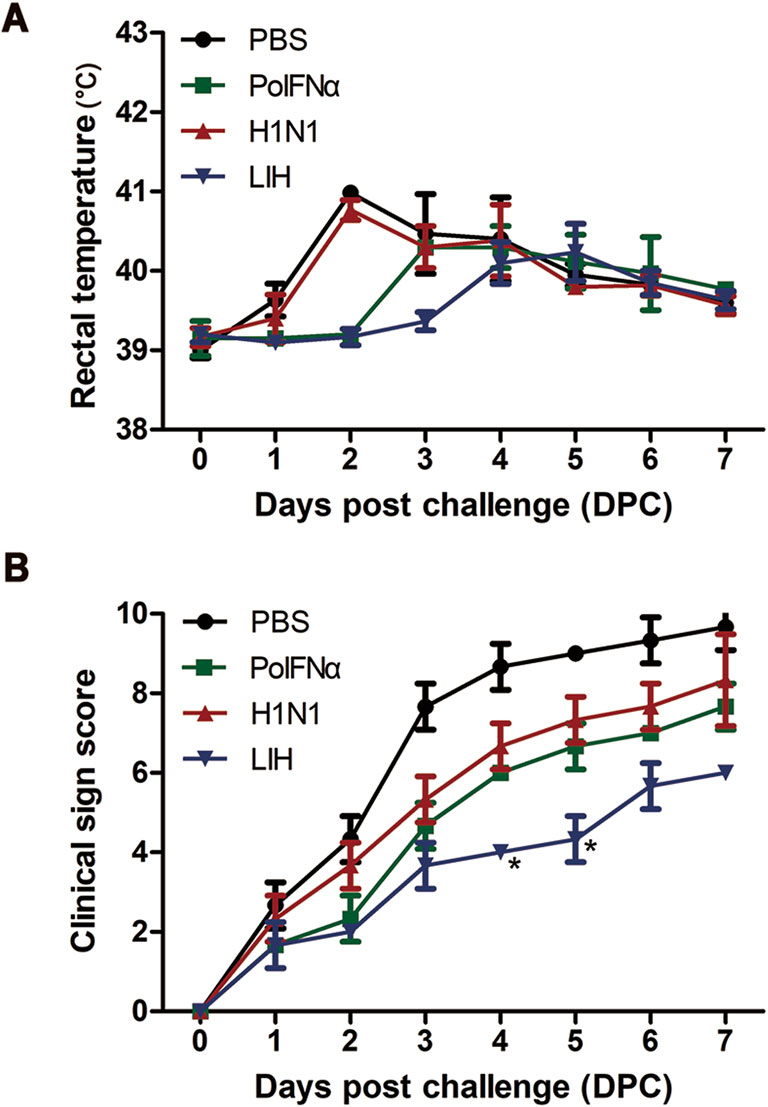
Figure 5. Clinical signs in pigs after challenge with H1N1-CAS01. A Rectal temperatures. B Clinical signs were scored for each group and recorded daily post-challenge, including respiratory rate (0–2 points), attitude (0–3 points), respiratory distress (0–3 points), and coughing (0–1 points). Data are the mean ± S.D. *P < 0.05 by t-test, comparing to 0 days post-infection (dpi) of each group. Groups included PBS, recombinant porcine interferon α (PoIFNα), H1N1 influenza virus-immunized (H1N1), and low-dose PoIFNα and H1N1 (LIH).
Expression and Purification of PoIFNα in E. coli
PoIFNα Induces High Antiviral Activity in Various Cells
PoIFNα Induces the Expression of Cytokines and CCRs After Intranasal Immunization of Pigs
Low-dose PoIFNα Induces Higher mRNA Levels of CCRs and Cytokines
Humoral Immune Response Regulated by Lowdose PoIFNα
Clinical Signs Post Influenza Virus Challenge
-
In this study, recombinant PoIFNα was combined with inactivated H1N1 influenza virus and administered in a porcine model. Clinical and immune responses were assessed and compared with those after the inoculation of inactivated H1N1 without PoIFNα. Pigs that were intranasally inoculated with the combined vaccines exhibited rapid, significant interferon responses and superior protection against H1N1 virus infection.
Type Ⅰ IFN was found to have a potent effect as an influenza vaccine adjuvant in a mouse model (Proietti et al. 2002) and PoIFNα has been used as an effective adjuvant in a recombinant protein vaccine against foot and mouth disease virus (Cheng et al. 2007). Furthermore, it is wellknown that genes encoding chemokines, receptors, and cytokines can be significantly up-regulated after type Ⅰ IFN administration via the mucous membranes. These genes regulate antigen processing, and lymphocyte activation and migration as well as other functions (Tovey 2002; Namangala et al. 2006; Tovey et al. 2008). However, the molecular mechanism associated with type Ⅰ IFN and the optimal dosage for immunomodulation has not been determined for mucosal administration.
Available data show that orally or intranasally administrated low-dose IFN-α exerts systemic immunomodulatory effects and reduces early virus replication in mice (Beilharz et al. 1997; Brod et al. 1999). Within 8 h, lowdose oral bovine IFN-α (200 U) was found to up-regulate genes involved in cytokine–cytokine receptor interaction, which encode proteins likely needed for antiviral immunity (Mamber et al. 2016). An additional study using type Ⅰ IFN as a mucosal adjuvant for an influenza vaccine showed that intranasal immunization with 5 μg of HA and high-dose IFN (4000 U) could induce high levels of HA-specific antibodies in sera of mice. (Santini et al. 2000). These studies provided the basis for our dose selection. Determining the optimal dose is the focus of our subsequent studies.
Previous studies have indicated that PoIFNα can induce rapid protection against foot and mouth disease virus in swine. Further, increased expression of the cytokines IL-15, IL-18, and IL-12, as well as the chemokine IP-10, is closely related to activation of the innate immune system (Diaz-San Segundo et al. 2010). In this study, levels of IL-2, IL-18, IFN-γ, IL-6, and IL-10 were increased in all three treatment groups at 12 hpi, whereas these cytokines were most up-regulated in the low-dose PoIFNα-treated group, indicating activation of the innate immune response after a single/combination vaccine immunization.
IL-2, IL-6, IL-18, and IFN-γ can increase lymphocyte activity and play an essential role in antiviral defense responses. IL-2 has an active role in inducing T cell proliferation, stimulating natural killer (NK) cell growth and cytotoxicity, and inducing the synthesis of B cell neutralizing antibodies (Hromadnikova et al. 2016). IL-6 stimulates NK cells and promotes T and B cell differentiation and antibody production (Hilbert et al. 1989; Rabinowich et al. 1993; Tormo et al. 2012), whereas IL-18 (which belongs to the IL-1 family) regulates a variety of cell development processes and cytokine secretion (Arend et al. 2008). IL-18 is also a Th1 cell growth and differentiation factor that can induce the production of IFN-γ by activated B, T and NK cells, which participate in anti-infection immunity. IFN-γ additionally activates macrophages and up-regulates the expression of MHC molecules (Giacomini et al. 1988; Foster et al. 2003). Moreover, IL-10 can promote B cell proliferation and differentiation and antibody production (Itoh and Hirohata 1995; O'Keefe et al. 1999). To understand the regulation of mucosal immunity by IFN, the immunoregulatory cytokines IL-2, IL-6, IL-18, IFN-γ, and IL10 were evaluated, as were CCR9 and CCR10, which are closely associated with lymphocyte homing. The up-regulation of these cytokines in PBMCs indicated changes in systemic cytokine transcriptional levels and activation of the innate immune system. Comparing immune response with or without PoIFNα treatment, the transcription of these cytokines in the LIH group was most remarkable at 12 hpi, and IL-6 and IL-10 levels were still significantly higher than those in the other groups at 24 hpi. Our results further suggest that the intranasal administration of low-dose PoIFNα can induce a strong systemic innate immune response. We therefore speculate that its administration could better activate subsequently recruited immune cells and adaptive immune responses.
CCR9 and CCR10 are expressed during mucosal immunity mediated by T lymphocyte subtypes and IgA+ plasmablasts and serve as homing signals for the digestive tract mucosa. Experimental data indicating the induction of local and distal mucosal immunity through oral immunization suggest that the mucosal immune system also exists in swine (Hyland et al. 2004). Additionally, da Cunha et al. demonstrated that intranasal immunization using the rhoptry protein of Toxoplasma gondii with Quli-A can stimulate a mucosal immune response (da Cunha et al. 2012). Based on this feature, lymphocytes sensitized in one mucosal tissue can be transmitted to other mucosa through the circulatory system. We thus examined the mRNA expression of CCR9 and CCR10 in PBMCs to explore the mucosal immune response after nasal mucosal administration. The combination of low-dose PoIFNα and H1N1 inactivated virus promoted the transcription of CCR9 and CCR10, especially at 12 hpi. It is likely that this has a positive effect on subsequent lymphocyte homing to mucosal sites and consequently the production of secretory IgA.
Serum antibody levels also comprise an effective indicator of general immunity. Our ELISA data indicated that the immunized swine produced IAV HA-specific antibodies at 7 dpi, which peaked at 21 dpi. PoIFNα can further enhance antibody production and the low dose was found to be more effective than the high dose. With respect to clinical indicators of infection, pigs in the LIH group displayed only mild signs within 7 days of H1N1 virus challenge, indicating an enhanced immune effect. Based on clinical manifestations, it was not difficult to determine that the immune effect in the LIH group was superior, and monitoring changes in the actual viral load after challenge will be the focus of subsequent studies. Taken together, our results indicate that intranasal immunization with low doses of PoIFNα and inactivated IAV can induce a more intense systemic immune response, a significant increase in serum antibody levels, and more effective protection against H1N1 virus challenge in swine.
IAV has caused epidemics that have harmed the swine industry and infection by IAV represents one of the major immunosuppressive diseases that are difficult to eradicate in intensive pig farms (Brown 2000). If the nasal delivery route can be incorporated into systems being developed to prevent H1N1 influenza virus infection, this would have the added benefits of convenience, efficiency, and low-cost, which will provide huge benefits for pig production. Overall, our study showed that the immunization of pigs via nasal administration using low-dose PoIFNα combined with inactivated IAV can activate a strong systemic innate immune response with obvious up-regulation of the transcription of lymphocyte homing-related signaling molecules and immunoregulatory cytokines. Meanwhile, low-dose PoIFNα induced higher levels IAV-specific neutralizing antibodies, which alleviated the early clinical signs of influenza virus infection. This study on the regulation of pig mucosal immunity by PoIFNα is expected to lead to the development of new applications for PoIFNα and suggest new strategies for the development of mucosal influenza vaccines.
-
We gratefully acknowledge the workers of Beijing Sinder Technology Co., Ltd. for their help with pig immunization and sample collection. This work was supported by Grants from the National Key R & D Programme of China (2017YFD051105), the National Natural Science Foundation of China (31630079), the National Science and Technology Major Project (2018ZX10101004), and the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB29010000). W.J.L. is the principal investigator of the Innovative Research Group of the National Natural Science Foundation of China (Grant No. 81621091). J.L. is supported by Youth Innovation Promotion Association of CAS (2019).
-
JL and WL supervised the Project and designed the study; JL and LL planned and conducted the experimental work, analyzed the data, and wrote the manuscript; WF, HZ, SZ, LC, MW, XB, WY, LY, and LS provided technical support. All authors read and approved the final manuscript.
-
All authors declare that they have no conflict of interest.
-
The pig experimental design and protocols used in this study were approved by the Institute of Microbiology, Chinese Academy of Sciences of Research Ethics Committee (Permit Number: PZIMCAS2017001). All pig experimental procedures were performed in accordance with the Regulations for the Administration of Affairs Concerning Experimental Animals approved by the State Council of People's Republic of China.
Conflicts of interest
Animal and Human Rights Statement
-

Table S1. Immunization program of inactivated H1N1 and PoIFNα in pigs

Table S2. Primer sequences used in this study

Table S3. Antiviral activities of PoIFNα toward different cell lines














 DownLoad:
DownLoad: