-
Avian influenza virus (AIV),a member of the Orthomyxoviridae family,is a kind of segmented,negative-stranded RNA virus. It is highly mutational,and can be divided into different serotypes. AIV is a serious threat to most kinds of birds,as well as to public health. In particular,in recent years,H5N1 AIV,has not only done great harm to poultry (2) ,but also caused many lethal infection cases in human (4, 9). The human infection cases caused by H5N1 AIV,as reported to the World Health Organization,is 230 in total,from January 1 in 2003 to July 14 in 2006,of which 132 were lethal corresponding to a mortality rate of 57.4%.
Current vaccines against H5N1 AIV in the world are mainly inactivated vaccine,recombinant live virus vaccine and a DNA vaccine. Among those three vaccines,the DNA vaccine,a new vaccine based on nucleic acid,which was developed in the 1990s,can induce high level CTL activity and antibody response. It also has many other advantages compared with traditional vaccines (10). For example,the DNA vaccine can induce lasting immune response,while the plasmid vector composing DNA vaccine has no immunogenicity; additionally,the DNA vaccine can be used repeatedly,easily produced,stored and transported because of its stability. Thus,in recent years,DNA vaccines against H5N1 AIV has held the attention of many researchers in view of its potential application in the future.
The genome of H5N1 AIV is composed of eight RNA segments with negative polarities,which encode 10 proteins,including PB1,PB2,PA,HA,NP,NA,M1,M2,NS1 and NS2. At present,domestic research on DNA vaccine against H5N1 AIV has mainly focused on he-magglutinin (HA) and neuraminidase (NA),and a few studies response,while theplasmid vector composing DNA vaccine has no immunogenicity; additionally,the DNA vaccine can be used repeatedly,easily produced,stored and transported because of its stability. Thus,in recent years,DNA vaccines against H5N1 AIV has held the attention of many researchers in view of its potential application in the future.
The genome of H5N1 AIV is composed of eight RNA segments with negative polarities,which encode 10 proteins,including PB1,PB2,PA,HA,NP,NA,M1,M2,NS1 and NS2. At present,domestic research on DNA vaccine against H5N1 AIV has mainly focused on hemagglutinin (HA) and neuraminidase (NA),and a few studies have provided convincing evidence that a DNA vaccine encoding HA or NA of H5N1 AIV could induce protective immunity effectively in experimental animals (4, 5, 11). However,a DNA vaccine encoding the M gene or the NP gene remains to be studied. In order to know whether an M-gene DNA vaccine or NP-gene DNA vaccine can provide sufficient protective effects against H5N1 AIV,we amplified the M and NP genes of H5N1 AIV (A/chicken/Hubei/489/2004) and constructed eukaryote expression plasmids pHM6-m and pHM6-np as DNA vaccines. Then we detected the protective effects of the two DNA vaccines in BALB/c mice.
HTML
-
H5N1AIV,A/chicken/Hubei/489/2004,used in this study,was provided by Professor Shu-wen WU and stored in our laboratory. pMD18-T vector was purchased from TaKaRa(Dalian),and Eukaryote expression vector pHM6 was purchased from Roche. Competent cells DH5α were developed by our laboratory. Reverse transcriptase M-MuLV and restriction endonucleases (Hind Ⅲ and EcoR Ⅰ) were purchased from MBI Fermentas. KOD Plus DNA polymerase and T4 DNA ligase were purchased from TOYOBO (Shanghai). Taq DNA polymerase was purchased from TaKaRa (Dalian).
SPF embryo eggs and BALB/c mice: 9-day-old SPF embryo eggs were purchased from Wuhan institute of biolo-gical products. BALB/c female mice (6-week-old) were purchased from the School of Medicine in Wuhan University.
-
H5N1 AIV,A/chicken/Hubei/489/2004,was inoculated into the allantoic cavities of 9-day-old SPF embryo eggs. Inoculated eggs were incubated for 72 hours at 34℃ before collection of the allantoid fluid. The allantoid fluid was harvested and stored at -70℃ for the use followed. Further to information provided in previous reports (3, 6, 7) ,M and NP gene sequences of H5N1 AIV in GenBank,universal primers for H5N1 AIV reverse transcription and two pairs of specific primers (contai-ning Hind Ⅲ and EcoR Ⅰ restriction sites) for respective amplification of viral M gene and NP gene were designed. All the primers were synthesized by Sangon (Shanghai). The expected size of the gene segments amplified was ~ 1.1 kb (M) and 1.6 kb (NP),respectively.
Universal primer for H5N1 AIV reverse transcrip-tion: 5'-AGCGAAAGCAGG-3'
M forward primer:5'-AAAGCTTAGCAAAAG-CAGGTAGATATTGAA-3'
M reverse primer:5'-AGAATTCAGTAGAAAC-AAGGTAGTTTTTTACTC-3'
NP forward primer:5'-AAAGCTTCAGCGAAA-GCAGGGTAGATAATCA-3'
NP reverse primer:5'-AGAATTCAGTAGAAAC-AAGGGTATTTTTCTTA-3'
-
Viral RNA was isolated from the allantoid fluid containing H5N1 AIV (A/chicken/Hubei/489/2004) according to the instruction for Trizol reagent.
-
With the two-step RT-PCR method,the M gene and NP gene were amplified,using their specific primers. The first cycle of the PCR program consisted of a 4-min period at 94℃ and was followed by 30 cycles with the following conditions: 94℃ for 30 sec,60℃ for 30 sec,and 70℃ for 2min. The program ended with one cycle at 70℃ for 10 min. KOD Plus DNA polymerase was used in the PCR reaction. The products amplified were confirmed by gel electrophoresis,and then extracted with the Gel Extraction Kit purchased from OMEGA.
-
An adenine deoxyribonucleic acid was added to the 3' end of the two kinds of extracted PCR products by Taq DNA polymerase,in virtue of its characteristic of adding an adenine deoxyribose nucleotide acid to the 3'end of the DNA segment. The reaction was performed at 72℃ for 30 minutes. Then the products were extracted with the Gel Extraction Kit.
-
Two kinds of PCR products,to whose 3' end an A had been added,were inserted into the pMD18-T vector; followed this,the two kinds of recombinant plasmids were introduced into competent cells (DH5α),respectively. Competent cells who took up the right recombinant plasmid were selected on Xgal/IPTG/Amp LB plates. Two kinds of plasmids were obtained from transformed competent cells,and identified by restriction enzyme digestion (Hind Ⅲ and EcoR Ⅰ) and gel electrophoresis. These plasmids were named pMD18-T-m and pMD18-T-np,respec-tively,and sequenced at UNITED GENE HOLDINGS LTD. (Shanghai).
-
After the digestion of the pMD18-T-m,pMD18-T-np and pHM6 with Hind Ⅲ and EcoR Ⅰ,the M gene,NP gene and linear pHM6 vector were obtained respectively by gel electrophoresis and extraction. The M gene was then linked to the linear pHM6 vector,forming orbicular eukaryote expression plasmid pHM6-m. A similar process using the NP gene,resulted in expression plas-mid pHM6-np. pHM6-m and pHM6-np were introduced into competent cells DH5α. The competent cells were then incubated in LB/ampicillin(50 μg/ mL) medium. Two kinds of recombinant plasmids were respectively extracted from the LB culture,identified by restriction enzyme digestion (Hind Ⅲ and EcoR Ⅰ) and gel electrophoresis. Both eukaryote expression plasmids pHM6-m and pHM6-np were sequenced at UNITED GENE HOLDINGS LTD. (Shanghai).
-
The E.coli cells containing plasmid pHM6-m or pHM6-np were cultured in 200 mL LB/ampicillin (50 μg/ mL) medium. Then pHM6-m and pHM6-np were extracted respectively with the Plasmid Rapid Isolation Kit (BioDev,Beijing). The two kinds of plasmids were quantified with a UV-VIS Spectrophotometer,dissol-ved in PBS buffer,and stored at -20℃ for later use.
-
32 BALB/c female mice (6-week-old) were divided into four groups at random,8 mice in each group. In the pHM6-m plasmid group,each mouse was injected with 30 μg pHM6-m (dissolved in 100 μL PBS),while in the pHM6-np group,each mouse was injected with 30 μg pHM6-np (dissolved in 100 μL PBS). In the mixed plasmids group each mouse was injected with 15 μg pHM6-m and 15 μg pHM6-np (dissolved in 100 μL PBS together). The control group of mice were injected with 100 μL PBS. All mice were injected via intramuscular route under ether anesthesia.
-
Two weeks after immunization,all the mice were challenged with H5N1 AIV A/chicken/Hu-bei/489/ 2004 (105.6 ELD50(50% egg lethal dose H5N1 AIV A/chicken/Hubei/489/2004),by intran-asal admin is-tration of 100 μL of the viral suspension (allantoid fluid collected in 2.2.1). The mice were observed every day,and the number of mice died was recorded.
1.1. Virus,vectors and enzynes
1.2. Proliferation and purification of the virus
1.3. RNA isolation
1.4. RT-PCR and extraction of products
1.5. Adding A to 3'end of PCR products
1.6. Cloning PCR products into pMD18-T vector
1.7. Construction of eukaryote expression plasmids
1.8. Preparation of abundant eukaryote expres-sion plasmids
1.9. Vaccine formulations and immunization protocol
1.10. Infection
-
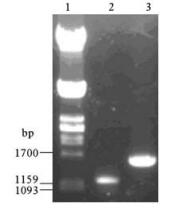
The M gene and NP gene of A/chicken/Hu-bei/489/2004(H5N1) were successfully amplified by RT-PCR with the designed primers,fragments for M (1.1 kb) and NP gene (1.6 kb) were obtained. The results from PCR reaction are shown in Fig. 1.
-
The two kinds of recombinant plasmids pMD18-T-m and pMD18-T-np were extracted and then confir-med by restriction enzyme digestion (Hind Ⅲ and EcoR Ⅰ) and gel electrophoresis. The results from gel electrophoresis are shown in Fig. 2. Products of expected size (1.1 kb and 1.6 kb) were obtained respectively after pMD18-T-m and pMD18-T-np were digested. Both plasmids were sequenced at United Gene Holdings Ltd.(Shanghai). The intact open reading frames of M1 and M2 could be found in pMD18-T-m and the intact open reading frame of NP was also found in pMD18-T-np. This confirms that the M gene and NP gene were cloned into pMD18-T vector successfully.
-
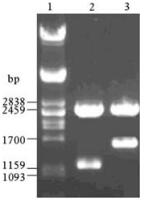
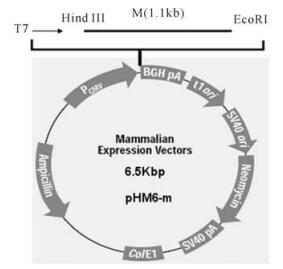
M fragments and NP fragments were respectively inserted into the multiple cloning site (between Hind Ⅲ and EcoR Ⅰ restriction sites) of the pHM6 vector (Fig. 3,Fig. 4). The eukaryote expression plasmids pHM6-m and pHM6-np were confirmed by restriction enzyme digestion (Hind Ⅲ and EcoR Ⅰ) and gel electrophoresis. Results from gel electr-ophoresis (Fig. 5) show that two kinds of fragments of the expected size (5.4 kb and 1.1 kb) were obtained after plasmid pHM6-m was digested. Similarly,digestion of pHM6-np created two fragments with expected size (5.4 kb and 1.6 kb). Digestion of pHM6-np created two fragments with expected size (5.4 kb and 1.6 kb).
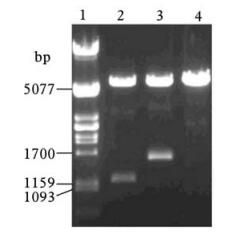
Figure 5. Gel electrophoresis of recombinant eukaryote expression plasmids pHM6-m and pHM6-np,digested with Hind Ⅲ and EcoR Ⅰ.1:λ/Pst I DNA marker; 2: pHM6-m (digested with Hind Ⅲ and EcoR Ⅰ); 3: pHM6-np (digested with Hind Ⅲ and EcoR Ⅰ); 4: pHM6 vector(digested with Hind Ⅲ and EcoR Ⅰ).
Plasmids pHM6-m and pHM6-np were then sequenced at United Gene Holdings Ltd.(Shanghai). Results confirmed that pHM6-m contains an intact M gene while pHM6-np contains an intact NP gene. We could therefore draw the conclusion that construction of the eukaryote expression plasmids pHM6-m and pHM6-np was successful. Plasmids pHM6-m and pHM6-np were then sequenced at United Gene Holdings Ltd.(Shanghai). Results confirmed that pHM6-m contains an intact M gene while pHM6-np contains an intact NP gene. We could therefore draw the conclusion that construction of the eukaryote expression plasmids pHM6-m and pHM6-np was successful.
-
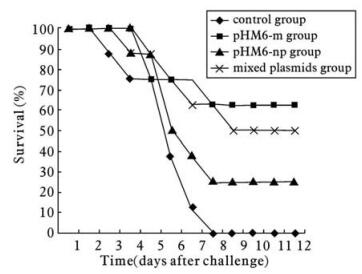
Two weeks after immunization,all mice were cha-llenged with 105.6 ELD50 (50% egg lathal dose) H5N1 AIV,A/chicken/Hubei/ 489/2004,and observed for the following 12 days. Table 1 and Fig. 6 showed that all mice in the control group died within 8 days. The final survival rates of mice in pHM6-m group and pHM6-np group were 62.5% and 25.0%,respectively. When mi-xed plasmids of pHM6-m and pHM6-np were immunized,50% protection was observed (Fig. 6).

Table 1. The survival rates of mice in different groups after H5N1 AIV challengea
2.1. Amplification of M gene and NP gene by RT-PCR
2.2. Cloning M gene and NP gene into pMD18-T vector and identification of products
2.3. Construction and identification of eukaryote expression plasmids pHM6-m and pHM6-np
2.4. Survival rates of mice after H5N1 AIV challenge
-
In this study,we amplified the full-length M gene and full-length NP gene of H5N1 AIV,A/ chicken / Hubei/ 489/2004,by RT-PCR,and inserted the M gene and the NP gene into pMD18-T vector respectively,and used these vectors for the study of the efficacy of a DNA vaccine con-taining the M gene or the NP gene against H5N1 AIV.
Previous research on DNA vaccines against H5N1 AIV have mainly focused on DNA expression plasmids containing the HA or the NA gene[4,5,11). We constructed pHM6-m and pHM6-np as DNA vaccines encoding the M gene and the NP gene respectively for the first time,and examined the protective effects of a M DNA vaccine and a NP DNA vaccine against homologous H5N1 AIV,A/chicken/Hubei/489/2004,were studied in BALB/c mice. Results show that a single intramuscular immunization with 30 μg pHM6-m could protect 62.5% of the mice challenged with homologous H5N1 AIV,whereas a single intramuscular immunization pHM6-np 30 mg gave 25.0% protection,sugges-ting that immunization with the M-gene DNA vaccine or the NP-gene DNA vaccine could protect against a homologous H5N1 AIV challenge. However,the NP-gene DNA vaccine provided weaker protection than the M-gene DNA vaccine. We also set a mixed plasmids group in which mice were immunized once with a mixture of 15 μg pHM6-m and 15 μg pHM6-np so as to investigate whether the M-gene DNA vaccine or NP-gene DNA vaccine is better. We expected a result that mixed DNA vaccines could protect against homologous H5N1 AIV more effectively than any single kind of DNA vaccine,but ultimately,the survival rate of mice in the mixed plasmids group only reached 50.0%,which was higher than the 25.0% in the pHM6-np group,but a little lower than the 62.5% observed in the pHM6-m group. That is,evidence of optimized protective effect was not observed when mixed DNA vaccines (M DNA vaccine and NP DNA vaccine) were used. However,the results provided convincing evidence that the protective effect of the M-gene DNA vaccine was better than that of the NP-gene DNA vaccine. It was also shown that the M gene and the NP gene could both be candidates for DNA vaccines against H5N1 AIV.
The M gene of H5N1 AIV encodes viral matrix proteins (M1 and M2),which is in the majority of all viral proteins. The DNA sequence of the M gene,in particular M1,is highly conserved (8) and in of all the genes of H5N1 AIV,the NP gene is the most conserved. So the M-gene DNA vaccine (pHM6-m) and the NP-gene DNA vaccine (pHM6-NP) are subjects for further studies,and their wide application can be expected in the future.
H5N1 AIV can cause lethal infection in human,and the mortality is high among infected subjects (4, 9) and so it presents a serious threat to public health. Our study results,based on BALB/c mice,showed that the M-gene DNA vaccine and the NP-gene DNA vaccine could provide reasonably effective protection against homologous H5N1 AIV in mammalians. This suggests that the M-gene DNA vaccine and the NP-gene DNA vaccine have much potential in clinical applications.
In this study,mice were immunized with pHM6-m or pHM6-np once via the intramuscular route. Although their protective efficacy could not meet the requirement for clinical application,results showed that pHM6-m and pHM6-np could provide obvious protection against H5N1 AIV. Previous research- (1, 4, 8, 9, 11) has show that protective efficacy of DNA vaccines can be improved by constructing DNA vaccines containing target gene with optimized codons,performing immunization se-veral times,improving the immunization route (for example,immunization by in vivo electroporation or gene gun),or immunized together with traditional vaccines. We can also try to improve the protection efficacy of M-gene DNA vaccine or NP-gene DNA vaccine by adding or deleting part of nucleotides of M gene or NP gene. Thus,based on this study,we think it is worthwhile to continue further research on a DNA vaccine encoding the M or NP genes of H5N1 AIV.













 DownLoad:
DownLoad: