-
Dear Editor,
Coronavirus disease 2019 (COVID-19) is a viral infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). From March 2020, several studies indicate that many subjects affected by mild-to-moderate COVID-19 presented olfactory/gustatory dysfunction (OD/GD) that appeared strongly correlated between them but not with the other symptoms suggestive of upper airway infection (Lechien et al. 2020a, b ; Hopkins et al. 2020; Paderno et al. 2020).
Isolated OD and GD, without any other general or otolaryngologycal complaints like rhinorrhea or nasal obstruction, were also described as the main or unique symptoms of the infection in a variable percentage of cases; these patients, not initially identified as infected, could have represented a potential way to rapidly spread the infection among the population (Lechien et al. 2020b; Vaira et al. 2020).
Post-viral gustatory dysfunction is well established, has been shown to be a key symptom of the coronavirus diseases 2019 (COVID-19), with more than 50% European and U.S mild to moderate patients reporting some degree of loss of taste (Hopkins et al. 2020; Lechien et al. 2020b; Yan et al. 2020). We have apparently overcome the worst part of the initial outbreak. However, persistent GD appears to be commonplace and will drive the demand for general practitioner, otolaryngology or neurology consultation in the next months—evidence regarding recovery will be essential in counselling our patients.
In order to evaluate patterns of gustatory recovery, data from patients with confirmed mild COVID-19 were collected prospectively from four university hospitals. Inclusion and exclusion criteria were described in Fig. 1. All patients had at least 30-days of follow-up after their last negative subsequent COVID-19 test. Information was collected using an online questionnaire created with Professional Survey Monkey (San Mateo, California, USA). Informed consent was obtained. Data may be available upon a reasonable request and an approval from the originating university hospitals.
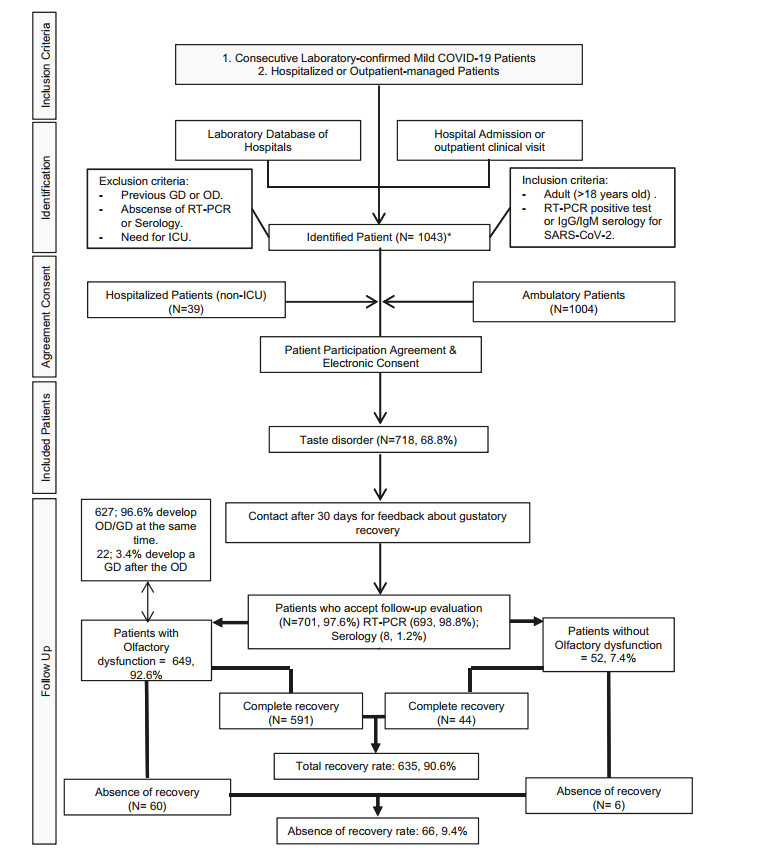
Figure 1. Gustatory dysfunction flowchart. * Those patients with symptom duration < 14 days were tested with a nasopharyngeal swab; in the case of three negative RT-PCR or patients with symptoms for ≥ 14 days, serology testing was performed.
Relevant epidemiological and clinical features contained within the questionnaire were collected by the COVID-19 Study Group of Young Otolaryngologists of the International Federation of Oto-rhino-laryngological Societies (YO-IFOS), and consisted of four subsets (demographic data, medical background, ENT symptoms and olfactory and gustatory dysfunction). All patients completed the Short version of Questionnaire of Olfactory Disorders-Negative Statements (sQOD-NS) (Mattos et al. 2019). The remaining olfactory and taste questions were based on the smell and taste component of the National Health and Nutrition Examination Survey (Bhattacharyya and Kepnes 2015). Physical examination (rhinoscopy, nasal endoscopy or objective olfactory or gustatory testing) was not performed in this study due to the risk of nosocomial infection.
Statistical Package for the Social Sciences for Windows (SPSS version 21, 0; IBM Corp, Armonk, NY, USA) was used to perform the statistical analyses. The potential associations between epidemiological, clinical and gustatory outcomes have been assessed through cross-tab generation between two variables (binary or categorical variables) and Chi square test. Incomplete responses were excluded from analysis. A level of P < 0.05 was used to determine statistical significance. A multivariate analysis (MA) was performed to address possible confounders. Part of this data was previously published in other papers (Lechien et al. 2020a, b, c ; Chiesa-Estomba et al. 2020).
All told, 1043 patients identified in the emergency room or primary care consultation were invited and agreed to participate in the study. Among them 718 (68.8%) described a GD. 701 patients completed the study (Fig. 1). The mean age of patients was 40 ± 13 (range: 18-78). There were 471 females and 230 males. Patients were grouped according the presence or absence of olfactory dysfunction. Both groups were comparable according to age, sex ratio, comorbidities and addiction (P = 0.273, Wilcoxon). Of those patients who reported GD, there were 649 patients (92.6%) who subjectively report a partial or total loss of smell. By contrast, 52 (7.4%) did not report OD. About general symptoms, headache, cough and fever were the most common in those patients who develop a GD. After a mean follow-up of 63 ± 9 days (range: 60-76) from the first consultation, 66 (9.4%) of patients still reported a persistent subjective GD and 635 (90.6%) reported complete recovery. There was a statistical correlation between GD and OD (P = 0.001). However, no difference in the rate of gustatory recovery according to the presence or absence of olfactory dysfunction was encountered (P = 0.952). The mean duration of the GD was 11 ± 5 days (range: 3-36) in those patients who recovered (Fig. 1). There was no significant association between comorbidities and the development or persistence of GD. Other possible confounders rather than OD were identified according to our MA (Table 1).
Characteristics All patients = 701 % P M.A. Median age (Years) 40 ± 13 (range: 18–78). Sex 0.001 0.003 Male 230 32.8 Female 471 67.2 Ethnicity 0.791 0.346 European 558 79.6 Latin-American 136 19.4 North American 2 0.3 North-African 2 0.3 Sub-Saharan African 2 0.3 Current Smoker 83 11.8 0.334 0.291 History of seasonal allergy 121 17.2 0.276 0.301 Comorbidities Diabetes 18 2.6 0.271 0.683 Heart problems 15 2.1 0.555 0.511 COPD 6 0.9 0.767 0.871 Hypertension 47 6.7 0.654 0.391 Asthma 41 5.8 0.433 0.213 Hypothyroidism 44 6.3 0.267 0.379 Autoimmune disease (LES, RA) 27 3.9 0.301 0.411 General symptoms Headache 539 72.6 0.001 0.009 Myalgia 494 66.6 0.411 0.395 Cough 373 51 0.062 0.008 Loss of appetite 418 56.6 0.598 0.541 Dyspnea 39 5.8 0.911 0.934 Diarrhea, Abdominal pain 281 39.3 0.466 0.093 Fever (> 38C) 261 36.2 0.051 0.035 Arthralgia 359 49.5 0.467 0.402 Nausea, vomiting 140 19.7 0.541 0.387 Sticky mucus/phlegm 121 17.3 0.219 0.314 Ear, nose and throat Symptoms Nasal obstruction 108 15.4 0.413 0.367 Olfactory dysfunction 649 92.6 0.001 0.001 Sore throat 189 26.2 0.404 0.635 Rhinorrhea 79 12.5 0.433 0.391 Postnasal drip 90 13.4 0.212 0.566 Face pain/heaviness 103 16.6 0.133 0.418 Ear pain 30 4.1 0.871 0.793 Dysphagia 64 9.2 0.398 0.501 Abbreviations: MA: multivariate analysis; SEL: systemic lupus; RA: Rheumatoid arthritis. Table 1. Demographic and clinical data.
Taste sense correspond to an integrative experience that involves the correlation of information from oral cavity mucosal surfaces through numerous peripheral cranial nerves and other sensory modalities, such as olfaction and somatosensation (Daly et al. 2012). The prevalence of self-reported gustatory dysfunction in our study was similar to those previously reported in COVID-19 patients (Lechien et al. 2020a, b ; Hopkins et al. 2020; Paderno et al. 2020; Vaira et al. 2020). However, the frequency of residual GD after 60 days of follow-up was significantly low. According to our results and similarly to previous report, GD is related to OD. Despite the rate of recovery was higher in those patients without OD, we were not able to find statistical differences among groups. According to our findings, we consider highlighting the presence of isolated GD in at least 7% of our patients.
At this moment, the molecular mechanisms of GD in COVID-19 patients remain still unclear. Regarding taste, it depends on the activity of specialized epithelial cells and taste cells, which are located mainly in the tongue mucosa. Shigemura et al. recently found that renin-angiotensin system (RAS) components as well as angiotensin-converting enzyme- 2 (ACE-2) are expressed in mouse taste organs and are present in the taste buds of fungiform and circumvallate papillae with ENaC (epithelial sodium channel -subunit, a salt taste receptor) or T1R3 (taste receptor type 1 member 3, a sweet taste receptor component). These preliminary results indicate the existence of a local RAS in the taste organ and suggest that taste function may be regulated by both locally-produced and circulating angiotensin II (Shigemura et al. 2019).
It is well known that SARS-CoV-2 binds directly to the ACE2 cell receptors to infect humans and it has been reported that ACE2 is the main host cell receptor of SARS-CoV-2, playing a crucial role in the entry of virus inside the cell to cause the final infection (Hoffmann et al. 2020). ACE2 receptors were also identified in brain and have been detected over glial cells and neurons (Netland et al. 2008). However, more evidence is necessary to elucidate the real mechanism for the GD.
Limitations of this study are the exclusion of patients with severe disease, the small proportion of older patients, the higher proportion of female respondents, loss to follow-up and recruitment from ENT-Clinics, potentially introducing a selection bias. Lack of objective testing to confirm GD is also a limitation. Also, the fact that retro-olfaction is essential for taste. However, at this relatively early point in the pandemic, subjective patterns of recovery of GD in COVID-19 patients are valuable for our patients, hypothesis generation and treatment development.
HTML
-
The authors declare that they have no conflict of interest.
-
Four ethics committees approved the current study protocol (HAP2020-011; CHUSP2003 2020; EpiCURA-2020-2303; CHU-Charleroi:B3252 2020).














 DownLoad:
DownLoad: