-
Hepatitis B virus (HBV) is the major etiologic agent of non-A non-C hepatitis. Approximately 400 million people are chronically infected with HBV and a death toll of 1-2 million people worldwide. In China alone, there are 170 million infected people (1-4) and many chronic carriers develop cirrhosis and hepatocellular carcinoma (5). Antiviral therapy of patients with chronic HBV infection aims to clear the HBV DNA in the peripheral blood. The nucleoside analogue Lamivudine, [(-)β-L-2', 3'-dideoxy-3'-thiacytidine, 3TC], is currently approved as anti-hepadnavirus drug for treatment of chronic hepatitis B targeting polymerase gene in HBV genome. Lamivudine has been shown to inhibit viral DNA synthesis (6) and has become the first agent worldwide in the treatment of chronic hepatitis B. Chinese patients are immunotolerant to interferon alpha because of acquisition of the disease during early childhood. The efficacy of interferon alpha in Chinese individuals is lower than that in patients of European origin (7). However, the efficacy of lamivudine is the same for both races. Clinical practice in the treatment of hepatitis B with lamivudine showed a drastic decrease of virus load even to an undetectable level. Similarly, the pathology of the liver showed improvement as indicated by the alanine/aspartate aminotransferases (ALT/AST) activity (6, 8, 9). Despite a safe profile and initial efficacy, lamivudine has no effects on HBV covalently closed circular DNA (cccDNA) and many patients need long period of antiviral therapy in order to exhaust the HBV DNA and eliminate the serum HBV DNA (10). Lamivudine-induced reduction in viral load is difficult to sustain over time due to the occurrence of viral drug resistance. Thus, the antiviral effects of the drug are gradually reversed in most cases and ensues outbreak of the infection. HBV polymerase gene seldom mutates under natural conditions. However, after 6 months of antiviral mono-therapy with lamivudine, there was a significant risk in the emergence of drug resistant HBV (8, 9). With continued application of the drug, the virus rebounded rapidly after developing resistance along with a decrease in drug competence to inhibit HBV DNA synthesis. Sequence analysis revealed the emergence of a specific mutation in the tyrosine-methionine-aspartate-aspartate (YMDD) motif of the polymerase gene under the selective pressure of lamivudine. YMDD motif, which is a highly conserved domain of the reverse transcriptase and is required for polymerase activity. In the mutated motif, the methionine is replaced by either an isoleucine or a valine (8, 9). There was an emergence of 16%-32% genotypic resistance to lamivudine after one year of treatment, 47%-56% after 2 years and 69%-75% after 3 years (11). To date, there is no report on the relationship between lamivudine selective pressure and virus evolution, the constitution of YMDD wild and mutation virus populations in sera of chronic carrier, YMDD motif evolutionary trait and its mechanism under the selective pressure of lamivudine. FT-RDB assay is a rapid, accurate, specific, reliable, versatile and an inexpensive method to detect the lamivudine resistance associated with mutations in human HBV genome (12). In this study we used FT-RDB to detect the constitutional changes of HBV YMDD wild and mutation motif in the HBV genome in sera of patients treated with lamivudine.
HTML
-
Sera from 1 021 patients (215 females, 806 males; mean age: 29.7 years; age range: 17 to 61) undergoing treatment with lamivudine were collected. The patients in the Guangzhou Liver Hospital and the First Affiliated Hospital of Sun Yat-Sen University (Guangzhou, Southern China) were clinically suspect of lamivudine resistance. The patients were selected according to the following criteria: (ⅰ) They were HBV positive for at least 2 years; (ⅱ) They had viral load of 1×103 to 1×108 copies/mL in the serum assayed with an HBV quantitative fluorescence PCR detection kit from DaAn Gene Corporation (Guangzhou, China) and higher ALT/AST levels, showing evidence of treatment failure with lamivudine; (ⅲ) For all patients, lamivudine had been used for at least 8 months (mean, 19.1; range, 8-50). In addition, we also extracted the corresponding archived sera before lamivudine treatment.
-
We aligned the HBV genome sequences in international DNA data banks (Genbank/EMBL/DDBJ) and selected the region conserved all genomes. Primers and probes were designed with Primer Express 2.0 (Applied Biosystem, Inc., Co), DNAMAN 4.0 software (Lynnon BioSoft) and Oligo 6.31 (Molecular Biology Insights, Inc., Co). The primers and probes (Table 1) were labeled with different groups: the reverse primer was labeled with biotin at 5' end and the hybridization probes were labeled with amino group at 5' end. In addition, we designed a color control probe (Table 1) for the operating control where a biotin was labeled at the 5' end and an amino group at the 3' end.

Table 1. Preferentially Selected PCR Primers for Amplification of YMDD Motif and Probes for RDB Analysis
-
HBV DNA was extracted from serum according to the report of Günther et al (13) with some modifications.
-
HBV DNA isolated from 15 μL serum was amplified by PCR in 50 μl of buffer containing 50 mmol/L Tris-HCl (pH 8.3), 50 mmol/L KCl, 2 mmol/L MgCl2, 0.01% gelatine, 200 μmol/L dNTPs, 2 U of Taq DNA polymerase (TaKaRa, Japan), and 0.2 μmol/L of each primer. In order to prevent contamination, we replaced dTTP with dUTP and added 0.5 U of Uracil-DNA Glycosylase in the PCR reaction system. The amplification was performed with an Applied Biosystems 9700 thermal cycler (Perkin-Elmer) under the following conditions: incubating at 50℃ for 3 min before an initial denaturation step at 95℃ for 5 min, 40 cycles of 93℃ for 30 s, 55℃ for 30 s, and 72℃ for 35 s. A final extension was performed at 72℃ for 5 min.
-
The PCR products were purified by phenol-chloroform (1:1) extraction, precipitated, dissolved in double distilled water and fragments of 233 bp were cloned into pMD 18-T simple vector (TaKaRa, Japan) according to the manufacturer's instructions. Cloned plasmids were authenticated by sequencing at BioAsia Corporation (Shanghai, China).
-
"Flow-through RDB", was carried out as described previously by Ou et al. (12) and performed on the KaiPuTM DNA HybriMax Rapid Hybridization Machine (HongKong DNA Ltd, HongKong, China). Briefly, the steps entailed: 1) preparation of hybrid membrane strip (Bigdye C membrane, Pall Co. USA; 2) denaturing of the PCR product; 3) pre-hybridizing the membrane; 4) hybridization of the target PCR product with the specific probe; 5) washing the membrane to eliminate un-hybridized products; 6) reaction of peroxidase (POD) with the biotin group on the PCR products; 7) washing the membrane to eliminate the un-combined POD; 8) coloring with 3, 3', 5, 5; -TetraMethylBenzidine (TMB) chromogen. Positive and negative controls were set for each test. The probe concentration was 5 μmol/L and the hybridization temperature was 42℃.
-
Data were analyzed with the statistical packages SPSS 11.0 (SPSS Inc., Chicago, Illinois, USA). Numerical data were assessed by the non-parametric Mann-Whitney two-sided U-test for sample comparison analysis.
Patients
Primer and probe design
Serum HBV DNA extraction
PCR amplification
Cloning and sequencing of the PCR products
Flow-through RDB analysis
Statistical analysis
-
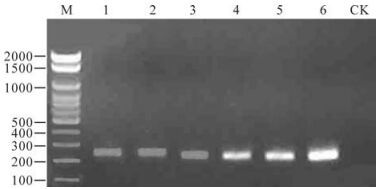
The data presented in Fig. 1 show that DNA extracted from the serum (103-108 copies/mL) and the positive template plasmid can be amplified specifically and efficiently with the primer pair. The length of the PCR products was 233 bp.
-
We have sequenced the PCR products and the plasmid clone containing HBV YMDD, YIDD and YVDD motifs. The sequence of HBV YMDD, YIDD and YVDD motif contained Atg, Att and Gtg, respectively (Fig. 2), which were in agreement with the data generated by our design strategy.
-
Various profiles of infection were found in the 1021 sera assayed, including single infection with viruses containing YMDD, YIDD, YVDD and mixed infections with YMDD+YIDD, YIDD+YVDD, YMDD+ YVDD and YMDD+YIDD+YVDD infections.
-
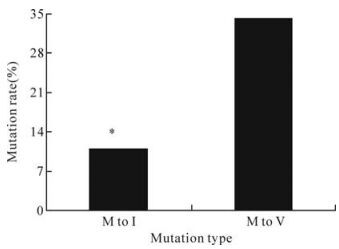
In the sera from 1021 highly suspected HBV lamivudine-resistant patients before and after lamivudine treatment, seven YMDD wild and mutated profiles were found. In 310 of mutated cases, 85 YMDD were mutated to YIDD and 266 YMDD were mutated to YVDD (Table 2). Before lamivudine treatment, the rates of YMDD, YIDD, YVDD, YMDD+YIDD, YMDD+YVDD, YIDD+YVDD and YMDD+YIDD+YVDD infection were 36.93%, 6.07%, 17.04%, 0.97%, 33.99%, 0.98% and 4.02%, respectively, but were 8.03%, 7.93%, 19.98%, 1.96%, 51.62%, 1.96% and 8.52%, respectively, after treatment for more than 8 months (Table 3). In total, infections including YIDD type increased slightly from 12.04% to 20.37%, YVDD type increased distinctly from 56.03% to 82.08%, and YMDD wild type decreased from 36.93% to 8.03% after more than 8 months of treatment, indicating the change from YMDD to YVDD but not to YIDD was the dominant mutation, i.e. the infection including YVDD motif increased dominantly under the selective pressure of lamivudine. The mutation rate of YMDD motif to YVDD motif which was 34.32% was significantly higher than that of YMDD to YIDD, which was only 10.97% (Fig. 3) (U=10.98, P < 0.05), as assessed by the non-parametric Mann-Whitney U-test.

Table 2. Infection cases on serum HBV YMDD wild and mutant type pre and post lamivudine treatment

Table 3. Serum HBV YMDD wild and mutant type infection pre and post lamivudine treatment
Serum and plasmids DNA PCR
Positive template plasmid sequencing
FT-RDB profiles of different serum HBV DNA
FT-RDB results of different serum HBV YMDD motifs pre and post lamivudine treatment
-
We have demonstrated that the FT-RDB assay can be successfully used to identifying the single or mixed infection with HBV YMDD wild and mutation motif types. We used the KaiPuTM DNA HybriMax Rapid Hybridization Machine, which is designed based on the particular principle of "flow-through hybridization" and the traditional RDB. The rapid method also offered an markedly enhanced efficiency in comparison to the traditional RDB assay that normally requires at least 4 h to complete. Our procedure required only 5-10 minutes to complete. The improved RDB method is also characterized by fast, clean, most versatile and least expensive. Nucleic acid hybridization occurs in a small volume rather than the conventional two-dimensional surface; the hybridization can be done with very small samples and reagent volumes, hence reduces the running cost. In this study, we designed and optimized not only the specific and scientific probes for the specific virus type but also the color control probe for the hybridization, indicating the method has a perfect quality control. The analysis can provide personalized data to guide the clinical hepatitis B treatment with lamivudine. The method can be used not only for genotyping of a large number of clinical samples but also for detecting single base mutation. (15 or more samples can be detected simultaneously and more than 24 probes can be integrated in a small membrane). All probes are integrated in a small hybridization membrane just like a low density microarray, making clinical high-throughput detection possible.
Lamivudine has been used in the treatment of chronic hepatitis B infections and it targets the polymerase gene in HBV genome. The HBV polymerase gene seldom mutates under natural conditions, but drug resistance develope after a long period of lamivudine therapy. Emergence of lamivudine resistance has been suggested by the association of increased viral load with the selective pressure of lamivudine and the mutations in YMDD motif. The assay of 1021 sera from highly suspected lamivudine resistant patients showed that there were 7 profiles of virus infections in sera characterized by dynamic changes (Table 2, Table 3). The mixed infection indicates that there were different components of the quasispecies co-existing in the serum of chronic carriers. The infection of different YMDD wild and mutation types was in agreement with the character of quasispecies populations (14), whichproved that HBV populations in sera were adapting to the environment. With the selective pressure of lamivudine, HBV YMDD wild type is supressed with a gradual increase in the YIDD and YVDD types. After more than 8 months of lamivudine treatment in the Guangzhou area, YVDD increased dominantly (Table 3), YMDD and YVDD mixed infection accounted for 51.62% and the infections including YVDD of 82.08%, which were 17.63% and 26.05% higher, respectively, than that of pre-treatment. The data in Fig. 3 show that 34.32% HBV YMDD motif mutated to YVDD, and only 10.97% YMDD mutated to YIDD. Our finding indicates the mutated type YVDD had an advantageous evolutionary trait and became predominant after 8 months or more of lamivudine treatment. Our results further confirmed that the functional or structural constraints associated with the substitution M5521 seem to be less compared to M552V (15).
Our data show that lamivudine had little effect on some patients who already had the YMDD phenotype as detected in the archived pre-treatment sera. With the use of lamivudine on carriers, often the HBV YMDD wild type changed to YIDD or YVDD type partly or completely (Table 2), which indicated lamivudine is responsible for the mutation and appears to be a selection pressure in HBV evolution, possibly affecting the replicative capacity of HBV variants. The HBV genome is partly double-stranded DNA that replicates via reverse transcription of an RNA intermediate (pregenomic RNA). Because of the lack of proof-reading by reverse transcriptase and the low efficacy of available therapies in eliminating HBV DNA, the spontaneous mutation rate during HBV replication is high (16). HBV is extremely prolific, generating up to 1010-1012 virions/day and mutants emerge because of the selective pressure exerted by long-term use of lamivudine (17; 18). Clearly, escape mutants are generated under the selective pressure of lamivudine. HBV YMDD motif variability plays a crucial role in escaping the host immune surveillance and establishing persistent infections under the selective pressure of lamivudine. When mutations occurs, the configuration of the wild-type YMDD motif becomes altered in such a way that the effectiveness of the drug is diminished with the appearance of resistant virus strains YIDD and YVDD in the infected livers. Perhaps YVDD motif has less evolutionary pressure than YIDD motif and finally YVDD population evolves into the dominant resistant population. The detailed mechanisms will be further pursued. In addition, our study also provided strong evidence that we should develop a new antihepadnavirus agents that directly inhibit the synthesis of HBV genome containing the YVDD motif.














 DownLoad:
DownLoad: