-
Dear Editor,
Duck virus hepatitis (DVH) is a significant concern in the duck industry as the disease causes a highly contagious infection in young ducklings that is often associated with liver necrosis, hemorrhage, and high mortality (Yugo et al. 2016). Duck hepatitis virus (DHV) was first described in 1949 on Long Island in the United States. Subsequent, outbreaks have been reported in England, Canada, Germany, Japan and elsewhere (Toth 1969). DHV is associated with at least two RNA viruses, duck hepatitis A virus (DHAV) and duck astrovirus (DAstV); however, no antigenic relationships have been identified between these two viruses (Yugo et al. 2016). DHAV is the primary causative agent of DVH. As the only member of the genus Avihep-atovirus, in the Picornaviridae family, DHAV has a linear, single-stranded positive-sense RNA genome. The genomic organization of DHAV is analogous to that of other picornaviruses with one large open reading frame (ORF) that encodes a polyprotein precursor, that is preceded by a 5'-untranslated-terminal-region (UTR) and followed by 3'- UTR (Tseng et al. 2007). Based on systematic phylogenetic analyses and neutralization assays, DHAVs have been classified into three serotypes: the classical serotype 1 (DHAV-1) (Kim et al. 2006; Ding and Zhang 2007; Tseng et al. 2007), the second serotype that has only been reported in Taiwan Province of China (DHAV-2) (Tseng and Tsai 2007), and the third serotype that was first reported in South Korea (DHAV-3) (Kim et al. 2007). DHAV-3 also accounts for an increasing proportion of DHV pathogens in China (Liu et al. 2011; Zhang et al. 2017; Wen et al. 2018), South Korea (Cha et al. 2013; Soliman et al. 2015) and Vietnam (Doan et al. 2016).
Previous studies have demonstrated that there is no cross-neutralization between DHAV-1 and DHAV-2 (Tseng and Tsai 2007) and that there is only limited crossneutralization between DHAV-1 and DHAV-3 (Kim et al. 2007). In many duck farms, DHAV-1 and DHAV-3 infections often occur at the same time, and their clinical symptoms and pathological developments are not easy to distinguish. Therefore, it is necessary to establish a rapid diagnostic technique for the simultaneous detection and differentiation of DHAV-1 and DHAV-3.
In the present study, we developed a SYBR Green I realtime RT-qPCR assay for the simultaneous detection and differentiation of DHAV-1 and DHAV-3 using amplicon melting curve analysis (MCA) of only one primer pair. The parameters were optimized, and the sensitivity, specificity and repeatability were evaluated. The diagnostic application of the assay was assessed on clinical samples.
At total of 77 published whole genome sequences (54 from DHAV-1 and 23 from DHAV-3) were retrieved from the GenBank database and aligned using the DNAStar (DNASTAR, Inc., United States) program. A highly conserved region located in the 5'-UTR was used to design primers for the detection and differentiation of DHAV-1 and DHAV-3. The optimized primers (5' UTR-F: 5' GTTGTGAAACGGATTACCGGTAGT 3'; 5' UTR-R: ACTCGACCAGCCGCGACCCTAT), yielded an expected PCR product of 202 base pairs that could bind all the DHAV-1 and DHAV-3 strains. In addition, a significant difference in the G+C content in the interior regions of the DHAV-1 and DHAV-3 sequences was used to facilitate an effective MCA. The SYBR Green-I RT-qPCR assay was conducted using the one step SYBR PrimeScript RT-PCR kit (Takara, China) following instructions in the kit manual.
To test the assay's sensitivity, two standard curves were generated with tenfold serially diluted DHAV-1 and DHAV-3 standard plasmids with concentrations of 1.0 × 101-1.0 × 109 copies/iL respectively. All the samples were tested in duplicate with three independentruns. As standard curve analysis demonstrated that when the copy numbers were in the range of 102 to 109 copies/^L, the efficiency of the qPCR reaction for DHAV-1 and DHAV-3 was 100% and 98%, respectively. The correlation coefficient R2 for the linear regression equation of DHAV-1 and DHAV-3 was 0.999 and 0.997, respectively (Fig. 1A and 1B). Furthermore, the detection limits forboth DHAV-1 and DHAV-3 were 10 copies/μL. The melting curves both of DHAV-1 and DHAV-3 displayed single peaks and had consistent Tm values of approximately 86.15 ℃ and 83.86 ℃, respectively(Fig. 1C).
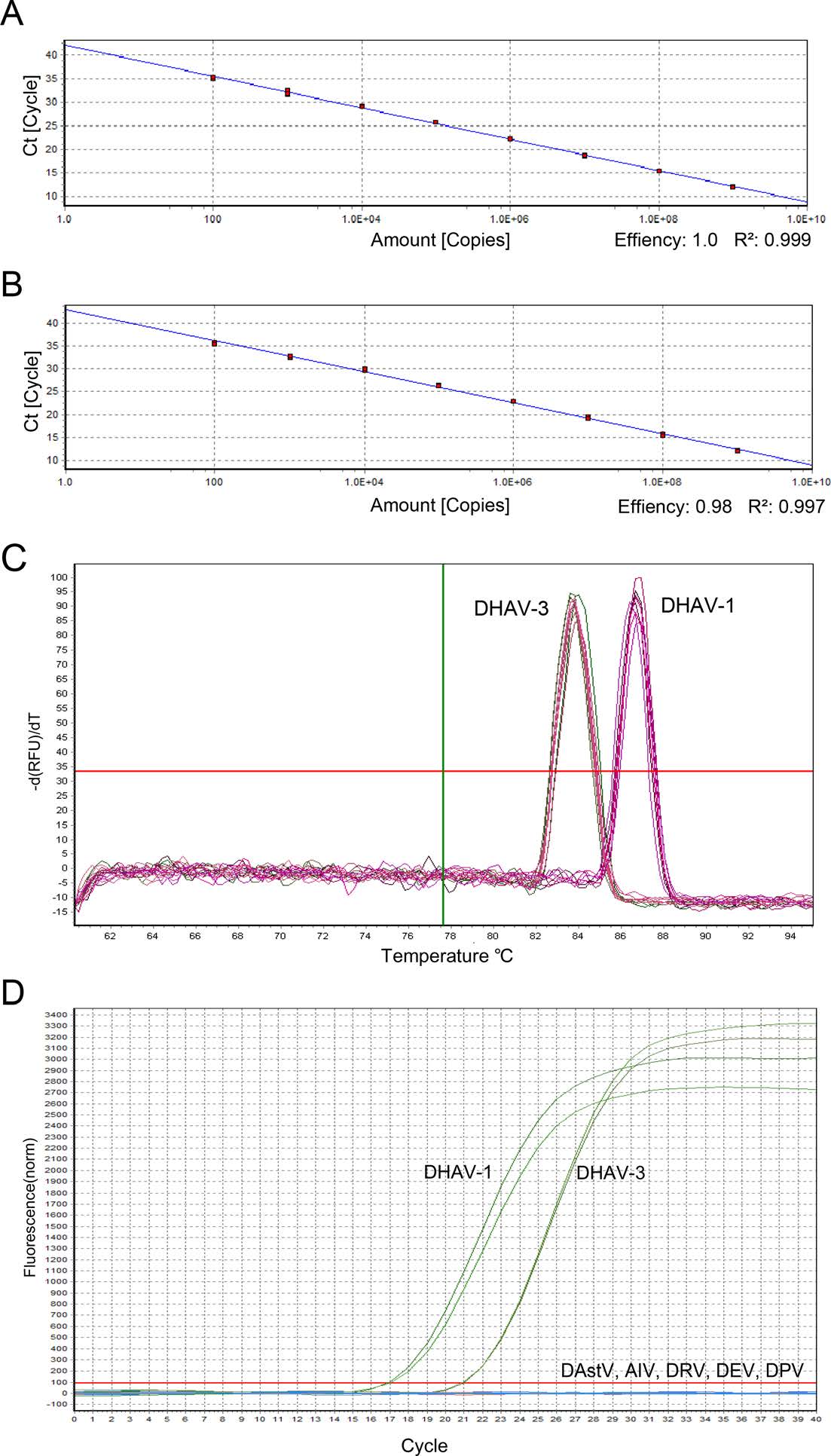
Figure 1. Simultaneous detection and differentiation of duck hepatitis A virus type 1 and 3 through melting curve analysis by one pair of primer. A, B Standard curves of real-time PCR for DHAV-1 and DHAV-3. Tenfold diluted standard plasmid of DHAV-1 and DHAV-3 tested with same pair of primer. Regression lines between the Ct values and the input concentrations of DHAV-1 (A) and DHAV-3 (B) plasmid DNA using SYBR Green I real-time PCR. C Melting curve analysis of DHAV-1 and DHAV-3. Amplification melting curve and melting temperature values (Tm) after SYBR Green I realtime PCR followed by melting curve analysis of DHAV-1 and DHAV-3. D Specificity of realtime PCR assay. The specific fluorescent signals were detected from cDNA of DHAV-1 and 3, and the dissociation curves showed that there were two specific product peaks for DHAV-1 and DHAV-3, respectively, but no specific amplification for negative control (DAstV, AIV, DRV, DEV and DPV).
To test the assay's specificity, DAstV, avian influenza virus (AIV), duck reovirus (DRV), duck virus enteritis virus (DEV), and duck parvovirus (DPV) genomic cDNA or DNA are used as templates in the SYBR Green I real time RT-qPCR assay, using the aforementioned reaction conditions (Fig. 1D). No consistently positive signals were observed for any of these samples, suggesting that the realtime RT-qPCR assay was specific for DHAV.
To assess the reproducibility of the RT-qPCR assay, the Ct values for two standard plasmids (DHAV-1 and DHAV-3, 103 to 107 plasmid copies/^L) were detected in inter and intra-assays that were performed in triplicate. As shown in Table 1, the coefficients of variation (CV) of the CT values were less than 5%. Specifically, the CV of the intra- and inter-assays ranged from 0.47% to 0.69% and from 0.51% to 1.5% for the DHAV-1 plasmids and, from 0.70% to 1.40% and from 0.34% to 0.87% for the DHAV-3 plasmids, respectively.
No. of DNA copies Intra-assay Inter-assay Ct (mean±SD) CV (%) Ct (mean±SD) CV (%) DHAV-1 1.0 × 107 18.43 ± 0.31 0.51 18.51 ± 0.087 0.47 1.0 × 105 25.07 ± 0.20 0.79 25.19 ± 0.24 0.95 1.0 × 103 28.40 ± 0.44 1.5 28.62 ± 0.20 0.69 DHAV-3 1.0 × 107 18.28 ± 0.16 0.87 18.21 ± 0.26 1.4 1.0 × 105 25.29 ± 0.15 0.59 25.22 ± 0.12 0.47 1.0 × 103 28.80 ± 0.10 0.34 28.56 ± 0.20 0.70 Table 1. Intra- and inter- assay reproducibility of SYBR Green I Real-time PCR.
To assess the assay's performance in a clinical setting, 40 liver samples from sick ducklings suspected of duck hepatitis virus infection were analyzed using the developed RT-qPCR assay and a traditional single RT-PCR assay. All the samples were obtained from duck farms located in the Zhejiang and Anhui provinces. The samples were mixed and ground into homogenates. All the positive clinical samples that were detected RT- qPCR assay were confirmed by DNA sequencing of the single RT-PCR products using the same specific primer. The DHAV-infected liver samples were characterized by sequencing the VP1 gene. The results of the two assays were 100% consistent, and both methods showed that 16 of the examined samples contained DHAV-1, while 24 contained DHAV-3.
Outbreaks with DHAV-1 and DHAV-3 at the same areas have recently been reported in China and South Korea (Gan et al. 2014; Soliman et al. 2015; Lin et al. 2016; Zhang et al. 2017; Wen et al. 2018). Since antibodies to DHAV-1 and DHAV-3 have almost no cross-neutralization ability, only ducklings injected with the genotype matched vaccine will receive protection. (Kim et al. 2007). Therefore, the new assay meets the clinical requirements for an accurate, rapid, sensitive and reliable method to detect and distinguish DHAV-1 and DHAV-3. Although, several diagnostic methods have been developed to detect and distinguish DHAV-1 and DHAV-3 based on RT-PCR and RT-qPCR (Yang et al. 2008; Huang et al. 2012; Chen et al. 2013; Hu et al. 2016; Chen et al. 2019).The previous differentiation methods required at two primer pairs, increasing the experiment's complexity. We have developed and validated a SYBR Green I real time RT-qPCR assay, which uses a single unlabeled primer pair to amplify a conserved region of 5' -UTR of DHAV.
Our experiments demonstrated that, the primer pair in this assay was able to detect virus samples in a range of concentrations. Also, this assay has no cross-reactivity DAstV, AIV, DRV, DEV, or DPV. Importantly, this diagnostic method can also distinguish DHAV-1 and DHAV-3 using MCA, based on the different Tm values for each genotype's amplicons. The results obtained by this diagnostic method were reproducible and can be easily applied to the clinical settings.
In conclusion, in this study, we developed an MCA- based SYBR Green I RT-qPCR assay to detect and differentiate DHAV-1 and DHAV-3. This new assay is rapid, sensitive, specific, cost-effective and easy to conduct. To our knowledge, this is the first description of a multiplex RT-qPCR that can detect and distinguish DHAV-1 and DHAV-3 in the same tube using one primer pair.
HTML
-
This work was supported by the National Natural Science Foundation of China Grants 31300141 (to CCM) and Shanghai Key Laboratory of Veterinary Biotechnology Grants klab201702 (to CCM).
-
The authors declare that they have no conflict of interest.
-
This research was approved by the Ethics Committee of Shanghai Veterinary Research Institute, Chinese Academy of Agricultural Sciences. The international guidelines for the care and use of animals have been followed.













 DownLoad:
DownLoad: