HTML
-
At the beginning of 2019, the World Health Organization published a list of the ten most serious threats to public health (WHO 2019). Antibiotic resistance appeared on this list. Acinetobacter baumannii was classified within the most dangerous critical priority group of multidrugresistant (MDR) bacteria, which may be resistant to third generation cephalosporins and carbapenems (WHO 2018).
The resistance of A. baumannii strains was based on three mechanisms: inactivation of antimicrobial enzymes, mutagenesis that changed antimicrobial targets or cellular functions in bacterial cells (efflux pumps and other protein expression), and protection of bacterial targets against antimicrobial factors (Rice 2006). As an example of the first mechanism, there are genes encoding enzymes called β-lactamases, which hydrolyze the β-lactam bound in carbapenems, cephalosporins, and penicillins (Bonomo and Szabo 2006; Thomson and Bonomo 2005). The second mechanism is related to mutations in the bacterial chromosome affecting bacterial function or bacterial cell targets for antimicrobials. Spontaneous mutagenesis can lead to higher expression of proteins building the efflux pumps and also to affinity reduction for antimicrobial factors (Li et al. 2005). The described mechanisms protected Acinetobacter cells against quinolone (Bonomo and Szabo 2006) and colistin (Li et al. 2005). The third mechanism consists of modification of outer membrane proteins (e.g., porin channel proteins) necessary for transport of antimicrobial agents into bacterial cells (Bou et al. 2000; Thomson and Bonomo 2005). The absence of the CarO protein (29 kDa) in the outer membrane of A. baumannii led to meropenem and imipenem resistance (Mussi et al. 2005). As a result of antibiotics overuse, bacteria are acquiring new and more sophisticated resistance mechanisms, which make them almost impossible to destroy using most available antibiotics.
Infections with MDR bacteria among severely ill patients in intensive care units cause high death risk ranging from 26% to 68% (Kwon et al. 2007; Seifert et al. 1995; Sunenshine et al. 2007). Additionally, mortality among patients infected with MDR A. baumannii was higher than for infections with MDR Pseudomonas aeruginosa (Gkrania-Klotsas and Hershow 2006). Interestingly, MDR strains of A. baumannii cause infections of the respiratory tract, bones, bacteremia, or wound infections, which were noted among seriously injured soldiers in Afghanistan and Iraq (CDC 2004; Davis et al. 2005; Hawley et al. 2007; Hujer et al. 2006).
A. baumannii has been classified as an opportunistic bacterium that did not usually infect healthy people (Towner 2009). However, higher mortality has been reported for A. baumannii infections than for infections with other gram-negative bacteria (e.g., Klebsiella pneumoniae) (Jerassy et al. 2006; Robenshtok et al. 2006). Multidrug resistance and resistance to unfavorable environmental conditions may lead to frequent nosocomial infections, such as urinary tract and wound infections, pneumonia, bacteremia, or meningitis (Fournier and Richet 2006; Jawad et al. 1996). Moreover, high virulence in A. baumannii was also associated with biofilm formation, which contributes to difficulties during treatment (Tomaras et al. 2003). The pathogen spreads in a medical environment. Infectious strains have been found on laryngoscopes, keyboards, curtains, mops, equipment for patient lifting, and door handles (Wilks et al. 2006). Current research showed that MDR A. baumannii is also an airborne bacterium present in intensive care units (Jiang et al. 2018). Equipment and procedures necessary in patient care (i.e., respiratory equipment, humidifiers, wound care) are serious infectious factors which facilitate the spread of this extremely dangerous pathogen (Aygun et al. 2002; Bernards et al. 2004; Das et al. 2002; De Jong et al. 2004; Denton et al. 2005; Maragakis et al. 2004; Podnos et al. 2001; Villegas and Hartstein 2003; Wilks et al. 2006; Zanetti et al. 2007).
-
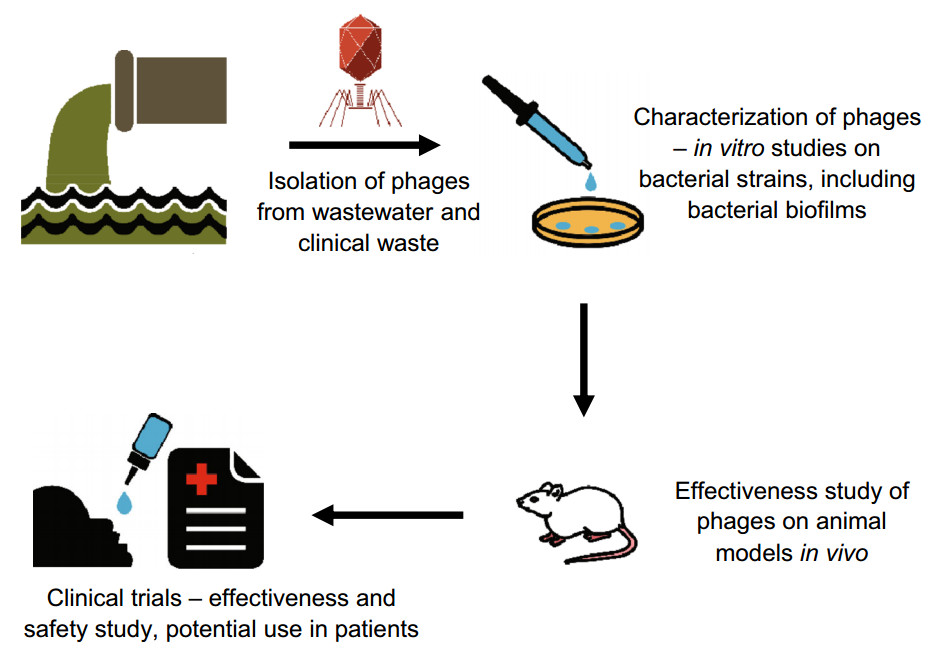
Based on the literature (Ghajavand et al. 2017) the best source of A. baumannii phages is hospital sewage. However, procurement and amplification of these phages entail many difficulties. Therefore, it is extremely important to adjust and optimize these methods for each individual phage. The methods for phage isolation and use are presented in Fig. 1.
Multidrug resistance among A. baumannii strains poses serious difficulties for treating infections (Perez et al. 2007). However, phage therapy could be a new weapon in the fight against A. baumannii infection (Sulakvelidze et al. 2001). Bacteriophages have no harmful effects on patients' microbiome and are known to be specific and selective to pathogens. Phage therapy against A. baumannii infection has therefore high potential to be an effective, natural, and safe treatment for patients with serious chronic infections (Clark and March 2006; Międzybrodzki et al. 2012; Van Helvoort 1992). Basic information about bacteriophages against A. baumannii is presented in Table 1.
Phage symbol Family Morphology Source of isolation Number of tested strains Type of life cycle Animal model application References IsfAB78 Myoviridae Six-sided symmetry, 100 nm long Water sample 43 MDR strains (12 of those strains were sensitive to phage) Lytic N.A. Ghajavand et al. (2017) IsfAB39 Podoviridae Six-sided symmetry, short tail and 50 nm head Water sample 43 MDR strains (11 of those strains were sensitive to phage) Lytic N.A. Petty phage Podoviridae Short 15 nm long tail and 60 nm head Sewage 40 strains (25 of those strains were MDR; 4 strains were sensitive to phage) N.A. N.A. Hernandez-Morales et al. (2018) Acibel004 Myoviridae 105 nm long contractile tail and 70 nm icosahedral head and formed 1–2 mm plaques Wastewater sample 34 MDR strains (28 of those strains were sensitive to phage) Lytic N.A. Merabishvili et al. (2014) Acibel007 Podoviridae 10 nm long noncontractile tail, 60 nm head and forms 3–5 mm plaques Wastewater sample 34 MDR strains (28 of those strains were sensitive to phage) Lytic N.A. vB_AbaS_Loki Siphoviridae Isometric 57 nm capsid and non-contractile 176 nm long tail (and 10 nm diameter); short spikes at the tail terminus Sludge 34 strains (2 of those strains were sensitive to phage) Lytic N.A. Turner et al. (2017) SH-Ab 15599 Myoviridae Head with tail (both 88 nm long), formed round, clear plaques (2–3 mm diameter) with haloes Sewage 48 carbapenem resistant strains (13 of those strains were sensitive to phage) Lytic N.A. Hua et al. (2018) SH-Ab 15708 Myoviridae Head (88 nm long diameter) and tail (63 nm long) formed round, clear plaques (2–3 mm diameter) with haloes Sewage 48 carbapenem resistant strains (14 of those strains were sensitive to phage) Lytic N.A. SH-Ab 15497 Siphoviridae Tail (125 nm long and 4 nm wide) and head (55 nm diameter) formed small plaques (0.5 mm diameter) Sewage 48 carbapenem resistant strains (14 of those strains were sensitive to phage) Lytic N.A. SH-Ab 15519 Podoviridae Short, non-contractile, straight tail (18 nm long) and polyhedral, symmetrical head (55 nm diameter) formed round, clear plaques (8–9 mm diameter) with haloes Sewage 48 carbapenem resistant strains (8 of those strains were sensitive to phage) Lytic Mouse model- lung infection; intranasally administration of phage; the survival rate 90% vB-GEC_Ab M-G7was (phi G7) Myoviridae 120 nm contractile tail and 100 nm diameter icosahedral head Sewage water 200 strains (136 of those strains were sensitive to phage) Lytic Rats’ wund model; phage added on the wound; the survival rate 100% Kusradze et al. (2016) PBAB08 Myoviridae Head 180 nm in diameter and 360 nm long tail Bacteriophage Bank of Korea 14 MDR strains (5 of those strains were sensitive to phage) N.A. Mice model- lung infection; intranasal phage cocktail (PBAB08, PBAB25, PBAB68, PBAB80, PBAB93) injection; the survival rate 35% Cha et al. (2018) PBAB25 Myoviridae Head of 80 nm in diameter and 90 nm long tail Bacteriophage Bank of Korea 14 MDR strains (1 of those strains were sensitive to phage) N.A. Mice model- lung infection; intranasal phage cocktail (PBAB08, PBAB25, PBAB68, PBAB80, PBAB93) injection; the survival rate 35% PD-6A3 Podoviridae Short tail (9 nm in length), isometric head (50 nm in diameter); the phage created significant 2–3 mm diameter halos Sewage 552 MDR strains (179 of those strains were sensitive to phage) Lytic Sepsis mouse model; intraperitoneal administration; the survival rate of endolysin therapy group, endolysin + phage therapy group, phage therapy group and phage cocktail (14 phages) therapy group were 70, 70, 60, and 50%, respectively Wu et al. (2019) AB3P1 N.A. 0.8–1.5 mm oval plaques Different regions in Baghdad city including sewage, farm soil, feces of sheep, chicken litter, and swab from surgical lounge of several hospitals in Baghdad 23: 11 extensive- and 12 pan-drug resistant strains (18 of those strains were sensitive to phage) Lytic Mice model; intreperitoneally administration of AB3 phages; the survival rate 100% Jasim et al. (2018) AB3P2 N.A. 0.5 mm oval plaques Different regions in Baghdad city including sewage, farm soil, feces of sheep, chicken litter, and swab from surgical lounge of several hospitals in Baghdad 23: 11 extensive- and 12 pan-drug resistant strains (18 of those strains were sensitive to phage) Lytic Mice model; intreperitoneally administration of AB3 phages; the survival rate 100% AB3P3 N.A. 2.5–4.0 mm round plaques Different regions in Baghdad city including sewage, farm soil, feces of sheep, chicken litter, and swab from surgical lounge of several hospitals in Baghdad 23: 11 extensive- and 12 pan drug resistant strains (18 of those strains were sensitive to phage) Lytic Mice model; intreperitoneally administration of AB3 phages; the survival rate 100% AB3P4 N.A. 35 mm oval plaque Different regions in Baghdad city including sewage, farm soil, feces of sheep, chicken litter, and swab from surgical lounge of several hospitals in Baghdad 23: 11 extensive- and 12 pan drug resistant strains (18 of those strains were sensitive to phage) Lytic Mice model; intreperitoneally administration of AB3 phages; the survival rate 100% Table 1. Bacteriophages against A. baumannii.
Ghajavand et al. (2017) examined bacteriophages against A. baumannii isolated from intensive care units of Ishan Medical University hospitals. The study collected 350 samples including urine, catheter, wound, blood, eye swabs, sputum, and cerebrospinal fluid for A. baumannii isolation. Examination of multidrug resistance of 43 isolates showed that 100% were resistant to ciprofloxacin; 93% were resistant to meropenem, imipenem, ampicillinsulbactam, and cefepime; 91% were resistant to trimethoprim-sulfamethoxazole; 86% were resistant to ceftazidime; 84% were resistant to tetracycline; and 54% were resistant to amikacin. Bacteriophages in these studies were obtained from water samples (environmental samples and hospital waste). After incubation of waste samples from hospitals, clear plaques formed on lawns of AB78 and AB39 bacterial strains. Twelve A. baumannii isolates were sensitive to IsfAB78 phage and 11 were sensitive to IsfAB39 phage. Morphology of both lytic phages was examined with transmission electron microscopy, which indicated that both phages had six-sided symmetry. They significantly reduced the turbidity of the bacterial culture from OD600—2.8 in the control to OD600—0.4 in A. baumannii cultures treated with IsfAB78 or IsfAB39 phage, which suggested that the examined phages have potential for application in phage therapy. Moreover, studies by Hernandez-Morales et al. (2018) resulted in isolation and characterization of Petty phage obtained from sewage. The latent period for this phage was 25 min and burst size was estimated as 240 particles. Among 40 Acinetobacter strains tested, 4 were multidrug resistant (resistance to cefazolin, cefotaxime, chloramphenicol, and tetracycline) and susceptible to Petty phage.
Merabishvili et al. (2014) described two phages (Acibel004 and Acibel007) selected from wastewater samples. Two A. baumannii strains isolated from the nose of a Queen Astrid Military Hospital patient were used for phage amplification. A host range study showed that non-A. baumannii strains (A. nosocomialis and A. pittii) were resistant to both phages. Out of 28 chosen A. baumannii strains, 2 were resistant and 15 were sensitive to the phages. Adsorption and/or propagation of phages was possible on 11 strains. Acibel007 propagated on 61% and adsorbed on 71% of 28 A. baumannii strains, while Acibel004 propagated on 75% and adsorbed on 89% of strains. Based on efficiency of the plating method, phage Acibel007 had higher lytic activity than Acibel004. Moreover, A. baumannii 070517/0072 showed low frequency of mutation during phage application, which suggests low capacity for selection of phage-resistant mutants among bacteria. Maximum adsorption for Acibel007 and Acibel004 was 95% in 10 min and 85% in 15 min, respectively. The latent period and burst size for Acibel007 were 21 min and 145 virions per infected bacterium, respectively, and for Acibel004 they were 27 min and 125 virions per host cell, respectively. Bacteria were incubated either with a mixture of two phages or with a single phage. After 24 h of incubation, OD600 was 0.13, 0.18, and 0.07 for Acibel004, Acibel007, and simultaneous incubation, respectively, compared to 0.42 in an untreated control. OD600 measurements suggested that simultaneous phage application resulted in greater bacterial titer reduction than single phage application.
Turner et al. (2017) characterized another phage against A. baumannii, the bacteriophage Loki (vB_AbaS_Loki), isolated from sludge. The latent period for Loki phage was 40 min and burst size was 43 PFU per infective center. Mutation in A. baumannii ATCC 17978 gene lpxA, encoding a protein required for LPS synthesis, prevented phage adsorption. The authors suggested that an LPS component could be used by Loki as a receptor on the bacterial host. Loki formed turbid plaques (0.5 mm in diameter) on A. baumannii ATCC 17978 and Ca2+ was necessary for plaque formation. Thirty-six out of 38 examined A. baumannii strains were sensitive to Loki phage. Genome analysis showed that Loki has genes gp50 (a putative class I holin) with two transmembrane domains and gp51 containing an N-acetylmuraminidase and a domain that binds peptidoglycan at the C-terminus (Briers et al. 2007).
Hua et al. (2018) looked for phages against MDR A. baumannii clinical isolates. They found that 48 isolated strains were resistant to ciprofloxacin, cefepime, ceftazidime, and piperacillin-tazobactam. The study obtained 30 bacteriophages but only four of them were described: phage SH-Ab 15599, SH-Ab 15708, SH-Ab 15497, and SH-Ab 15519. The phage cocktail containing all these phages was effective against 88% of A. baumannii isolates but no synergic effect was described (i.e., strains resistant to single phage application were also resistant to the phage cocktail). Phages SH-Ab 15599, SH-Ab 15708, SH-Ab 15497, and SH-Ab 15519 were effective against 27%, 29%, 29%, and 17% of A. baumannii isolates, respectively. For phage SH-Ab 15519, an enzyme was suggested as cause of bacterial exopolysaccharide degradation. This phage demonstrated high absorption (90%) within 10 min. Subsequently, the phage was shown to be stable at pH 5 to 12 and temperatures from 4 to 50 ℃.
Kusradze et al. (2016) described another phage against A. baumannii, phage vB-GEC_Ab-M-G7 (phi G7), which was classified in the Myoviridae family (Matsuzaki et al. 2005). The phage proved to be active against 65% of the 200 A. baumannii strains tested (Kusradze et al. 2016). Phage activity was determined at different temperatures and it was observed that incubation at 37 ℃ had no effect on phage activity. After 24 h incubation at 50 ℃, 90% of phage particles were still active but total inactivation was obtained after 24 h incubation at 70 ℃. Moreover, phage phi G7 remained stable when incubated for 24 h in the presence of chloroform and for 5 h at pH from 5 to 11. Phage titer reduction to 103 was caused by 24 h incubation at pH 3.
Cha et al. (2018) examined the sensitivity of 14 MDR A. baumannii strains to nine bacteriophages: PBAB05, PBAB06, PBAB07, PBAB08, PBAB25, PBAB68, PBAB80, PBAB87, and PBAB93. Seven of the tested bacterial strains were sensitive to four or five phages. A. baumannii 28 was sensitive to five out of nine phages. These bacteriophages were used to prepare a phage cocktail, which was used in in vivo studies. Burst size for PBAB25 and PBAB08 was 630 and 215, respectively, and both phages were able to cause bacterial cell lysis 25 min after infection. PBAB08 and PBAB25 showed stability at pH 5–10 and temperatures from 4 to 55 ℃ for 1 h. Moreover, the authors sequenced whole genomes of phages PBAB08 and PBAB25 and they found that the genome of these phages was linear, with dsDNA of 42312 bp (PBAB08) and 40260 bp (PBAB25). Sequenced genomes were compared with reported genomes of phages against Acinetobacter. The results showed that phage PBAB08 had 57% sequence coverage and 99% similarity to AB1 phage (Yang et al. 2010), whereas phage PBAB25 had 78% sequence coverage and 97% similarity to IME_AB3 phage (Zhang et al. 2015).
Wu et al. (2019) presented the characterization of 14 lytic bacteriophages, which were tested on 552 A. baumannii strains. The phage cocktail combining the mentioned phages lysed 54% of 552 bacterial isolates. One of the lytic phages (phage PD-6A3) had a wider lytic spectrum (32% of 552 bacterial isolates) than the other 13 bacteriophages. PD-6A3 bacteriophage was stable at temperatures between 4 and 50 ℃ and at pH from 5 to 10. The burst size for this phage was 129 PFU per infected bacterial cell.
Liu et al. (2019) described another phage against A. baumannii. Phage IME200 formed 2 mm diameter clear plaques with halos. The halo was associated with depolymerase activity. Phage IME200 was able to infect 10 out of 41 A. baumannii isolates and it encoded an enzyme that might be useful in the treatment of biofilm formed by A. baumannii strains. This biofilm is resistant to disinfectants, so the use of phages with depolymerizing genes in their genomes creates a chance for treatment of infections caused by biofilm-forming resistant A. baumannii strains.
In another study, Jasim et al. (2018) isolated 23 A. baumannii strains from patients with septicemia, urinary tract infection, meningitis, wound infection, or pneumonia. Twelve out of 23 isolates were pan-drug resistant and 11 of them were extensive-drug resistant. Pan-drug-resistant clinical isolates are defined as strains causing difficulties in treatment and resistant to all antimicrobial drugs (Göttig et al. 2014). Analyses of bacterial sensitivity to bacteriophages indicated that 111 out of 136 A. baumannii-specific bacteriophages had high lytic properties and formed clear plaques with diameter greater than 3 mm (Jasim et al. 2018). For 10 randomly selected bacteriophages, the burst time ranged from 30 to 45 min and the minimum and maximum burst size was 130 and 245, respectively. Moreover, a phage cocktail (64 bacteriophages specific against A. baumannii isolates) was examined on 23 isolates (AB1-AB23). The results showed that 21 bacterial isolates (except AB2 and AB8) were infected by more than one bacteriophage, and AB3 was infected by six bacteriophages.
Wang et al. (2018) studied the use of phage ϕm18p against A. baumannii clinical isolate KM18. Murine RAW 264.7 macrophages were infected with KM18 and treated with the phage φm18p. After phage application [multiplicity of infection (MOI) 4, 0.4, and 0.04], the cell number was around two times higher than among cells infected with bacteria without treatment. A. baumannii titer in infected cells without treatment was around × log CFU/mL, but in cells treated with phage at MOI of 4, 0.4, and 0.04 it was around 4, 6, and 7 log CFU/mL, respectively.
-
Among the four bacteriophages characterized by Hua et al. (2018), SH-Ab 15519 was chosen for treatment in a mouse model of lung infection. A. baumannii was administered by the intratracheal route to neutropenic mice. The level of survival among mice infected with 106 CFU of A. baumannii was 91%. The survival level among mice infected with 107, 108, and 109 CFU of bacteria was 36%, 18%, and 18% respectively. Subsequently, mice were also infected with 108 CFU of A. baumannii and phage SH-Ab 15519 was administered intranasally at 0.1, 1, and 10 MOI. Different phage doses significantly increased the survival level among mice to the same value (90%) compared to the nontreated control (10%).
The potential of phi G7 phage application was examined in a rat wound model (Kusradze et al. 2016). In the control group, with an artificial wound and without inoculation of A. baumannii, no mortality was detected. No toxic effect after phage application was noted and all wounded rats treated with phages survived. Samples taken from rats using a sterile swab during the experiment showed no contamination with A. baumannii. Furthermore, samples from wounds taken using sterile swabs, which were added to 4.5 mL of BHI broth, showed a phage titer not more than ~ 2 × 101 PFU/mL of BHI broth. Two groups of rats were infected with bacteria in a titer of 5 × 108 CFU/mL, the first with A. baumannii T-10 and the second with A. baumannii G7. After 2 days, inflammation and pus formation were noted. Infection with A. baumannii G7 was more complicated than A. baumannii T-10 infection. In the untreated group infected with bA. baumannii G7, 70% of rats survived 5 days after inoculation. For animals infected with A. baumannii G7 and treated with 5 × 107 PFU/mL phage phi G7 12 h after infection, no mortality was reported and there were no purulent lesion after 6 days of the experiment. Furthermore, bacterial titer was reduced to 3 × 102 CFU/mL. In the group infected with the A. baumannii T-10, phage therapy led to full recovery of animals before 5 and 6 days of experiment (bacterial titer decreased to 9 × 102 CFU/mL).
A phage cocktail (consisting of five phages against A. baumannii 28) was used to treat mice infected intranasally with 1 × 108 CFU of A. baumannii 28 (Cha et al. 2018). Phages were also administered intranasally (1 × 109 PFU) every day of the experiment (7 days). At days 0 and 1, the phage cocktail was administered 4 h after bacterial infection. In the control group (i.e., mice infected with bacteria without phage therapy), the survival rate was 15% compared to mice treated with the phage cocktail where survival rate was 35%, 7 days after A. baumannii infection. Moreover, the bacterial titer (i.e., bacteria resistant to ampicillin and kanamycin) in mice lungs was reduced more than 100-fold, at 3 and 4 days after infection. Intranasal and oral phage administration had no effect on IgE level in mice serum; only intraperitoneal injection caused a 20% increase of IgE, which may suggest the potential route of phage administration. Furthermore, Soothill (1992) described in vivo studies concerning BS46 phage action against A. baumannii AC54. In the first study, mice were injected into the peritoneal cavity with 0.25 mL of 8 LD50 (LD50 for A. baumannii AC54 = 1.9 × 107 CFU). Five mice couples were treated with either 8.3 × 106, 1.7 × 106, 3.3 × 105, 6.6 × 104, or 1.3 × 104 PFU of phage BS46, and one last couple had no treatment with phage BS46. All mice in the treatment group survived, whereas all mice died in the non-treatment group. In the second study mice were injected with 0.25 mL of 3 LD50—five groups with one mouse per group. Each mouse was treated with either 1200, 120, 12, or 1 PFU of phage BS46, and the last mouse had no treatment with phage BS46. The first three mice survived but the last two died. Moreover, the author described a third study where mice were injected with 0.25 mL of 3 LD50—five groups with five mice per group. Each group was treated with 108, 36, 12, or 4 PFU of phage BS46, and the last group had no treatment with phage BS46. Mice survived only in the first group (four out of five treated mice), while all mice died in the other groups. Additionally, there was no toxicity of the phage BS46 for mice in the control group (i.e., treated only with phage). No differences in health status between mice with phage therapy and without phage administration were observed.
The phage PD-6A3, a phage cocktail, and Ply6A3 endolysin were tested in intraperitoneally infected mice (sepsis mouse model) with 1 × 109 CFU/mL of A. baumannii 32 (Wu et al. 2019). The survival rate among mice treated with Ply6A3 endolysin (intraperitoneal administration of 1 mL Ply6A3 at 2 mg/mL), Ply6A3 endolysin with PD-6A3 phage (intraperitoneal administration of 1 mL Ply6A3 at 2 mg/mL and 1 mL PD-6A3 at 109 PFU/mL), PD6A3 phage (intraperitoneal administration of 1 mL PD-6A3 at 109 PFU/mL), and phage cocktail (intraperitoneal administration with 1 mL of phage cocktail at 109 PFU/mL) was 70%, 70%, 60%, and 50%, respectively, on the last (7th) day of the experiment. Mice in the control group (infected with A. baumannii 32) died on the third day of the experiment.
Jasim et al. (2018) described in vivo studies where mice were injected intraperitoneally with AB3 isolate (0.3 mL at 106 CFU/mL) and treated, also intraperitoneally, with phage specific to the AB3 strain (0.3 mL at 108 PFU/mL administered 2 h after phage injection). All mice in this group survived more than 6 weeks. Contrastingly, all mice died 4 days after infection in the group without phage therapy. Moreover, in the group treated with phage but without bacterial infection mice also survived. In vivo studies of BALB/c mice showed that KM18 intraperitoneal injection at 2–2.5 × 108 CFU resulted in mice death after 24 h. Infection with KM18 at 2–3 × 107 CFU resulted in a survival rate of 50% after 14 days of the experiment, but after infection at 2–3 × 106 CFU all mice survived 14 days of the experiment (Wang et al. 2018). Phage therapy with φm18p at different MOI (i.e., 10, 1, and 0.1) protected mice infected with 2–3 × 108 CFU of KM18 (100% of mice survived 14 days of experiment). Moreover, levels of cytokines in mice infected with A. baumannii were high (tumor necrosis factor α [TNF-α] = 3000 pg/mL; interleukin 6 [IL-6] = 35000 pg/mL). However, mice treated with phage had low levels of TNF-α and IL-6 (250 pg/mL and 1000 pg/mL, respectively), even at the lowest MOI. This showed that the phage was able to increase the survival rate among mice infected with A. baumannii and also reduced the inflammatory response of infected mice.
All of these animal models have demonstrated that phage therapy can efficiently eliminate Acinetobacter infection, which creates an opportunity for future clinical application in humans.
-
The studies on phage PD-6A3 showed that it encoded Ply6A3 endolysin, which degraded peptidoglycan (forming a transparent ring) compared to a negative control (Wu et al. 2019). The phage (106–1010 PFU/mL) and endolysin (0.25–2.0 mg/mL) were incubated with HEK293T (human renal epithelial cells) and THP-1 (human mononuclear macrophage) cells for 8 and 24 h to study potential cytotoxicity of PD-6A3 and Ply6A3. The results showed that the level of viability for both types of cells with phage and endolysin was around 100% after both 8 and 24 h of incubation. This showed PD-6A3 and Ply6A3 safety of use. Moreover, the endolysin was able to lyse such bacterial strains as Staphylococcus aureus, Enterococcus faecium, Escherichia coli, and Klebsiella pneumonia.
Furthermore, Liu et al. (2019) reported that 10 out of 41 A. baumannii isolates were sensitive to depolymerase Dpo48. Activity of Dpo48 was examined under different pH conditions and the optimal pH was determined to be 5.0, although Dpo48 was active at pH from 5.0 to 9.0. The optimal temperature range for this depolymerase was 20 ℃–70 ℃. Additionally, the authors showed that overnight incubation of A. baumannii AB1610 (109 CFU/mL) with Dpo48 (10 μg/mL) and 1 h incubation with 50% human serum led to a five-log reduction of bacterial titer compared to the control with inactive serum. This showed the important role of complement combined with endolysin in bacterial killing.
-
Using a purified phage cocktail, Schooley et al. (2017) combined local and intravenous phage therapy application for the first time on a 68-year-old diabetic patient with necrotizing pancreatitis and pancreatic pseudocyst complicated by MDR A. baumannii infection. Three pathogenic A. baumannii strains (TP1, TP2, and TP3) were isolated from the patient's pancreatic drainage. Among 98 lytic bacteriophages against TP1, TP2, and TP3, nine phages were selected to obtain three phage cocktails. ΦPC cocktail was administered for 18 weeks from day 109 of the illness to drain the gallbladder, pseudocyst cavity, and third intra-abdominal cavity through percutaneous catheters. ΦPC contained AC4, C1P12, C2P21, and C2P24 Myoviridae phages against the TP1 strain. ΦIV cocktail was administered intravenously at 5 × 109 PFU for 16 weeks from day 111 of the illness. ΦIV contained ABNavy1, AB-Navy4, AB-Navy71, and AB-Navy97 Myoviridae phages against the TP1 strain. ΦIVB cocktail was administered intravenously at 5 × 109 PFU for 2 weeks from day 221 of the illness. ΦIV contained AB-Navy71 (Myoviridae) phage against the TP1 strain and AbTP3Φ1 (Podoviridae) against TP1, TP2, and TP3 strains. ΦPC, ΦIV, and ΦIVB contained 2.4 × 103, 5.89 × 103, and 1.64 × 103 EU/mL endotoxin, respectively. Phage therapy was supplemented with minocycline 4 days after the initial ΦPC treatment. Two days after ΦIV administration the patient's condition had improved and the patient awoke from a coma. Phage therapy was continued for the next 8 weeks and the patient left hospital on day 245. The above results indicate high potential for phage therapy to treat patients with a serious condition in whom MDR bacterial strains were detected. Furthermore, the phages can also restore the sensitivity of bacteria to antibiotics.
Ho et al. (2016) examined phage aerosol activity on A. baumannii strains in a hospital in Taiwan. According to this study, eight active phages were selected (ΦAB1, ΦAB2, ΦAB6, ΦAB7, Φ4C08, Φ8C07, ΦAB11, and Φ5C05), which were used sequentially 52 times. During the first period of the experiment (1.01.2012–28.02.2013), 191 new carbapenem-resistant A. baumannii strains were identified in intensive care units, and during the second period (1.03.2013–31.12.2013) the number of identified bacterial strains was 73. Cleaning with phage aerosol decreased the rate of new carbapenem-resistant A. baumannii from 8.57 to 5.11. Therefore, the use of phage aerosols may be of use for decontamination of hospital surfaces in the future.
-
Our experience with phage therapy is encouraging; good clinical results may be observed in many cases, while its safety appears to be unquestionable (Górski et al. 2018; Międzybrodzki et al. 2012). Furthermore, the first doubleblind, controlled, randomized clinical trial of phage therapy has recently confirmed phage therapy's safety and suggested that phage preparations even with very low phage titers may reduce the burden of bacteria in treated patients (Jault et al. 2018). Results of experimental phage therapy of A. baumannii infection in animals (including humans) are scarce, but very encouraging. Evidently, clinical trials are urgently needed to confirm the value of phage therapy against this pathogen in accordance with evidence-based medicine.
-
This work was supported by the statutory funds from the Hirszfeld Institute of Immunology and Experimental Therapy, Polish Academy of Sciences. The authors also thank to Norbert Łodej for making figure to this paper.
-
Andrzej Górski, is co-inventor of patents owned by the Institute and covering phage preparations. Other authors declare that they have no conflict of interest.
-
This article does not contain any studies with human or animal subjects performed by any of the authors.














 DownLoad:
DownLoad: