-
Filovirus family consists of Ebola virus and Marburg virus which can cause severe hemorrhagic fever in human and non-human primates with high mortality. Ebola viruses are classified into five species named after the location of their first outbreaks: Zaire, Sudan, Ivory Coast, Bundibugyo, and Reston [46]. Three species of fruit bats, Hypsignathus monstrosus, Epomops franqueti, Myonycteris torquata, captured near the sites of 2001 and 2005 outbreaks of Ebola, are implicated as the natural reservoir of Ebola virus[20, 21]. Ebola virus infects humans and non-human primates through direct contact with body fluid, while aerosol transmission has also been suggested[19]. Since there are no vaccines or therapeutics available for humans, Ebola virus has become one of the major concerns on bio-terrorism.
Ebola viruses are enveloped viruses with an ~19 kb single-stranded RNA genome containing 7 genes. The GP gene encodes two glycoproteins, a secreted form called sGP and a transmembrane form called GP which is generated by RNA editing[34, 35, 44]. GP is responsible for viral entry, including attachment of viruses to the target cells and fusion of the virus-cell membranes[43, 48, 49]. GP is modified by N-linked glycosylation in the Endoplasmic Reticulum to form PreGP, and further undergoes O-linked glycosylation in Golgi to become mature GP[10].
Ebola GP processing by proteases has been well documented. First, the N-terminal signal peptide (32 residues) of GP is cleaved cotranslationally. The GP precursor is then cleaved into GP1 and GP2 in trans-Golgi by a furin-like cellular protease, and GP1 and GP2 are linked by disulfide bonding[14, 36, 45]. Although the cleavage site between GP1 and GP2 is conserved among the Ebola species, this cleavage event appears not required for Ebola entry[13, 28, 29, 49]. Lastly, GP was shown to be cleaved at D637 by the tumor necrosis factor a-converting enzyme (TACE) to shed the truncated GP from the surface of infected cells[7]. In addition to these cleavage events during GP maturation, two intracellular proteases, Cathepsin B and L, residing in the endosomes, have been shown to process GP1 into an 18-19 kDa fragment during viral entry[5, 16, 37]. The endosomal cleavage appears to be critical on viral entry. It is interesting to note that these endosomal proteases play a role in entry of other viruses[9, 12, 30, 31, 39]
Many cellular proteins have been implicated in facilitating Ebola entry, including asialoglycoprotein receptor, folate receptor a, dendritic cell-specific ICAM3 grabbing non-integrin, liver/lymph node-specific ICAM3 grabbing non-integrin, human macrophage galactose and N-acetylgalactosamine-specific C-type lectin, and Tyro3 receptor kinase family [1, 4, 8, 22, 32, 33, 38, 42]. However, none of them seems to be absolutely required for the entry. Interestingly, expression of Ebola GP on the target cells can specifically enhance Ebola GP-mediated viral entry [23].
The N-terminal 200 residues of GP1 are relatively conserved compared to the highly variable C-terminal mucin-like domain (MUC, amino acids 309~476). The MUC region is heavily modified by O-and N-linked glycosylation. It has been shown that MUC is responsible for the cytopathic effects induced by GP expression, but it is dispensable for GP-mediated viral entry, at least in tissue culture using pseudotyping systems[14, 24, 40, 50]. Previous studies have shown that the N-terminal region of GP1 (approximately 150 residues in length after cleavage of the signal peptide) is critical for receptor binding, which is referred to as the receptor-binding domain, or RBD[3, 24]. Extensive mutational analyses have been performed to charac-terize the roles of RBD residues in protein folding and function in viral entry. In this report, we examined the roles of the RBD residues by substitutions in protein expression, virion incorporation, and viral entry[3, 14, 24, 26]. By entry interference assay and binding assay, we identified several key residues involved in receptor binding and the post-binding steps in viral entry. In addition, the results further substantiated the notion that Ebola and Marburg viruses use the same or similar receptor.
HTML
-
Cell lines and antibodies
Human embryonic kidney cells 293T were maintained in Dulbecco's modified Eagles Medium supplemented with 10% fetal bovine serum with penicillin and streptomycin plus 300 μg/mL Geneticin. The mouse monoclonal antibody, 12B5-1-1, which recognizes the GP1 of Ebola Zaire GP, was kindly provided by Dr. Mary K. Hart[47]. The mouse anti-HIV p24 monoclonal antibody was obtained from NIH AIDS Research and Reference Reagent Program[41]. Mouse anti-b actin antibody, goat anti-mouse IgG HRP, and anti-human IgG FITC were purchased from Sigma (St. Louis).
Mutagenesis of Ebola glycoprotein gene
Ebola Zaire glycoprotein gene was synthesized by multiple rounds of overlapping PCR based on the Ebola Zaire genome sequence (GenBank accession number L11365[15]).ΔMucin GP was constructed by PCR-directed mutagenesis. All alanine substitution mutations of GP gene were generated by site-directed mutagenesis using Stratagene's Quick-Change Mutagenesis kit according to supplier's protocols. All mutations were confirmed by DNA sequencing of flanking regions (on average approximately 500 bp).
Pseudotyping
Pseudotyped viruses were produced by cotansfecting DNA of wt GP or mutants with Env-deficient HIV vector carrying a firefly luciferase reporter gene[6, 11] into producer cells. Briefly, 2μg DNA of wt GP or mutants and 2μg of pNL4-3-Luc-R--E- were used to transfect 293T cells (90% confluent) in six-well plates by Lipofectamine 2000 according to supplier's protocol (Invitrogen). To produce other pseudotyped viruses, vesicular stomatis virus protein G (VSV-G), or Marburg GP, instead of Ebola GP was co-transfected with HIV vector. The supernatants containing the pseudotyped viruses were collected twice (24 h and 48 h post-transfection, respectively) and combined, clarified from floating cells and cell debris by low-speed centrifugation and filtered through 0.45μm filter (Nalgene). Then 1 mL supernatant was used to infect cells and the rest was stored at -80℃ until use.
Western blotting
To evaluate the Ebola GP expression, the producer cells (293T) were lysed in 0.2 mL Triton X-100 lysis buffer (50 mmol/L Tris-HCl pH7.5, 150 mmol/L NaCl, 5 mmol/L EDTA, 1% Triton X-100 and protease inhibitor cocktail: 10μg/mL leupeptin, 5μg/mL aprotinin and 2mmol/L phenylmethylsulfonyl fluoride) 48 h post-transfection. The protein samples were subjected to SDS-PAGE, and transferred to PVDF membrane. The membrane was first incubated with anti-Ebola GP1 monoclonal antibody 12B5-1-1(1:5 000 dilution) for one hour, and then probed with peroxidase-conjugated goat anti-mouse antiserum (dilution 1:20 000) for one hour. The bands were visualized by the chemiluminescence method according to supplier's protocol (Pierce). As a loading control, b actin in transduced cells of the entry interference assay was visualized by Western blotting similar as described above except that the anti-b actin antibody (1:10 000) was used as the primary antibody.
Detection of glycoprotein incorporation in pseudotyped viruses
To evaluate the incorporation of wt GP protein or mutants into the pseudotyped viruses, 2 mL of pseudotyped viruses was layered onto a 3mL cushion of 20% sucrose in PBS (w/v) and centrifuged at 55 000 r/min for 30 min. Pelleted pseudotyped viruses were lysed in 50μL Triton X-100 lysis buffer and 25 μL sample was subjected to SDS-PAGE. Expression of Ebola GP protein or mutants was detected by Western blotting as described above. Mouse anti-HIV p24 monoclonal antibody (1:5 000 dilution) was used as the primary antibody to detect the HIV p24 protein.
Infectivity assay of pseudotyped viruses
Human 293T cells (3×105 cells) were seeded in six-well plates one day prior to infection. These targeted cells were incubated with 1 mL of the pseudotyped viruses for 5 h. The cells were lysed in 200 μL of cell culture lysis reagent (Promega) 48 h post-infection. The luciferase activity was measured using the luciferase assay kit (Promega) and a FB12 luminometer (Berthold Detection System) according to the supplier's protocol. Each experiment was in triplicate and repeated at least three times.
Entry interference assay
To construct plasmids carrying a Δmucin GP gene (with mutation), PshAI-BamHI fragment of full-length Ebola GP gene (with mutation) in pcDNA3.1(+) was replaced by PshAI-BamH I fragment of Δmucin Ebola GP gene. Δmucin GP gene with/without mutation in these plasmids was amplified by PCR and inserted between BamH I and Xho I sites of the HIV vector, pHR'-CMV-Luc [equal to pHR' in [27]], to replace luciferase gene. GFP or puromycin-resistant gene (puro) was used to replace the luciferase gene in the same vector to construct pHR'-CMV-GFP and pHR'-CMV-puro.
Human 293T cells in 6-well plate were transfected with 0.25 μg of VSV-G plasmid encoding VSV-G protein, 1μg of HIV-trans plamsid encoding HIV structural protein gag-pol [equal to pCMVDR8.2 in [27]], and 1 μg of pHR'.CMV plasmid carrying different genes (GFP, puro, or wt/mutant Δmucin GP). The supernatants containing pseudotyped viruses were collected at 48 h post-transfection and filtered as above. The viruses were incubated with 293T cells in 6-well plate and the media were replaced with fresh media after overnight incubation. Forty-eight hours later, the transduced cells expressing different proteins (GFP, puro, or wt/mutant Δmucin GP) were seeded into 12-well plates and incubated with VSV-G, Ebola GP, or Marburg GP pseudotyped pHR'-CMV-Luc viruses carrying the luciferase reporter. Forty-eight hours later the cells were lysed and luciferase activity was measured as described above to determine the effect of expression of these proteins (GFP, puro, or wt/mutant Δmucin GP) on entry of different pseudoty-ped viruses (VSV-G, Ebola GP, or Marburg GP).
Cell surface binding assay
To study direct binding of Ebola GP to cells, the gene encoding a fusion protein (Ebola receptor-binding domain [RBD] fused to Fc of human IgG) was constructed and the fusion protein was purified. Nhe I-BamH I fragment of S1-hIgG[17] was replaced with PCR product of Ebola RBD (residue 54-201) fragment. Plasmids of wt or mutant Ebola RBD-hIgG were transfected into 293T cells using the calcium phosphate method. After overnight incubation, the transfection mix was replaced by protein-free VP-SFM media supplemented with 4 mmol/L L-glutamine (Gibco). Supernatants were collected twice at 48 and 72 h post-transfection and filtered through a 0.45mmol/L membrane (Millipore). The supernatants were applied to a column of protein A beads (Santa Cruz Biotechnology) followed by three washings with 10 mL PBS. The proteins were eluted three times with 1 mL 0.1 mol/L glycine (pH 2.8) and immediately neutralized with 60 μL Tris (pH 8.0) each time. Then the proteins were dialyzed in 3.5K Slide-A-Lyzer Dialysis Cassettes (Pierce) and concentrated by centrifuging in YM-10 Centricon (Millipore). The protein concentrations were measured using the BCA Protein Assay Kit (Pierce).
Different amounts of the purified proteins (0, 0.25, 0.5, 0.75, 1.0 or 2.0 μg) were incubated with 2×105 293T cells in a 50 μL volume of PBS/2% BSA on ice for 1 h. Then the cells were washed twice with PBS/2%BSA and incubated with 0.5 μL anti-human antibody conjugated with FITC (Sigma) in a 100mL volume of PBS/2%BSA on ice for 0.5 h. Then cells were washed three times with PBS/2%BSA and subjected to flow cytometry. FITC-positive cells were counted as indicators of the binding of protein to cells.
-
Alanine scanning analysis of the receptor-binding domain of Ebola GP
Previous work on the conserved hydrophobic and charged residues in GP1 has shown that most of the residues involved in protein folding or viral entry are at the N-terminus. This suggests that the N-terminal region of approximately 150 residues (aa 33-185) consists of an important domain of GP, receptor-binding domain, or RBD [24]. To further dissect RBD in viral entry, mutational analysis was carried out to study other residues in this domain. In total, 41 single alanine substitutions were generated on Ebola GP. The wild type (wt) or mutant GP plasmid was co-transfected with an Env-deficient HIV vector (pNL4-3 Luc.R-E-) into 293T cells to produce pseudotyped viruses as described[24]. 293T cells were challenged by these viruses. Infected cells were lysed 48 h post-infection, and luciferase activities were measured as an indicator of entry efficiency of different pseudotyped viruses, as previously described[24]. Western blot analysis was used to determine protein expression in 293T cells and GP incorporation on HIV particles.
Only four of the forty-one substitutions (V52A, V66A, F160A, and V181A) appeared to show low levels of protein expression and/or virion incor-poration, which led to impaired GP phenotype in mediating viral entry (Fig. 1). The lower amount of V52A, V66A, and V181A GP incorporated onto virions compared to that of wt GP could explain the inefficient viral entry (29%, 44%, and 15% of wt). Low expression of F160A GP led to low virion incorporation and viral entry (2% of wt). In contrast, none of the remaining 37 mutant GPs showed a major defect in protein expression and GP incorporation. Among them, two (T60A and V79A) were defective in mediating viral entry (~30% of wt), suggesting that T60 and V79 are involved in receptor binding or post-binding steps. In addition, two substitutions (T42A and N61A) did not have major effect on GP expression and viral incorporation, but they appeared to be somewhat defective in mediating viral entry (~40% of wt, Fig. 1 C and D). In contrast, the remaining 33 substitutions did not greatly impair the ability of GP in mediating viral entry (more than 60% of wt). Indeed six of them (V96A, S119A, F151A, E156A, S167A, and T168A) displayed enhanced entry (over 200% of wt).
Figure 1. Analysis of alanine substitution mutants. Individual GP mutant were co-transfected into 293T cells with HIV vector to produce pseudotyped viruses to infect target cells (293T). Western blotting was performed to determine GP expression and incorporation onto pseudotype virions. (A, B, E, and F) GP expression and incorporation onto virions. Mock, only HIV vector was transfected to produce Envelope-deficient viruses. (C, D, G, and H) Relative infectivity of the pseudotyped viruses. The infectivity of wt GP-pseudotyped virus was expressed as 100%, while infectivity of mutant GP-pseudotyped viruses were expressed as the percentage of wt. Bars represent standard deviations calculated from at least three independent repeats
Analysis of double mutants
Among the 41 substitution mutants of GP characteri-zed above, four (T42A, T60A, N61A, and V79A) behaved like wt GP in protein expression and viral incorporation, but were defective in mediating viral entry, displaying less than 50% of wt GP in mediating viral entry (measured as RLUs, see Fig. 1 C and D). Previously, we showed that four substitution mutants in this region (L43A, R54A, K56A, and E100A) displayed similar phenotypes, that is, they were expressed and incorporated onto virions like wt GP, but somewhat defective in mediating viral entry[24]. To further analyze the role of these residues in viral entry, fifteen double substitution mutants were generated. Each of these mutants was characterized in protein expression, viral incorporation, and in mediating viral entry. Five mutants (V79A/L43A, V79A/L51A, V79A/ R54A, V79A/E100A, and T60A/L51A) displayed defects either in protein expression or incorporation onto viral particles (Fig. 2A), thus. leading to the reduced ability of GP in mediating viral entry (Fig. 2B). In contrast, the remaining 10 double substitution mutants did not display detectable defects in protein expression or viral incorporation, but they were impaired in mediating viral entry, ranging from 2% to 44% of wt GP (Fig. 2). Among them, five double substitutions (V79A/K56A, V79A/N61A, T60A/L43A, T60A/R54A, and T60A/K56A) further decreased viral entry compared to the single substitutions ( < 20% versus < 50%), suggesting that these residues (L43, R54, K56, and N61) play a role in receptor binding or post-binding steps of viral entry.
Figure 2. Analysis of double mutants. A: GP expression in producer cells and incorporation efficiency onto virions. B: Relative infectivity of the pseudotype viruses
Ebola and Marburg entry interference
We have previously shown that expression of Δmucin Ebola GP in 293T cells by transient transfection can inhibit Ebola GP-mediated viral entry[25]. The choice of Δmucin Ebola GP instead of wt Ebola GP was to avoid the observed cytopathic effect of expressing wt Ebola GP in the cells. This specific inhibition is consistent with the notion that virus envelope protein expressed in target cells interacts with its cognate receptor(s) and interferes with the reinfection by the viruses using the same receptor(s). However, this entry interference assay is not highly efficient (about 60% inhibition) and variable due to unreliable transfection efficiency (60%-80% of cells are transfected, depending on experiments). Here we developed a more efficient interference assay by stably expressing Ebola GP in the target cells using a retrovirus-based transduction. Briefly, to maximize the number of cells expressing the gene of interest, a pilot experiment was carried out to transduce 293T cells by a replication-defective HIV vector carrying GFP as the reporter. As shown in Fig. 3A, the number of GFP positive cells increased as more virions were used in transduction. Approximately 99%-100% cells expressed GFP when 1-3 mL of virions were used. This protocol gives virtually 100% of transduction efficiency compared to the previous transient transfection protocol.
Figure 3. Interference of viral entry by stable expression of Ebola GP. A: Different amounts (0.1~3 mL) of VSV-G pseudotyped pHR'-CMV-GFP viruses were used to challenge 293T cells. Images of GFP fluorescence were taken 48 h post-infection. NC, negative control, uninfected cells. Percentage of GFP positive cells determined by flow cytometry is shown at the upper corner. B: Entry interference to Ebola GP-mediated viral entry. 293T cells were infected by different amounts of VSV-G pseudotyped HIV viruses to express either GFP or Δmucin Ebola GP. Forty-eight hours later cells were challenged again with either VSV-G or Ebola GP pseudotyped viruses carrying luceriferase gene. Cells were lysed to measure luciferase activity 48 h post-challenge. The EGP or VSVG-mediated entry in the EGP or GFP-expressing cells is presented as the percentage of the luciferase activity of the cells pre-challenged by 0.5 mL of the GFP-carrying virions
To develop an interference assay for Ebola GP-mediated viral entry, the Δmucin Ebola GP construct was introduced to 293T cells by transducing the HIV vector carrying Δmucin gene following the aforementioned protocol (Fig. 3B). The transduced target cells were challenged by the HIV-luc virions carrying either VSV-G or Ebola GP. As controls, the same VSV-G or Ebola GP pseudotyped HIV virions were incubated with the target cells expressing GFP (by transduction). As shown in Fig. 3B, expression of Δmucin Ebola GP in target cells displayed some effect on VSV-G mediated viral entry. This effect was likely due to the cytotoxicity by increasing the amount of viruses used in transducing the Δmucin construct into target cells, since the same trend was observed for the cells expressing GFP (by transduction with the HIV-GFP construct) which were then challenged by VSV-G or Ebola GP pseudotyped virions. In contrast, expression of Δmucin Ebola GP in target cells could interfere with Ebola GP-mediated viral entry up to 95%. Thus we have developed a more reliable and efficient interference assay for Ebola GP-mediated viral entry.
This improved entry interference assay was used to examine 12 substitution mutants of interest identified in this study and our previous work. These mutant GP genes were introduced to the target cells (293T) by transduction, and all of them were expressed at similar levels (Fig. 4A). The cells expressing these different mutants were challenged by either wt Ebola GP or VSV-G pseudotyped virus. In this experiment, the target cells expressing puromycin-resistant gene (puro) or GFP gene were used as controls. Entry of pseudoty-ped virus into puro and GFP-expressing cells was expressed as % wt. As expected, expression of wt or mutant Ebola GP proteins did not greatly affect VSV-G-mediated viral entry (Fig. 4B). In contrast, entry of Ebola GP pseudovirions was decreased dramatically in cells expressing either Dmucin or GP mutants (Fig. 4B). Expression of Dmucin GP reduced Ebola GP-mediated viral entry to 7% of controls. Interestingly, two mutant GP proteins were less efficient in inhibiting entry of wt Ebola GP pseudotyped virus: R64A (31%) and K95A (18%). These results suggest that R64 and K95 of Ebola GP1 are directly involved in receptor binding.
Figure 4. Interference of Ebola and Marburg GP-mediated viral entry by Ebola GP expression. A: Expression of wild type (WT) or mutant Δmucin Ebola GPs in cells. B: Entry interference of VSV-G or Ebola GP-mediated viral entry. C: Entry interference of Marburg GP-mediated viral entry. VSV-G pseudotyped HIV viruses (0.5mL) were used to transduce 293T cells to express WT or mutant Δmucin Ebola GPs, GFP, or puromycin-resistant protein (Puro) as described in Materials and Methods. These pre-transduced cells expressing different proteins were challenged again with VSV-G, or Ebola GP (B), or Marburg GP (C) pseudotyped viruses carrying luciferase gene. Entry of these viruses was measured as the luciferase activity 48 h later. Average of viral entry into cells expressing GFP and cells expressing puromycin is expressed as 100%, and other viral entries were expressed as percentages of that average. Bars represent standard deviations of three repeats
The cells expressing Dmucin or mutant Ebola GP proteins were also challenged by Marburg GP pseudotyped virus. Expression of Δmucin Ebola GP inhibited Marburg GP mediated viral entry to 10% of controls (Fig. 4C). Furthermore, the overall inhibitory profile of viral entry mediated by Marburg GP was highly similar to that by Ebola GP. Two mutants (R64A and K95A), which were impaired in inhibiting viral entry mediated by Ebola GP, were also less effective in inhibiting viral entry mediated by Marburg GP. These results substantiate the notion that Ebola and Marburg viruses share a common receptor/co-receptor for viral entry. These results further strengthen the argument that R64 and K95 are involved in direct interaction with the cellular receptor.
Mutants R64A and K95A are impaired in cell surface binding
To further characterize the role of R64 and K95 in receptor binding, we performed a binding assay of the recombinant Ebola GP proteins following a published protocol[17]. We constructed and expressed the wt and mutant Ebola GP RBD-hIgG fusion constructs (Fig. 5A). The purified proteins were used to bind the target cells (293T). As expected, the control protein S1-hIgG displayed little binding to 293T cells, as determined by flow cytometry (Fig. 5B). In contrast, wt Ebola RBD-hIgG bound to the target cells (Fig. 5B), in a dose-dependent manner (data not shown), consistent with the previous finding[17]. Mutant I170A bound to the target cells at a level similar to that of wt. However, mutants R64A and K95A bound to the target cells less efficiently compared to wt. These results provided direct evidence that R64 and K95 are involved in receptor binding. Since mutant I170A is similarly defective in mediating viral entry compared to R64A and K95A, but not defective in receptor binding, we conclude that I170 is involved in the post-binding steps during Ebola entry.
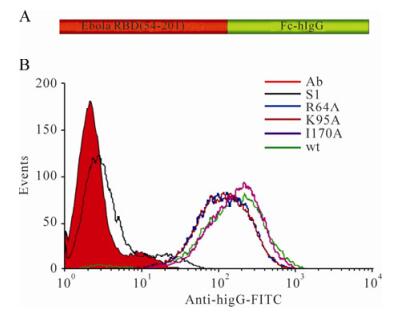
Figure 5. Cell surface binding of purified RBDs (A) Schematic representation of Ebola RBD-hIgG. (B) Binding of hIgG fusion proteins to 293T cells. The SARS virus glycoprotein S1 was used as a negative control for the binding assay. The purified proteins (final concentration of 40 μg/mL) were used to bind 293T cells. Relative binding efficiencies were measured using an anti-human IgG conjugated to FITC by flow cytometry
-
In this study, we focused on identification and analysis of critical residues of Ebola GP1 in receptor binding and post-binding steps. Previously we identified that residues 33 to 185 of the N-terminus consist of the receptor-binding domain, confirmed by others using a biochemical approach[17]. In this report, 41 residues in receptor-binding domain of Ebola GP were studied for their role in protein expression/ folding and viral entry by alanine scanning muta-genesis. Our results showed that only 8 residues are critical for protein structure and viral entry function while alanine substitution of other 33 residues did not have major effect. Four residues (V52, V66, F160, and V181) appear to be important for protein expression/folding, and four residues (T42, T60, N61, and V79) are critical for viral entry. Furthermore, the role of several GP residues which displayed moderate effects by single amino acid substitution was investigated by double substitutions. It was found that four residues (L43, R54, K56, and N61) were important for viral entry. Using an improved viral entry interference assay and cell surface binding assay, we demonstrated that two residues (R64 and K95) are directly involved in receptor binding. In contrast, I170 is not defective in receptor-binding, suggesting a role in the post-binding step during viral entry. Finally, our results further strengthened the notion that Ebola and Marburg viruses use a common cellular molecule for viral entry.
The role of Ebola GP residues in protein expression and viral entry was also studied by others. It has been shown that Y162A caused a defect in protein expression, virion incorporation, and entry[26]. Although it was generated on a DMucin GP (deletion of 315-504), the mutation displayed a similar phenotype to wt GP studied by us. Our previous study showed that F88 is important for viral entry[24]. Consistent with our results, it has been shown that F88A rendered the infectious Ebola virus non-infectious[26]. Recently, another group studied Ebola GP function extensively using DMucin GP (deletion of aa 309-489) pseudotyped FIV viruses[3]. Ten mutations (T83A, S90A, V97A, N98A, T144A, E156A, F160A, T168A, V169A, and T174A) displayed similar phenotype to this work in virion incorporation and viral entry.
Study of Marburg GP-mediated viral entry showed similar results to our findings. K79, the conserved residue in Marburg GP corresponding to K95 of Ebola GP, has also been shown to be important for viral entry[25]. For the other receptor-binding residue (R64) and the putative receptor-binding residues of Ebola GP (L57, L63 and F88), the corresponding residues in Marburg GP are H48, L41, V47, and F72, respectively. Alanine substitution of these residues in Marburg GP caused defects in protein expression, and/or virion incorporation[25]. Therefore it is more difficult to distinguish their roles in protein folding, receptor-binding, or post-binding steps.
Although a lot of host cell surface molecules have been implicated in Ebola entry, the receptor is not identified yet. Therefore we have used an entry interference assay to study the role of Ebola GP residues in receptor binding. Entry interference is one of the classical methods to determine whether different viruses use the same receptor. In infected cells, glycoprotein expressed by the virus can interact with its cognate receptor(s) to inhibit reinfection by the same virus or viruses using the same receptor. Previously using entry interference, it was shown that expression of Dmucin Ebola GP blocked Ebola GP-mediated viral entry[25]. However the assay is less effective (with only 60% inhibition of viral entry) due to low efficiency and high variation of transient transfection. In this report, we improved the effectiveness ( > 90% inhibition of viral entry) and consistency using a retroviral vector to express GP protein in cells. It was shown that two alanine substitutions on Dmucin Ebola GP (R64A and K95A) were less efficient in inhibiting Ebola GP-mediated viral entry (31% and 18%) compared to wt (7%). This is consistent with the cell surface binding data. In contrast, alanine substitution of the other 10 residues of interest (L43, R54, K56, L57, T60, N61, L63, V79, F88, and I170) inhibited entry of Ebola GP pseudoviruses as efficient as wt. These 10 residues are critical for viral entry, but not important for entry interference, suggesting some of them might be involved in the post-binding steps of viral entry. In addition, I170A is defective in mediating viral entry, but not impaired in receptor binding or entry interference, thus we conclude that I170 is important in the post-binding steps in viral entry.
Several pieces of evidence suggest that Ebola and Marburg viruses share a common cellular factor(s) for viral entry. First, conservation of protein sequence suggests that RBD of Ebola and Marburg GP might adopt a similar structure: 68% (32 of 47) of the residues that are important for protein structure or viral entry are conserved in both. Second, it has been shown by an entry interference assay that expression of Ebola or Marburg GP inhibited entry of each other. In this report, expression of mutant Ebola GP proteins displayed the same profile in Marburg entry interference as in Ebola entry interference. In particular, the same two mutations, R64A and K95A, were defective in inhibiting both Ebola and Marburg GP entry (Fig. 4 B and C). Third, purified fusion proteins containing the Ebola RBD can block Marburg GP-mediated viral entry and vice versa[17]. In addition, small molecule compounds and their derivatives inhibited both Ebola and Marburg GP mediated viral entry while they displayed no effect on the VSV-G mediated entry [2, 51]. Taken together, these results strongly imply that the Marburg RBD adopts a similar structure as the Ebola RBD, and a common cellular factor is used by both Ebola and Marburg viruses for infection.
The results in this report and our previous study are summarized in Fig. 6. Twelve residues (green) are important for viral entry. Among them, R64 and K95 are shown to be directly involved in receptor binding, while I170 plays a critical role in post-binding steps. The roles of the remaining 9 residues in viral entry are not very clear at present, for example, they may be involved in either receptor-binding or the post-binding steps. Nevertheless, the results from the current study and the previous work by us and others, together with the x-ray structure of Ebola GP [18], give insights to the structure and function of Ebola GP RBD.
Figure 6. Structural and receptor-interacting residues in RBD of Ebola GP. A: Summary of functional analysis of individual residues of Ebola GP1 represented in different colors. Red, residues involved in protein structure; green, putative receptor binding residues; grey, no detectable defect in protein structure and function. The putative receptor-binding pocket is highlighted by a line on the top. B: Domain organization of Ebola GP. SP-signal peptide, RBD-receptor-binding domain, MUC-mucin-like domain, FP-fusion peptide, N-H-N-terminal helix, C-H-C-terminal helix, and TM-transmembrane domain
Acknowledgements
The laboratory research was supported by National Institutes of Health grant AI 059570 and AI077767. J.W was partially supported by a University of Illinois at Chicago Fellowship













 DownLoad:
DownLoad: