HTML
-
The great disruption of viral infection, such as currently circulating novel coronavirus to the health of human beings, African swine fever virus (ASFV) to the benign development of swine industry, is being witnessed, thereby appealing for more attention to the virus induced diseases. Since the first document of pseudorabies virus (PRV) in Hungary in 1902 (Lee and Wilson 1979), this pathogen has been regarded as one of the most significant causative agents, leading to fatal losses to pig industry worldwide (Xia et al. 2018). PRV, also called suid herpesvirus (SuHV-1) or Aujeszky's disease virus (ADV), is an enveloped and double-stranded linear DNA virus which belongs to the subfamily Alphaherpesvirinae, within the family Herpesviridae (Sun et al. 2016). PRV can infect a wide variety of mammals, including pigs, wild boars (Minamiguchi et al. 2019), bears (Zanin et al. 1997), ruminants (e.g. sheep, goat, and cattle) (Kong et al. 2013; Cheng et al. 2019), carnivores (e.g. minks and foxes) (Jin et al. 2016), and rodents, etc. In particular, the latest researches revealed that human beings might also be another potential host of this pathogen (Ai et al. 2018; Liu Q et al. 2020). Although PRV has a wide range of hosts, pigs (including other wild boars) are the unique natural host and reservoir (Freuling et al. 2017). The clinical symptoms of PRV-infected pigs are characterized by diarrhea, vomiting, nervous system disorders (such as tremor, dyskinesia, and lethargy), with high morbidity and mortality for sucking piglets less than two weeks, dyspnea and slow growth for fattening pigs, reproductive disorder for breeding pigs (Sun et al. 2016).
The first identification of pseudorabies (PR) occurred in cat was reported in 1947 in China (Zhou and Sun 1957), thereafter, PRV infection in diverse species has been extensively documented. Given its significant impact on Chinese swine industry (Sun et al. 2016), China has accordingly made a lot of efforts to investigate the epidemiology and molecular biology, and develop the detection methods and commercialized vaccines for the control and prevention of PRV infection. Though significant progress in this field has been made, the threat posed by PRV does not vanish, mainly due to numerous factors such as huge pig herds with distinct management levels and various breeding conditions, the occurrence of PRV variants, and voluntary policy of vaccination. In the present review, an update understanding of PRV in China, with an emphasis on the diagnosis methods, prevalence, clinical conditions of PR in different hosts, molecular characteristics, and the prevention of this disease, is presented. The potential threat of PRV infection to public health is highlighted, meanwhile, the effective agents against PRV infection is also described.
-
In 1947, the first case of PRV infection in a domestic cat was reported in eastern China (Zhou and Sun 1957), and those in pigs, cattle, goats, and other hosts were subsequently documented. Owing to the lack of accurate detection technology and poor biosafety measures in pig industry, PR had been widely prevalent in most regions of China in the early 1980s. With the frequent communication among pig raising markets and the increasing intensity of pig herds in China, the outbreaks of PRV infection with higher morbidity and mortality had frequently occurred in the following years. Fortunately, the prevalence of PR in China has been largely controlled, mainly via the wide application of the commercial Bartha-K61 vaccine (a live-attenuated vaccine based on the PRV Bartha-K61 strain) which was introduced from Hungary. However, it seems that the battle to control PR has not came to the end, since this infectious disease currently remains the major disaster influencing the development of the swine industry in China (An et al. 2013; Sun et al. 2016).
Since 2011, the outbreaks of PR have frequently been reported in many pig farms with a fatality rate up to 50% in infected newborn piglets in China, though these pig herds had been immunized with Bartha-K61 vaccine (An et al. 2013; Tong et al. 2015; Wang et al. 2014). Further molecular biological analysis confirmed that the pathogenic agents were PRV variants which displayed distinct genetic variations compared to the classical PRV strains. Moreover, growing evidence indicated that Bartha-K61 vaccine could not confer complete protection for piglets against these new variants (An et al. 2013; Luo et al. 2014; Tong et al. 2015). More recently, this pathogen has received increasing attention since the first case of human endophthalmitis caused by PRV infection was reported in 2017 (Ai et al. 2018).
-
Traditional clinical and pathological conditions based diagnostic approaches fail to accurately diagnose PR. In recent years, numerous diagnostic approaches for PRV detection have been invented and applied. They can be divided into two categories: serological technologies detecting PRV specific antibodies and molecular biology methods probing PRV nucleic acids. Several representative PRV detecting approaches of these two types are listed in Table 1 and briefly introduced as below.
Target Methods Sensitivity and specificity Reference PRV antibody Blocking ELISA: targeting to the gB antibody High sensitivity (80.9%) and specificity (96.4%) compared with the commercial ELISA kit (IDEXX) Sun HF et al. (2018) Indirect ELISA: targeting to the gE antibody Total 89.1% positive coincidence rate compared with the commercial ELISA kit (IDEXX) (allowing DIVA) Zheng et al. (2017) Indirect ELISA: targeting to the gE antibody High 88.0% sensitivity and 91.5% specificity compared with the commercial ELISA kit (IDEXX) (allowing DIVA) Kou et al. (2018) Indirect ELISA: targeting to the gB antibody Highly total positive coincidence rate (97.8%) compared with the commercial ELISA kit. and the lowest detection limit was 1: 128 dilution of the positive serum Liu MM et al. (2019) ICA: targeting to the gE antibody The lowest detection limit was 1:1280 dilution of the positive serum, and 95.3% positive coincidence rate compared with the commercial ELISA kit (IDEXX) (allowing DIVA) Lei and Zhang (2016) DFM: 2 targeting to the gE antibody Total 74.7% positive coincidence rate compared with the conventional PCR, while which was more sensitive than the later one (allowing DIVA) Xu LH et al. (2017) Liquid chip technology: targeting to the gE antibody This method could be applied to detect both PRV and PRRSV antibodies with higher sensitivity than the commercial ELISA kits (allowing DIVA) Xiao et al. (2018) IFAT: targeting to the gE antibody High sensitivity (93.8%) and specificity (91.7%) compared with the commercial ELISA kit (IDEXX) (allowing DIVA) Zhu et al. (2019) PRV antigen LFA: targeting to the gB antigen Detection limit of inactivated PRV antigens were lower than 1 × 106.6 TCID50/0.1 mL with 86.7% positive coincidence rate compared with the conventional PCR Wang TH et al. (2018) PRV DNA NanoPCR: targeting to the gB, gE, and gG genes This test could be used for the differentiation of wild PRV and gene-deleted vaccine strains with higher sensitivity than the conventional PCR (allowing DIVA) Ma et al. (2013) DdPCR: targeting to the gE gene Higher sensitivity (6.1 copies/μL) than qPCR with high specify (96.2%) compared with viral isolation (allowing DIVA) Chen et al. (2017) LAMP: targeting to the gB gene Showed 100 times higher sensitivity than conventional PCR with the detection limit of 1 fg/μL Xu ZL et al. (2017) PCR combined with nucleic acid probe spot hybridization: targeting to the gE gene The sensitivity was 100 times higher than conventional PCR, being 10 pg/μL and 1 ng/ul, respectively (allowing DIVA) Wunaerhan et al. (2017) PCR: targeting to the gE, gB and TK genes This approach could be used to distinguish between PRV wild virus, SA215 and Bartha-K61 vaccine strains with detection limit of 1.8 × 106 copies/μL (allowing DIVA) Jiang et al. (2018) RPA: targeting to the gE and gB genes Both the sensitivities of gE and gB gene were 100 copies/μL, this approach could be used to distinguish PRV wild and attenuated virus with 100% specify compared with qPCR (allowing DIVA) Liu LB et al. (2018) TaqMan qPCR: targeting to the gE and gD genes The detection limits of gE and gB genes were 12.1 and 39.4 copies, respectively, which was more sensitive than qPCR and conventional PCR (allowing DIVA) Lan et al. (2018) QPCR: targeting to the gB gene The detection limit was lower than 1000 copies/μL, showing higher sensitivity than conventional PCR Hua et al. (2019) QPCR: targeting to the gE gene The detection limit was 10 copies/μl with high specificity (100%) compared with conventional PCR and commercial kits Wen et al. (2019) Multiple RT-PCR: targeting to the gE gene The approach could detect the nucleic acids of PRV, PCV2, PPV, PPRSV, JEV, and CSFV with the detection limit pf 10-3 ng/μL for PRV (allowing DIVA) Li et al. (2019) QIAxcel CGE: targeting to the gE gene This method could be applied for the detection of PRV, CSFV, JEV, PCV2, PRRSV, PPV and ASFV, with the detection limit of 4.53 × 103 copies/μL for PRV (allowing DIVA) Wu et al. (2019) Multiple PCR: targeting to the gE gene This method could be applied for the detection of PRV, PCV2 and PPV, with the detection limit of 72 pg/μL for PRV (allowing DIVA) Xin et al. (2019) NGS This approach could be used for detecting undetermined pathogens with high sensitivity Ai et al. (2018) DIVA: Distinction between the infected and vaccinated animals; ICA: Gold immunochromatographic assay; DFM: Direct immunofluorescence method; IFAT: Indirect immunofluorescent antibody detection method; LFA: Lateral flow assay; NanoPCR: Nanoparticle-assisted PCR assay; DdPCR: Droplet digital PCR assay; LAMP: Loop-mediated isothermal amplification; RPA: Recombinase polymerase amplification; QIAxcel CGE: QIAxcel capillary gel electrophoresis; NGS: Next generation sequencing. Table 1. Various diagnostic approaches developed in China.
-
Owing to the extensive application of PRV glycoprotein E (gE)-deleted vaccines in China, a lot of PRV-gE antibody based serological tests have been developed for rapid and effective differentiation of infected and vaccinated animals (DIVA), while gB antibody based serological approaches have been designed for evaluating the immunological levels induced by vaccine immunization. Various serological methods including enzyme-linked immunosorbent assays (ELISA) (Zheng et al. 2017; Kou et al. 2018; Sun HF et al. 2018; Sun et al. 2018b; Liu MM et al. 2019), serum neutralization test (SNT), and direct-immunofluorescence method (DFM) (Xu LH et al. 2017) have been also applied in recent years. However, the most widely applied approach is the ELISA based one, with higher sensitive and more simple properties relative to others. Among these distinct ELISA tests, competitive ELISAs (cELISA) targeting to the gB or gE antibody are the most widely used in China (Hu D et al. 2015; Liu Y et al. 2018; Wu et al. 2018; Xia et al. 2018).
Besides cELISAs, other types of serological approaches have also been developed for reducing cost and increasing sensitivity and specificity (shown in Table 1). Notably, no unitary standard has been established for comparing the features between these various formats of serological approaches. Therefore, the sensitivity and specificity of the newly designed ELISA tests including blocking ELISA (Sun HF et al. 2018), indirect ELISA (Zheng et al. 2017; Kou et al. 2018; Liu MM et al. 2019), indirect fluorescence antibody test (Zhu et al. 2019), are usually compared to these of commercial cELISA kits. Fantastically, some of them, such as immunochromatogragphic assay (Lei and Zhang 2016) and liquid chip technology (Xiao et al. 2018), display higher sensitivity than commercial cELISA kits. In particular, a novel serological approach based on blocking fluorescent lateral flow immunoassay exhibits advantages of less time and high sensitivity in differently diagnosing wild PRV-infected and vaccinated pigs relative to a commercial gE-ELISA kit (Chen H et al. 2020). In addition, one serological test targeting to the inactivated PRV gB antigen presents relatively high positive coincidence rate (86.67%) with the conventional PCR, while it is not universal thus far (Wang JH et al. 2018).
-
To further improve the sensitivity and specificity for PRV detection, increasing molecular biology approaches targeting to the specific sequences of PRV genes (e.g. gE, gC, gD, gB, and gG) have been created, such as polymerase chain reaction (PCR), real-time PCR (RT-PCR), nano PCR, loop-mediated isothermal amplification (LAMP) (Table 1). In general, PCR and RT-PCR approaches are the most widely applied ones for rapidly detecting the existence of PRV or distinguishing between the PRV wild type and gE-gene deleted strains in China (Jiang et al. 2018; Ren et al. 2018). Especially, multiplex PCR assays have also been developed for synchronous detection of PRV and other pathogens (Li et al. 2019). Among other approaches, next generation sequencing (NGS) is the most powerful and supersensitive method for detecting the presence of PRV in humans (Ai et al. 2018; Yang H et al. 2019), while this approach might be not suitable for large-scale clinical detection due to its high cost. Relative to conventional PCR, the features of higher sensitivity and no requirement for expensive facilities of LAMP approach make it more suitable for rapid and on-site PRV detection in the field (Zhang et al. 2010), while the easily contaminated property of it remains to be overcome in the future.
Apart from the approaches described above, direct pathogen detection is another effective method, which also facilitates vaccine design for the prevention of PR greatly. This method mainly consists of viral isolation and subsequent laboratory diagnosis involving serological, molecular biology, and electron microscopy technologies. Viral isolation is regarded as "gold standard" in pathogen diagnosis, and a mass of PRV strains from different hosts have successfully been isolated in China via this way (Liu et al. 2017; Cheng et al. 2019; Lian et al. 2020), whereas this approach is only suitable for professional laboratories.
-
In order to better control this infectious disease, the serological and molecular epidemiology of PRV infection in Chinese pig population have been extensively performed in recent years, while its prevalent status is affected by numerous factors including regions, sample sizes and seasons, etc.
-
A series of sero-prevalence approaches, mainly targeting the gE antibody, have been developed to comprehensively assess wild PRV prevalence in pigs and explore potentially affecting elements in China. To provide a comprehensive and update view on these, we collect a total of 108 representative studies investigating PRV sero-prevalence in pigs across distinct regions covering 29 provinces of China, which are available in Chinese and English databases from 2011 to the present. Thus, 256, 326 serum samples are included and analyzed in this review. Among them, 76, 553 specimens manifested positive for PRV gE antibody, yielding the average 29.87% positive rate of PRV infection in pigs, as supported by two latest reports (Chen C et al. 2020; Ning et al. 2020). The sero-prevalence rate of PR in the three regions (Jiangsu, Henan, and Jiangxi) from 2018 to 2019 was over 35% (1398/3675) (Chen C et al. 2020), while that for 24 provinces from 2016 to 2018 was 34.2% (32083/93912) (Ning et al. 2020). These demonstrate that in spite of the improved feeding and management levels and widely applied effective vaccines, the prevalence of wild PRV in pig population is still severe in recent years, indicating the continuous threat of PR to the pig industry in China.
The sero-prevalence results of wild PRV infection in pigs from different regions of China are summarized and shown in Fig. 1, which shows an over 30% PRV-gE antibody positive rate in 9 provinces/regions (31.03%, 9/29) (red, purple and orange marked). Furthermore, the sero-prevalence in northern China, eastern China, and central and southern China are higher than those in northeastern China, northwestern China, and southwestern China. Several risk factors including feeding patterns, seasons, and categories of pigs, are considerably associated with the occurrence of PR in pig population. For example, the sero-prevalence of PR in free-range or small pig farms is much higher than that in middle or extensive pig farms. Additionally, other factors, like times of vaccine immunization, altitudes of pig farms, also have significant impact (Wu et al. 2018), while sex and variety of pigs exhibit less influence (Wu et al. 2018).
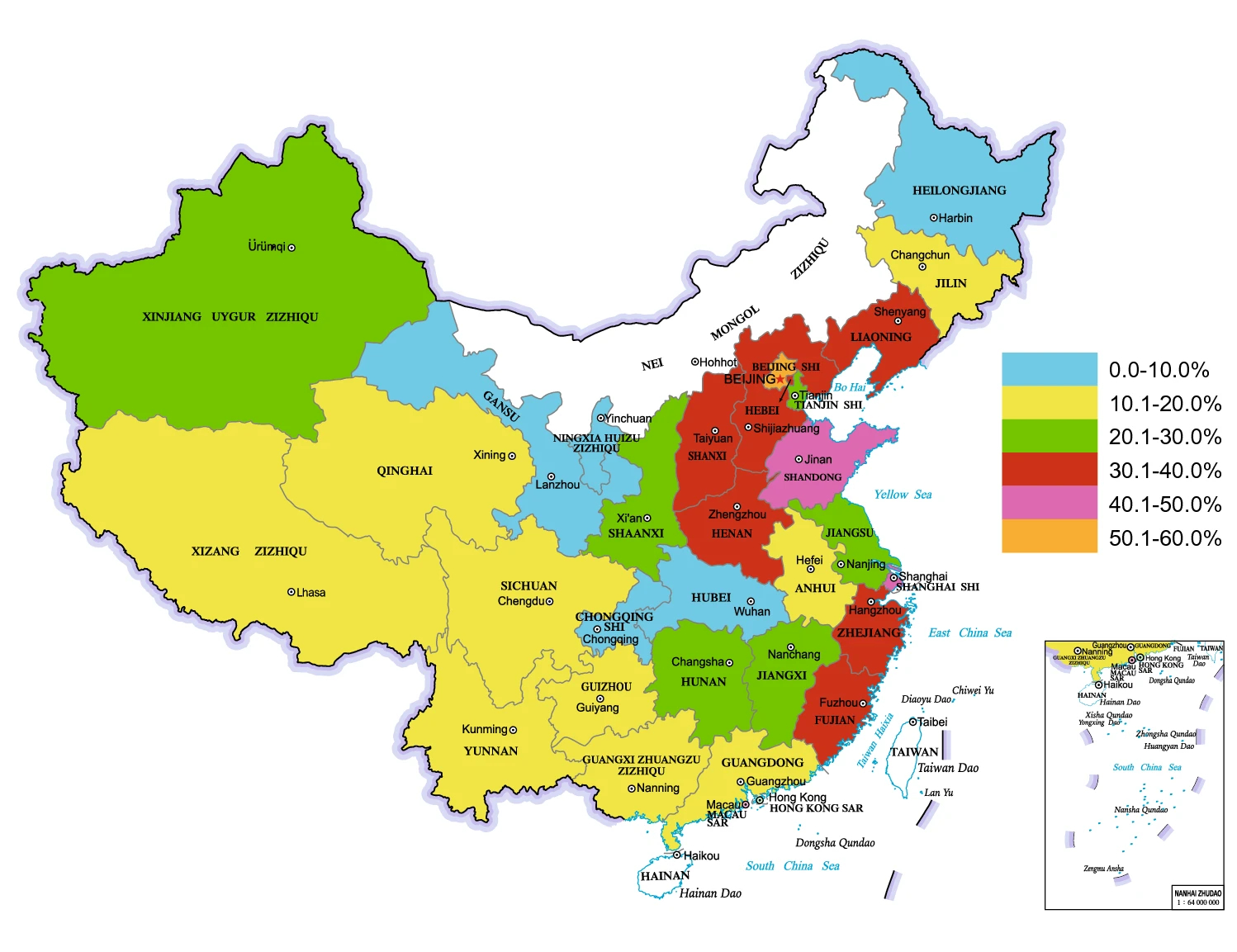
Figure 1. Current sero-prevalence of pseudorabies virus in pigs. Results of 108 representative studies investigating PRV sero-prevalence in pigs across distinct regions covering 29 provinces of China. These data collected from Chinese and English databases from 2011 to the present were analyzed. Data of the regions marked with white were not available.
-
Recently, PCR and RT-PCR methods have been widely employed to investigate the PRV epidemiology of pigs in China (Sun Y et al. 2018; Ma et al. 2020). A total of 27 representative articles available in Chinese or English database from 2011 to the present are chosen and summarized. The analytic results showed that the overall nucleic acid positive rate of PRV in pigs reached 11.5% (4742/41237), as also supported by a recent study, which indicated a nearly 8.0% of PRV-positive rate among 16, 256 tissue specimens collected from 27 provinces in China. Furthermore, the average rate in eastern and central China was over 10.0%, while that of northwestern China was below 7.0%, suggesting that the occurrence of this disease is markedly related to regions (Sun Y et al. 2018). Similarly, another recent study focusing on the PRV prevalence in the years of 2015, 2017, and 2018 in Shandong Province revealed an average positive rate of 11.8% (147/1243) (Ma et al. 2020), as consistent with ours. Notably, the general epidemiological trend of PRV infection is similar to that of the sero-prevalence in different regions (Sun Y et al. 2018). Additionally, pigs showing nervous symptoms or reproductive failures are at high risk of PRV infection, although these consequences may be caused by the infection of various pathogens.
-
In China, a variety of animals (including some wild species) are raised for satisfying the demands of consumers, while some of these animal species are susceptible to PRV. Accordingly, growing reports of PR in different mammals (apart from pigs) have been documented in recent years. For example, the outbreak of PRV infection has caused the death of nearly 8000 minks in Shandong Province, China (Liu et al. 2017). Similarly, a morbidity rate of 80% in foxes has led to the death of 1200 animals due to PRV infection (Jin et al. 2016).
Representative characteristics of PR in non-nature host species are summarized in Table 2. In general, the clinical and pathological signs of these PRV infected species (cow/cattle, dog, wolf, goat/sheep, etc.) have some common features, including pruritus, hyperemia or hemorrhage in some organs (Zhang L et al. 2015; Jin et al. 2016; Liu et al. 2016; Zhang et al. 2016; Sun Y et al. 2018; Cheng et al. 2019; Lian et al. 2020). Additionally, reports of PRV infection with other animals (tigers and wild pigs) have also been documented in China (Li et al. 2014; Qin et al. 2015). Overall, the major causes for PRV infection in plant-eating animals (i.e. cow/cattle and goat/sheep) can be ascribed to living spaces adjacent to pig breeding sites with low feeding management (severe rat plague) (Cheng et al. 2019), while the reasons for carnivorous animals (i.e. wolf, mink, dog, and fox, etc.) may be stemmed from feeding raw or undercooked PRV-infected pork meat or offal to these animals (Liu et al. 2017; Jin et al. 2016; Lian et al. 2020).
Species Case numbers Clinical signs Pathological characteristics Reference Cattle/cow 9 Pruritus, nervous symptoms, being excited and manic, etc Leptomeningeal hyperemia, consolidation of lung lobes, etc Cheng et al. (2019) Dog 12 Pruritus, hypersalivation, broken winded, etc Endocardial and thymic hemorrhage, pulmonary hemorrhage and/or congestion, etc Zhang L et al. (2015) Wolf 1 Pruritus, vomiting, quadriplegia, etc hemorrhagic spots and edema in the meninges Hemorrhagic spots and necrosis in the liver, etc Lian et al. (2020) Goat/sheep 11 Pruritus, nervous symptoms, muscle spasm, etc Leptomeningeal hyperemia, consolidation of lung lobes, etc Zhang et al. (2016) Fox 2 Pruritus, vomiting, broken winded, etc Sugillation in the lung, hemorrhage in the spleen, thymus and liver, etc Jin et al. (2016) Mink 9 Pruritus, diarrhea, muscle spasm, etc Hemorrhage in the thymus and submandibular lymph node, liver and spleen tumefaction, etc Liu et al. (2017) Raccoon 3 Pruritus, vomiting, etc Not mentioned Liu et al. (2016) Table 2. Clinical signs and pathological characteristics of different PRV-infected species in China.
Though PRV infection is fatal to these animal species, studies on the prevalence of this disease remain limited. A report performed by Wang et al. indicated that 38 (5.8%) among 566 dead mink specimens collected in Shandong province displayed PRV-positive (Wang GS et al. 2018). Another study revealed that PRV infection led to 21 of 300 (7.0%) cows suffering from abortion related diseases (Gu et al. 2012). These scant information still hint that these animal industries should pay attention to the occurrence of PRV infection.
-
Since the first identification of PRV in 1902, whether this pathogen can infect humans remains controversial (Wong et al. 2019). Though several reports indicating high possibility of PRV infection in humans with different symptoms (such as fever, weakness and dysphagia) have been documented, the causes of these cases were not determined due to the lack of substantial evidences (Mravak et al. 1987; Anusz et al. 1992; Skinner et al. 2001).
In July 2017, the first case of human endophthalmitis with clinical symptoms of fever, visual impairment and headaches, etc. in China was proved to be PRV positive by NGS combining with PCR analysis (Ai et al. 2018). Further phylogenetic analysis confirmed that it was caused by an emerging PRV variant, suggesting that in spite of the case occurred in China, the potential threat of PRV infection to humans should receive more attention worldwide (Ai et al. 2018). The latest research showed that 1.27% (6/472) patients with clinically suspected central nervous system (CNS) infection were PRV-positive (Fan et al. 2020). Likewise, another retrospective seroepidemiological survey conducted in nine provinces of China indicated that 12.16% (75/617), 14.25% (63/442) and 6.25% (18/276) patients with encephalitis exhibited positive for PRV infection in 2012, 2013, and 2017, respectively (Li et al. 2020). Up to now, increasing cases of PRV infection in humans have been recorded in several provinces of China, and some of patients were eventually blind or dead (Ai et al. 2018; Fan et al. 2018; Zhao et al. 2018; Wang Y et al. 2019; Yang H et al. 2019; Yang X et al. 2019; Fan et al. 2020; Liu Q et al. 2020; Wang et al. 2020). The detailed information of these cases are summarized in Table 3. Notably, almost all patients in these reports were engaged in the works associated with pigs (e.g. slaughterer, veterinarian, and swine herder). Moreover, most of them had been injured when dealing with (sick) pigs or pork. In addition, all infected patients displayed similar clinical symptoms, including fever, headache, respiratory failure, seizure, and even visual impairment or loss, while the occurred clinical symptoms mainly depended on the development stages of the disease. It should be mentioned that most of the cases above were diagnosed as PR via detecting the presence of PRV DNA or specific IgM antibodies. Live viruses (namely hSD-1/2019) have successfully been isolated only from one case (i.e. the cerebrospinal fluid sample) (Liu Q et al. 2020). Further analysis showed its close phylogenetic relationship with those emerging PRV variants prevalent in China. Moreover, the in vivo pathogenicity of this isolated virus in experimental mice or pigs was higher than that of other variants (such as JX2/CHN2015, HuBXY/2018, and HeN1/CHN2012 strains) (Liu Q et al. 2020). However, no experiments investigating the mechanisms of PRV infecting humans have been carried out thus far.
Date Age/Sex Occupation Injured at work Interval after injury Symptoms Diagnostic methods Treatment Reference NA 43/male Veterinarian Yes 4 days Fever, headache, tonic-clonic seizures and coma, etc NGS; PCR and ELISA methods Acyclovir treatment for 2 weeks; antibiotics Yang H et al. (2019) NA 59/male Swineherd Yes 6 days Fever, seizures, tonic-clonic seizures and coma, etc NGS, ELISA method Antimicrobial therapy, penciclovir and foscarnet sodium treatment for 17 days Zheng et al. (2019) NA 44/male Sick pig handler NA NA Fever, seizures, visual loss, etc NGS and PCR methods Acyclovir and other treatments Wang Y et al. (2019) 2011 A young female NA NA Least 5 days Fever, chotic behavior, recurrent seizures, etc NGS (CSF) Acyclovir, foscarnet, VPA treatment, etc. (death) Fan et al. (2020) Nov 2016 42/female Pork cutter/cooker NA 4 days Fever, tonic-clonic seizures, coma, blindness, etc NA Acyclovir and other treatments Zhao et al. (2018) Jun 2017 46/female Swineherd Exposed to the sewage 3 days Fever, visual impairment, headaches, etc NGS, real-time PCR and PCR methods NA Ai et al. (2018) Dec 2017 55/male Pork cutter/cooker NA 4 days Fever, headaches, cough, coma, etc NGS (CSF) NA Zhao et al. (2018) Dec 2017 51/man Pork cutter/cooker NA NA Fever, headache, coma, tonic-clonic seizures and death, etc NGS (CSF) Acyclovir and other treatments Zhao et al. (2018) Nov 2017 38/male Pork cutter/cooker NA 4 days Fever, headache, coma, tonic-clonic seizures, etc NA Acyclovir and other treatments Zhao et al. (2018) 2018 A mid-aged male NA NA NA Fever, bilateral retinal necrosis, consciousness loss, etc NGS (CSF) Acyclovir treatment (death) Fan et al. (2020) 2018 A mid-aged male Pig farmer NA One month Fever, cognitive decline, consciousness loss, etc NGS (CSF) Acyclovir, foscarnet, and methylprednisolone treatment, etc Fan et al. (2020) 2018 A young male NA NA Least 3 days Fever, seizures, consciousness loss, etc NGS (CSF) Continues renal replacement, clonazepam, VPA, acyclovir, etc Fan et al. (2020) Before Mar 2018 59/male Pig farmer Yes 40 days Fever, weakness, tonic-clonic seizures, respiratory failure, etc NGS (CSF) Penciclovir treatment combined with Sodium phosphonate Fan et al. (2018) Feb 2018 50/male Pig slaughterer Yes 5 days Fever, headache, visual disturbance, coma, etc NGS IVIG treatment for 5 days, Glucocorticoids, antiviral and other treatments Yang X et al. (2019) Mar 2018 50/female Pork cutter Yes 7 days Fever, coma, respiratory failure, seizure, etc NGS Yang X et al. (2019) Mar 2018 43/male Sick pig handler Yes 7 days Fever, extremity tremors, respiratory failure, etc NGS Yang X et al. (2019) Apr 2018 59/male Pork cutter Yes 10 days Fever, seizures, respiratory failure, etc NGS Yang X et al. (2019) Apr 2018 50/male Pork cutter NA NA Fever, seizure, respiratory failure, etc NGS Yang X et al. (2019) Apr 2018 25/male Veterinarian Exposed to the PRV infected pigs 6 days Fever, headache, cognitive impairment, seizures, consciousness loss, and meningeal irritation sign NGS (CSF), DdPCR, etc Ganciclovir and foscarnet treatment Liu Q et al. (2020) Aug 2018 43/male Veterinarian Exposed to the diseased pigs 11 days Fever, respiratory failure, headache, cognitive impairment, seizures, consciousness loss, and meningeal irritation sign Virus isolation, NGS (CSF), ddPCR, etc Acyclovir treatment Liu Q et al. (2020) Mar 2019 44/male Pork cutter Yes 14 days Fever, seizure, tonic-clonic seizures and coma, etc NGS and PCR methods Acyclovir, dexamethasone and other treatments Wang et al. (2020) Mar 2019 35/male Pig butcher Exposed to the pigs and pork 7 days Fever, respiratory failure, headache, cognitive impairment, seizures, and consciousness loss NGS (CSF), ddPCR, etc Acyclovir and foscarnet treatment Liu Q et al. (2020) Feb 2019 49/male Pig butcher Exposed to the pork 3 days Fever, respiratory failure, headache, cognitive impairment, seizures, and consciousness loss NGS (CSF), ddPCR, etc Ganciclovir and foscarnet treatment Liu Q et al. (2020) NA: no available; NGS: next generation sequencing; CSF: cerebral spinal fluid; DdPCR: droplet digital PCR. Table 3. Human PRV infection cases in China.
In spite of these, the potential factors contributing to the PRV cross-species transmission events from animals to human are mainly derived from the speculations. The natural recombination events in some PRV variants circulating in Chinese pig population may alter viral virulence and immune response to novel hosts (He et al. 2019; Liu J et al. 2020). In addition, it has been confirmed that multiple alphaherpesviruses including PRV, herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2), and bovine herpesvirus 1 (BHV-1) could utilize nectin-1 as an efficient cellular receptor for cell entry (Geraghty et al. 1998; Campadelli-Fiume et al. 2000; Milne et al. 2001; Li et al. 2017; Menotti et al. 2000; Yue et al. 2020). Moreover, the amino acid residues in nectin-1 among distinct species (e.g. human, swine, sheep, dog, cat, etc.) are relatively conserved (Wong et al. 2019). However, whether nectin-1 plays an important role in the cross-species infection of PRV needs further investigation.
Current Prevalence of Pseudorabies Virus in Pigs
Sero-prevalence of Pseudorabies Virus in Pigs
Epidemiology of Pseudorabies in Pigs
Pseudorabies in Other Animals
PRV Infection as a Potential Threat to Humans
-
Undeniably, PRV remains the major pathogen that posts huge threats to pig industry worldwide. Currently, PRV isolates from the world could be divided into two genotypes, genotype I strains have been widely prevalent in different countries from Europe, America, and parts of China, while most of genotype II PRV isolates are from Asian countries, mainly in China (He et al. 2019a). Since China has suffered from two PR outbreaks, which were caused by PRV classical strains in 1990s and variant PRV strains in 2011 respectively, PRV isolates circulated in China represent emerging variant strains and classical local strains (Sun et al. 2016). Due to the high genetic variations of PRV gC, gB and gD gene sequences, they are often employed to analyze the genetic evolution of PRV strains (Ye C et al. 2015; Zhai et al. 2019). To investigate genetic characteristics of Chinese PRV strains from various species, we construct neighbor-joining phylogenetic trees based on the partial gC, gB, and gD gene sequences of the representative PRV strains isolated after 1970s from China and other countries. The results indicate that all PRV strains from different species including humans can be clustered into two major genotypes (Fig. 2). As shown in Fig. 2A, a tiny minority of PRV Chinese isolates and isolates from Europe and USA, including Bartha vaccine strain from Hungary (accession number: KF779466), are the members of genotype I group. While the genotype II group is composed of other Chinese strains and Japan strains, which can be further subdivided into two groups, namely classical and variant PRV strains, the former group mainly includes vaccine strains Ea (accession number: KX423960) and Fa (accession number: KM189913) which are widely used in China, and other PRV strains, the latter group is composed of a recently licensed PRV vaccine (accession number: KP72 2022) and other strains isolated after 2011. In line with our findings, numerous studies have shown that PRV strains in genotype II but not genotype I were more prevalent in China (He et al. 2019a; Zhai et al. 2019). This finding partially explains why currently available vaccines derived from Bartha or Bartha-K61 strains (genotype I) cannot provide complete protection against PRV variants (genotype II). Furthermore, PRV strains from other species including fox, mink, sheep, dog, and raccoon are randomly distributed with these from pigs in China in genotype I or II, together with the latest research conducted by Liu J et al. (2020), suggesting that PRV strains from the above species including humans may have the similar origin with those of pigs (Moreno et al. 2015; He et al. 2019a).
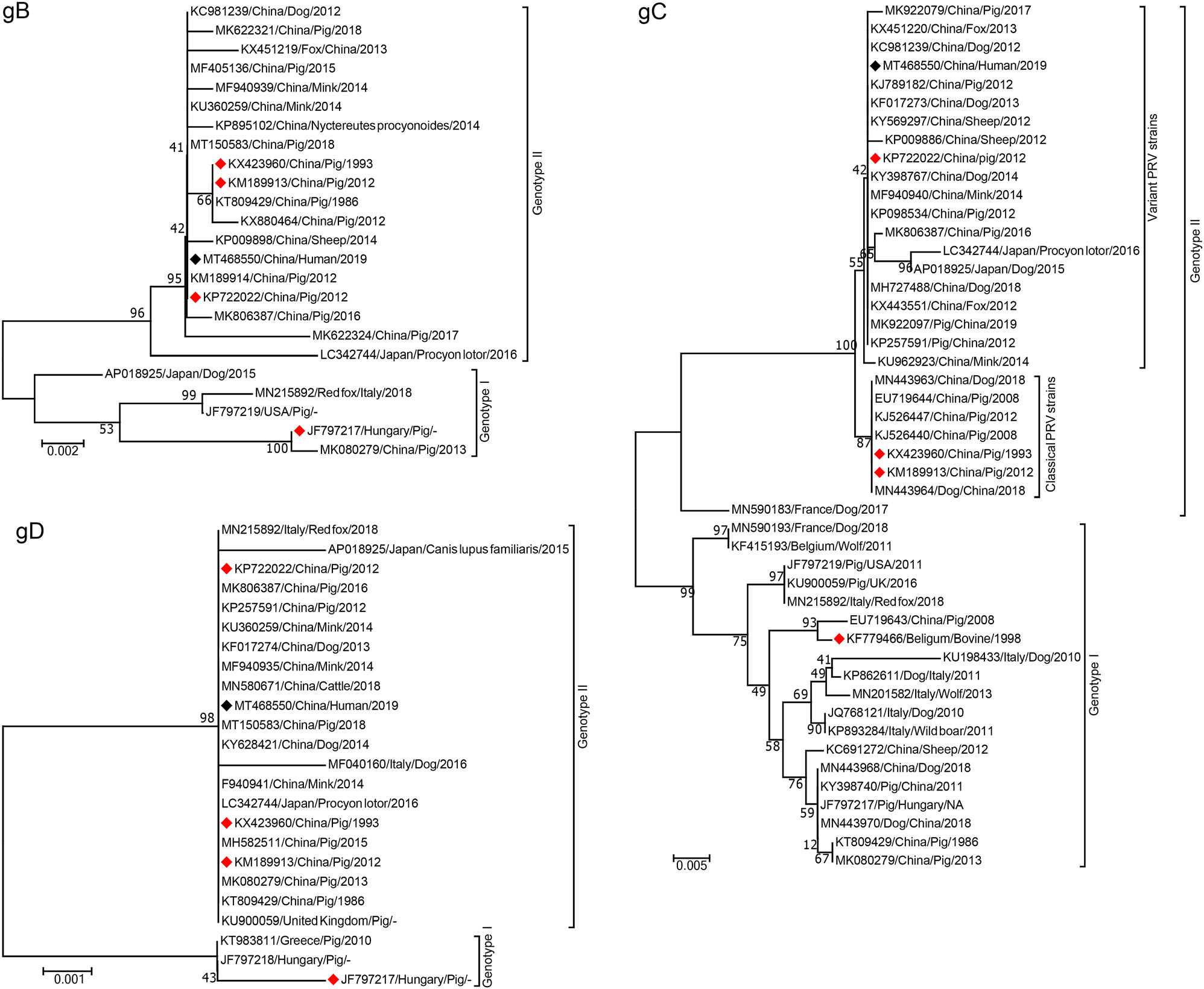
Figure 2. Phylogenetic trees based on the different gene sequences, showing the genetic relationships among PRV strains from different hosts/regions, were constructed by the neighbor-joining method with a bootstrap test of 1000 replicates using MEGA 7.0 software. The hosts, countries, years, and GenBank accession numbers of the reference strains employed in this phylogenetic tree are labeled, Black and red diamonds represent isolates from human and vaccines generated.
-
According to the analysis results of He et al. an emerging PRV variant (Qihe547 strain) in China showed that the average amino acid (aa) differences with PRV genotype I strains, classical and variant strains in genotype II are 4.94%, 1.16%, and 0.46%, respectively (He et al. 2019a). Compared with PRV genotype I strains such as Bartha and Becker, currently circulating genotype II PRV strains in China displayed high genetic variations in some hypervariable regions, such as internal and terminal repeat regions (Ye C et al. 2015). Furthermore, unique aa insertions and deletions (indels), and mutations are frequently found in various protein-coding gene sequences of PRV strains in genotype II. Thus, there are many unique genetic features to differentiate PRV genotype I and II strains. For example, both classical (e.g. Ea and Fa) and emerging (e.g. JS-2012, HeN1, and hSD/2019) PRV strains in genotype II show a 9-nucleotide deletion (encoding 75VPG77) in UL27 (gB) gene, and a 21-nucleotide insertion (encoding 63AASTPAA69) in UL44 (gC) gene compared with PRV strains in genotype I (Sun et al. 2018b). Also, there are some key features of PRV variants that are different from the classical local strains, such as a 6-nucleotide insertion (encoding 288SP299) in US6 (gD) gene of classical local strains compared to emerging PRV variants (Fig. 3).
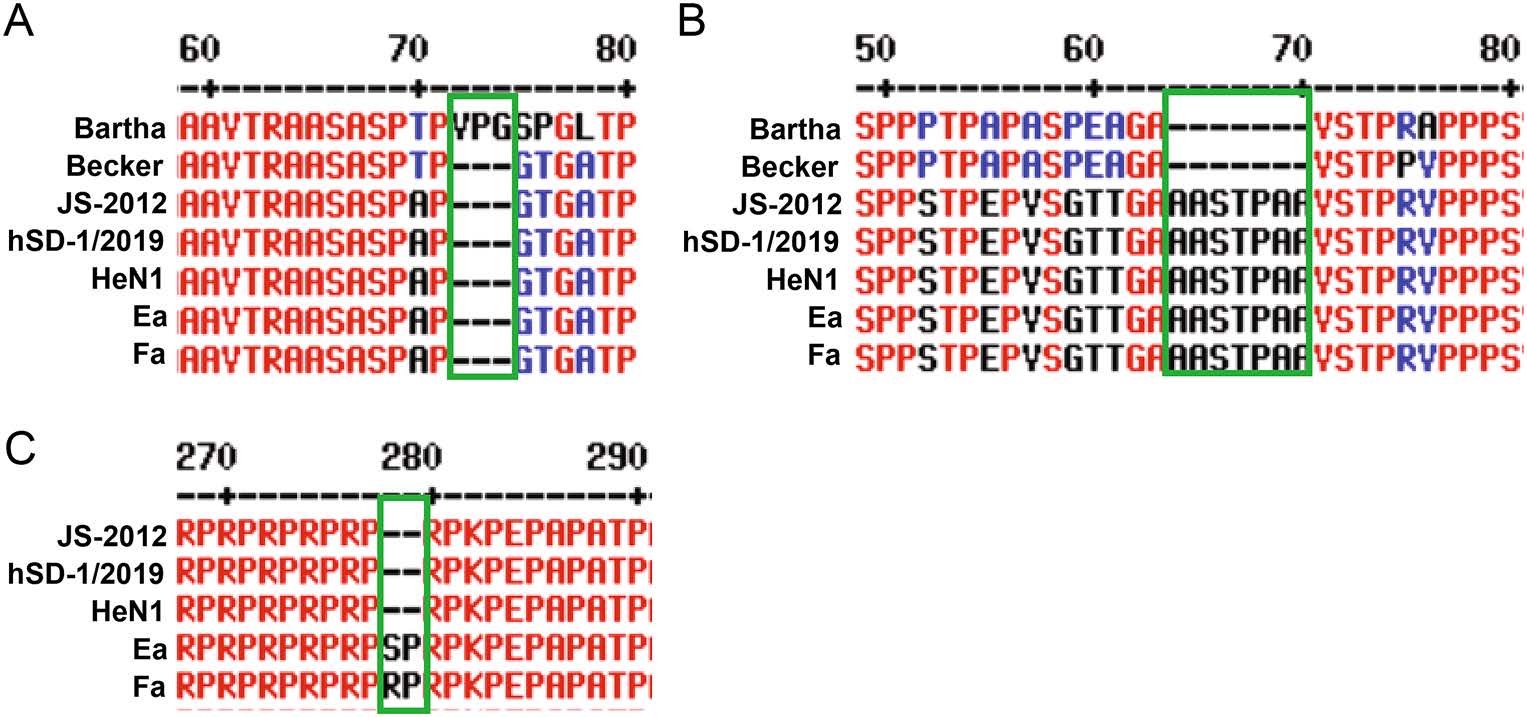
Figure 3. Alignment of partial amino acid sequence of gB (A), gC (B), and gD (C) genes of representative PRV strains (genotype I strains (eg. Bartha and Becker), classical (eg. Fa and Ea) and variant strains (JS-2012, hSD-1/2019, and HeN1) in the genotype II). Unique aa deletions/insertions existed between different PRV strains are shown in the green box.
Besides aa mutations and indels, recombinant events also accelerate the evolution of PRV genome and the change of viral virulence. He et al. has confirmed that inter- and intra-clade recombinant events occurred among various PRV strains using SimPlot software (He et al. 2019a). Recently, two inter-clade recombinant PRV strains (FJ-W2 and FJ-ZXF) have been reported, the gB genes of these two strains belonged to the genogroup I, whereas the gD, gE, and gC genes of which were located in the genogroup II (Zhai et al. 2019). Similarly, the gB gene of another PRV strain FJ62 from Sichuan Province of China had a 100.0% sequence identity with the MY-1 strain isolated from a Japanese wild boar, while its gC, gD, and gE sequences displayed higher homology with Chinese strains relative to others, namely 99.5%, 99.9%, and 99.9%, respectively (Huang et al. 2020). This suggests that the variant strain FJ62 may be generated from the natural recombination of PRV strains from genotype I (from Japan) and genotype II (from China) (Huang et al. 2020).
Genotypical Features of Prevalent PRV Strains in China
Mutation and Recombination of PRV Strains in China
-
To better control and prevent PR in China, the Chinese Government has issued a series of policies with the intent to eradicate PR in pig breeding farms by the end of 2020 (Sun et al. 2016). Accordingly, lots of efforts have been made for the development of effective means to control PRV infection, mainly including vaccines and other novel viral inhibitors.
-
As a major challenge, PRV has in fact been prevalent in pig farms of China for nearly seventy years, vaccination is one of the most effective approaches for preventing it and minimizing the economic losses caused by PR (Freuling et al. 2017). The first live gene-modified vaccine (attenuated Bartha-K61 strain) widely applied in China was imported from Hungary in 1970s, immunization with it has effectively controlled PR in some regions in China (Sun et al. 2016; Zhou et al. 2017). However, outbreaks of PR in farms where pigs had been vaccinated with Bartha-K61 strain based live modified vaccines have frequently occurred in most of regions in China since 2011, which were mainly caused by emerging PRV variants with high sequence divergence comparing with classical PRV strains in China (Ye et al. 2016). These suggest that the existing vaccines derived from Bartha-K61 strain cannot confer complete protection for pigs against the current PRV variants (Hu D et al. 2015; Tong et al. 2015). With the rapid development of biotechnologies and deep understanding of the biological functions of PRV encoding genes, some gene-modified and other types of vaccines derived from local virulent PRV strains have also been generated.
A series of novel gene-modified vaccines based on classical or variant PRV strains have been created in recent years (Summarized in Table 4). It has been confirmed that gE, gI, and TK genes are close to the virulence of PRV strains, while they do not affect viral immunogenicity, therefore these genes are ideal targets for researchers to create genetic engineering vaccines against PR (Cong et al. 2016; Zhao et al. 2020). Before the emergence of variant PRV strains in 2011, some gene-modified vaccines based on classical PRV strains had been licensed in China, including the triple-gene-deleted (gE/gI/TK) vaccine generated based on the PRV Fa strain licensed in 2003, which was regarded as the first genetically modified vaccine against PR in China (Zhu et al. 2004). Likewise, another TK/gG-deleted vaccine developed based on the PRV Ea strain was licensed in 2006 (He et al. 2006). These two licensed vaccines have been widely applied in Chinese pig population against PR until now, however, whether those two vaccines could provide complete protection against variant PRV strains are undefined (Sun et al. 2016). Additionally, a large number of researchers have developed novel genetic engineering vaccines based on emerging PRV variants since 2011, but only two types of vaccines have been licensed so far, including the gE-gene-deleted inactivated vaccine based on the PRV HeN1201 strain in 2019 (Wang T et al. 2015), and another natural four-gene-deleted (gI/gE/Us9/Us2) vaccine based on the PRV C strain in 2017 (Gao et al. 2015). Though other candidates including killed and live attenuated vaccines based on the variant PRV strains have been proved to offer effective protection against PR caused by variant strains, considering the safety issue of these candidates applied in the field, further clinical trials need to be conducted.
Gene-deleted vaccines Strain Features Technologies Main advantages Status References Single gene-deleted vaccine (inactivated) AH02LA (variant) gE-deleted BCA High safe without virulence reversion; Allowing DIVA with more complete protection than Bartha K61 vaccines Not available Wang J et al. (2016) HN1201 (variant) gE-deleted HR Licensed Wang T et al. (2015) Double gene-deleted vaccine (inactivated) ZJ01 (variant) gE/gI-deleted BCA Not available Gu et al. (2015) Single gene-deleted vaccine TJ (variant) gE-deleted HR Safe to piglets without visible gross pathological lesions; Effective immune response; Completely provides protection against emerging PRV variants; Allowing DIVA; Not available Wang et al. (2014) Double gene-deleted vaccine Ea (classical) TK/gG deleted NA Licensed He et al. (2006) JS-2012 (variant) gE/gI-deleted HR Not available Tong et al. (2016) XJ (variant) Yin et al. (2017) AH02LA (variant) TK/gE deleted HR Not available Wang J et al. (2018) JS-2012 (variant) gE/US2 deleted High-temperature passage Not available Liang et al. (2017) Triple gene-deleted vaccine Fa (classical) gE/gI/TK-deleted HR Safer than double gene-deleted vaccines to piglets and growing pigs; Effective immune response; Completely provides protection against emerging PRV variants; Allowing DIVA; Licensed Zhu et al. (2004) SA215 (classical) ZJ01 (variant) Not available Dong et al. (2017) SMX (variant) Not available Hu RM et al. (2015) HN1201 (variant) Not available Zhang C et al. (2015) HeN1 (variant) gE/gI/TK-deleted CRISPR/Cas9 Not available Tang et al. (2016) NY (variant) Not available Zhao et al. (2020) 201, 715 (variant) gE/gC/TK-deleted CRISPR/Cas9 Not available Lin et al. (2020) Four gene-deleted vaccine C (variant) gI/gE/Us9/Us2-deleted Natural losses Licensed Gao et al. (2015) HR: homologous DNA recombination; DIVA: distinction between the infected and vaccinated animals; BCA: bacterial artificial chromosome; CRISPR/Cas9: clustered regularly interspaced short palindromic repeats/Cas9. Table 4. List of genetic modified vaccines against PRV infection.
Considering the large genome of PRV, the property of tolerating gene modification can also be used as an excellent vector for expressing other foreign protein(s), meanwhile, the infectivity and immunogenicity of PRV itself are not affected (Kimman et al. 1992). Some live attenuated PRV recombinant vaccines which could encode key antigens of other animal pathogens have been created and described in a previous review (Dong et al. 2014). As updated in Table 5, researchers usually selected the gene regions encoding nonessential glycoproteins (eg. gG, gI, gE, and TK) to insert exogenous sequences (Chen et al. 2011; Tong W et al. 2020; Zheng et al. 2020). To date, various exogenous genes encoding key antigens of animal pathogens have been successfully inserted into PRV genome, such as the VP2 gene of Porcine parvovirus (Zheng et al. 2020), the E2 gene of Classical swine fever virus (Wang YM et al. 2015; Lei et al. 2016), as well as the Bp26 gene of Brucella melitensis (Yao et al. 2015). In general, most of PRV recombinant vaccines could provide effective protection against multiple infectious disease including PR, but these candidate vaccines are not available in the markets yet. Fantastically, the latest research showed that using Bacillus subtilis as a recombinant vaccine expressing PRV gC and gD proteins could efficiently induce mucosal immune responses against this disease (Wang J et al. 2019), and the efficient protection provided by a gD-based subunit vaccine against PRV variant infection in pig models was also confirmed recently (Zhang et al. 2020).
Insertion sites in PRV genome Parental PRV strains Insertion genes Function (animal model) References gG gene HB-98 strain Porcine IL6 gene and VP2 gene of PPV Provided partial protection against the virulent PPV and PRV challenges (mice) Zheng et al. (2020) gI gene gE/gI/TK-deleted SA 215 strain VP2 gene of PPV Completely protected pigs against maternal PRV infection and significantly reduced the death rate (1/28) after PPV challenge compared with the control (7/31) (pig) Chen et al. (2011) gG gene HB-98 strain Porcine IL18 gene and Cap gene of PCV2 Protected mice against PRV variants infection and significantly reduced the amount of PCV2 viremia (mice) Zheng et al. (2015) Between gE and gI gene gE/TK-deleted strain prM and E genes of JEV Provided 100% and 80% protection against PRV and JEV infection, respectively (mice) Qian et al. (2015) Between gE and gI gene gE/gI-deleted strain E2 gene of CSFV Provided complete protection against maternal PRV and CSFV infection (pig) Wang YM et al. (2015) gE/gI/TK-deleted TJ strain Lei JL et al. (2016) Between gG and gD gene gE/gI-deleted JS-2012 strain Provided complete protection against maternal PRV and CSFV infection without MDAs (pig) Tong W et al. (2020) Between gG and US9 gene gE/gI/TK-deleted TJ strain Cap gene of PCV2 and E2 gene of CSFV Only protected pigs against PRV infection (pig) Abid et al. (2019) gG gene gG deleted strain SiRNA targeting to the N gene of HP-PRRSV Safe to pigs and efficiently inhibited HP-PRRSV replication in vivo (pig) Cao et al. (2015) Between gE and gI gene gE/TK-deleted strain Bp26 gene of Brucella melitensis Induced good humoral and cell-mediated immune response in mice (mice) Yao et al. (2015) gG gene HB-98 strain SAG1 and MIC genes of Toxoplasma gondii Induced partial protection against a lethal challenge with Toxoplasma gondii strain (mice) Nie et al. (2011) Between gE and gI gene gE/gI/TK-deleted TJ strain P12A and 3C genes of FMDV Significantly increased the survival rate (3/5) after FMDV challenge compared with the control (0/5) (pig) Zhang et al. (2011) PPV: porcine parvovirus; PCV2: porcine circovirus type 2; JEV: Japanese encephalitis virus; CSFV: classical swine fever virus; HP-PRRSV: high pathogenic porcine reproductive and respiratory syndrome virus. Table 5. List of live attenuated recombinant vaccines against PRV infection.
In China, various types of vaccines against PR have been developed recently, mainly including inactivated vaccines, live attenuated vaccines, and live virus-vectored vaccines. Most of vaccines aforementioned could generate high level of neutralizing and gB antibodies, and offer effective protection against PRV challenge. Furthermore, these candidate vaccines also allow DIVA, thereby accelerating the eradication of PR in China. As shown in Table 4, the inactivated vaccines display high safety to the vaccinated animals without viral virulence reversion, while they are generally less efficacious compared to genetically modified-live vaccines (the live attenuated and virus-vectored vaccines) (Freuling et al. 2017). Notably, genetically modified-live vaccines summarized in the Tables 4 and 5, have their own unignorable disadvantages. The important one is their safety to pig populations should be confirmed via lengthy testing (Wang J et al. 2016). Moreover, the safety issues of these PRV modified-live vaccines to non-target species has also attracted widespread concern. For example, vaccination with attenuated Bartha-K61 strain could cause PR in sheep with severe clinical signs (Kong et al. 2013). Similarly, immunization with Bartha-K61 and HB98 (another commercial live attenuated PRV vaccine generated based on Ea strain in China) vaccines also posed potential health threat to dogs (Lin et al. 2019). In addition, the latest report verified that infection with a gE-deleted PRV strain could cause PR in an adult red fox in Italy (Moreno et al. 2020). Finally, as the origin of variant PRV strains circulating since 2011 in China remains undefined, potential contribution of these genetically modified-live vaccine to the evolution of this pathogen should also be considered (Wang T et al. 2015; Bo et al. 2020).
-
Considering the great impact of PR on pig industry, Chinese scientists have also devoted to identifying inhibitory agents against PRV infection. The antiviral activities of Chinese herbal medicines have received comprehensive attention, as summarized in Table 6. For instance, the druggable components derived from leaves of Panax ginseng exhibited effective anti-PRV activity in vitro, while the detailed antiviral mechanisms remain undetermined (Hou et al. 1998). Likewise, resveratrol, as a polyphenolic stilbenoid initially exacted from Veratrum grandiforum and leaves of vines (Pezzuto, 2019), has a variety of bioactivities, such as high anti-oxidative and anti-inflammatory properties (Malaguarnera et al. 2020). In particular, the antiviral activities of resveratrol against PRV infection have well been recognized in a series of observations. Resveratrol showed efficient anti-PRV activities with a low 50% inhibition concentration (EC50) of 17.17 μmol/L in vitro, the underlying mechanisms of it in suppressing PRV replication were at least partially associated with the inhibition of the IκB kinase activation (Zhao et al. 2017). Its anti-PRV and immune-adjuvant abilities were also confirmed in both mice and pig models (Yao et al. 2015; Su et al. 2016; Zhao et al. 2018). Additionally, the ability of resveratrol to inhibit ASFV replication in vitro previously described (Galindo et al. 2011), together with interesting observations as stated above, suggest that this natural herb should warrant intensive investigations in the future, thereby pushing it forward to be an efficient agent for protecting animals or humans from PRV infection.
Source Extracts Mechanism IC50 CC50 In vitro In vivo PRV strain MOI References Resveratrol Ethanol Inhibition of viral replication; Inhibition of IKB kinase activation > 262.87 μmol/L 17.17 ± 0.35 μmol/L √ √ Rong A 0.01 Chen et al. (2019), Zhao et al. (2017) Germacrone Dimethyl sulfoxide Inhibition of viral replication 233.5 μmol/L for Vero, 184.1 μmol/L for PK15 54.51 μmol/L for Vero, 88.78 mol/L for PK15 √ × PRV variant Bartha K61 vaccine 0.1-10 He et al. (2019b) Isatis indigotica (leaf) Ethanol Inhibition of viral replication 226 μg/mL 11 μg/mL √ × TNL 100 pfu/well Hsuan et al. (2009) Radix isatidis Ethanol and water Inhibition of viral replication; Killing virus directly Not mentioned Not mentioned √ × MinA 100 TCID50 Tong C et al. (2020) Marine Bacillus S-12-86 lysozyme Water Inhibition of viral replication 100 μg/mL 0.46 μg/mL √ × Attenuated Not mentioned Zhu et al. (2013) Diammonium glycyrrhizin Not mentioned Killing virus directly 1.25 mg/mL Not mentioned √ √ Bartha K61 vaccine 104 pfu/mL Sui et al. (2010) Vanadium-substituted Heteropolytungstate DMEM Killing virus directly 400 μg/mL 5 μg/mL √ × Bartha 200 TCID50/mL Liu et al. (1998) Graphene Oxide DMEM Killing virus directly Not mentioned Not mentioned √ × HNX variant 0.01 Ye S et al. (2015) Ivermectin Dimethyl sulfoxide Blocking the nuclear translocation of viral DNA polymerase UL42 Not mentioned Not mentioned √ √ Not mentioned 0.01 Lv et al. (2018) Phosphonoformate sodium Not mentioned Inhibition of viral DNA polymerase 480 μg/mL Nearly 60 μg/mL √ × Kaplan 2 Ren et al. (2011) Table 6. Different types of compounds with anti-PRV infection activity.
-
Other compounds with antiviral properties also receive extensive attention, as described in Table 6. For example, Vanadium-substituted Heteropolytungstate should be the first compound exhibiting anti-PRV activity through directly killing the virus (Liu et al. 1998). Similarly, Graphene Oxide has been confirmed its antiviral function against PRV infection (Ye S et al. 2015). Therefore, these agents might hold the promise to be natural disinfectants in the future, if their killing characteristics of multiple viruses prevalent in pig farms are further verified. Additionally, ivermectin, an effectively anti-parasitic drugs, also manifested a nearly 100% inhibitory activity against PRV replication under the concentration of 2.0 or 2.5 μmol/L in vitro. The molecular mechanism of it was ascribed to blocking the nuclear translocation of viral DNA polymerase UL42 (Lv et al. 2018). Moreover, further in vivo study revealed that ivermectin treatment could decrease the death rate of PRV infected mice, and relieve the lesions in mouse brains caused by PRV infection (Lv et al. 2018). However, the anti-PRV activity of ivermectin in pig models warrants further investigations.
-
Small RNAs including small interfering RNAs (siRNAs) and microRNAs (miRNAs) are widely used to explore gene functions due to their features of targeting mRNA degradation (Tan et al. 2019), and also to be useful tools for efficiently inhibiting virus replication and interfering protein synthesis (Du et al. 2009; Wong et al. 2020). For example, application of siRNA targeting to PRV DNA polymerase processivity factor UL42 could efficiently suppress viral replication in cultured cells (Wang YP et al. 2016). Similarly, over-expression of miRNA-21 significantly inhibited PRV replication via directly targeting to interferon-γ inducible protein-10 gene in PK15 cells (Huang et al. 2014). Additionally, another research group proved the capability of PRV-miRNA-LLTTa to reduce the numbers of PRV genomic copies in vitro, while the potential mechanism requires further investigations (Liu H et al. 2019).
-
PRV remains the major infectious pathogen which greatly affects the development of pig industry in China. Chinese Government and scientists have spent nearly seventy years in fighting PR. The past efforts devoted to investigating the prevalence of PR in China, and developing diagnostic approaches for detecting PRV, vaccines and other novel inhibitors against this virus, are well summarized as described above.
In terms of PRV prevalence in China, the literatures collected from English and Chinese databases clearly indicate that this virus has widely spread in different provinces/regions of this country with intense epidemic. The factors contributing to the severe PRV infection in China are complex. Overall, the biosecurity levels in Chinese large-scale pig farms have been greatly improved, and PR in some breeding pig farms has been successfully eradicated via implementing effective elimination strategies. However, pigs in these PR purified farms are still faced with high risks of being re-infected upon introducing PRV infected breeding pigs, even PRV infected semen (Song et al 2017) and animal feeds (Dee et al. 2018) from the outside. Additionally, as a voluntary policy, vaccination for PR eradication in some pig farms (especially free-range and small farms) in remote regions of China is not always performed (Sun et al. 2016), thus, poor biosecurity leads to more severe prevalence of PR in these farms.
Also, severe prevalence of PRV in Chinese pigs could increase the possibility of other susceptible animals to get infected with this pathogen, which in turn increases the opportunities of human infection. Though the cases of human infection with PRV currently are still limited, potential threat of its infection to humans should truly receive widespread concerns. Common characteristics in these cases are summarized in Table 3. In particular, it has been proved that PRV may infect humans via wound (Yang H et al. 2019, Yang X et al. 2019) and eyes (Ai et al. 2018). Additionally, considering these facts that intranasal immunization is the main approach for vaccination and PRV genome is detected in the air specimens from pig farms (Li et al. 2019), whether airborne route is another pathway for PRV infection in humans needs further investigations. It is therefore suggested that self-protection for these susceptible population is very essential upon working. Moreover, molecular analysis revealed the occurrence of PR in other animals and humans might be caused by PRV variants circulating in Chinese pig population (Ai et al. 2018; Chen et al. 2019; Jin et al. 2016; Liu Q et al. 2020), while the understanding of PRV transmission from pigs to other species (especially humans) should further be explored.
In recent years, different types of diagnostic approaches for PRV diagnosis have been created. It should be noted that despite not reaching to commercialization, some diagnostic products designed by veterinary detection institutes have widely been applied for the diagnosis of PR or the investigation of the PRV prevalence in China. Notably, available approaches for determining PRV infection in humans remain very limited. The confirmation of PRV infection in humans by most hospitals mainly relies on NGS, while this approach is time-consuming and relatively expensive compared with others. Furthermore, considering that PRV infection may be fatal to humans, NGS seems not to be suitable for patients with severe infection. In particular, diagnostic standards for detection of PRV infection in humans are not available in Chinese hospitals, including clinical and laboratory tests, which are essential for early diagnosis of this fatally infectious disease in humans. Therefore, these should be the emphases for future efforts.
Additionally, various types of gene-modified vaccine candidates have been generated with efficient protection for pigs against PRV infection. However, they have not been commercial for application mainly due to multiple factors, such as their safety to pig populations and the possibility of viral virulence reversion. Additionally, as different species show various susceptibility and immune response to PRV infection, safer vaccines with efficient protection against PRV infection in other species including goat, dog, mink, also need to be developed in the future. Furthermore, several antiviral drugs treating other herpesvirus such as HSV-1 can be introduced for curing human PRV infection, such as valacyclovir, acyclovir (Wang Y et al. 2019), ribavirin (Pancheva 1991). Meanwhile, some newly developed therapies including monoclonal antibodies, small molecule inhibitors, are also available approaches for the treatment of PR in humans.
In conclusion, though effective vaccines have widely been used in pig population against PRV infection, this pathogen is still prevalent in Chinese pig herds. The degree of PR elimination in pig population seems to rely heavily on whether the mandatory vaccination policy is conducted or not. In addition, PRV infection in other susceptible species including humans should receive more concerns. Some effectively diagnostic and treatment approaches urgently need to be exploited, especially in term of human infection.
-
This work was supported by the General Program of National Natural Science Foundation of China (Grants No. 31571432/31802252), the Hunan Provincial Natural Science Foundation of China (Grant No. 2015JC3097/2020JJ4041), and the Postgraduate Scientific Research Innovation Project of Hunan Province (CX20200659). Support was also provided by "Shennong" Scholar funding to Aibing Wang. Additionally, we sincerely thank Fan Liao and Kuan-kuan Xiong for the assistance in data collection.
-
The authors declare that they have no competing interests.
-
The present manuscript does not include any animal or human experiments by any of the authors.














 DownLoad:
DownLoad: