-
The use of a macaque model to monitor of plasma viral RNA in acquired immune deficiency syndrome (AIDS) has become an essential way to access disease progression and evaluate the effect of prophylactic or therapeutic interventions (15, 17, 19). For these reasons, studies on viral load quantification have increased in recent years. Available technology for quantitative evaluation of HIV-1 viral load includes RT-PCR (10, 12, 18), branched DNA (b-DNA) (18), and nucleic acid sequencebased amplification (NA SBA) (11). These methods are implemented in several standard commercial kits employed for HIV-1 infection monitoring. In simian immunodeficiency virus (SIV) infected macaque models of HIV, the current commercial test for detection of SIV is the branched-chain DNA assay (Bayer, Emeryville, CA) however its drawbacks are it is expensive and its sensitive is limited to about 1 500 viral RNA copies/ mL plasma. Recently, quantitative Real-Time reverse transcription-polymerase chain reaction (RT-PCR) assays mainly based on TaqMan or SYBR green due to its highly sensitive and reliable detection have found wide application in SIV viral loads quantification (2, 3, 16). In SYBR green RT-PCR assay, as the double-stranded PCR product accumulates during cycling, more SYBR green dye binds and emits fluorescence. Thus, the fluorescence intensity increases proportionally with dsDNA concentration (20). The specificity of the PCR can be confirmed through gel analysis and dissociation curve analysis where different PCR products are reflected in the number of first derivative melting peaks (14).
In the current study, we have succeeded in establishing a one-step assay using SYBR green as a fluorescent dye to quantify SIVmac251 and SIV-mac239 RNA purified from virus stocks produced in CEM×174 cells. The detection limit of this assay was 10 copies per reaction or 215 copies/mL of plasma. The accuracy of the assay was further confirmed in a SIVmac251 infected rhesus macaque. This method does not requiring any hydrolysis probes and can be performed conveniently and economically with a sensitivity equivalent to the TaqMan assay (2, 5).
HTML
-
A healthy Chinese rhesus macaque (Macaca mulatta) which was seronegative for SIV, simian retrovirus, and simian T cell leukemia virus type-1 and B virus was housed at the animal lab in Guangzhou Institute of Biomedicine and Health. The monkey was intravenously infected with approximate 30 TCID100 of uncloned pathogenic SIVmac251 (donation courtesy of Dr. Chuan Qin). Whole blood samples were collected from experimental animal by venepuncture in tubes containing EDTA at various time points (1, 2, 3, 4, 8, 12, 16, 20, 24 weeks post inoculation). The plasma was collected by centrifuging whole blood at 2500 r/min for 20 min and stored in aliquots at -80 ℃. SIVmac251 and SIVmac239 (donated by the NIH AIDS and Reference Reagent Program) was propagated and titrated in the CEM×174 cell line (9). The virus culture supernatants were collected and stored at -80 ℃.
-
The viral RNA in cultured SIVmac251 supernatant was purified with QIAamp viral RNA minikit (Qiagen, Valencia, CA) and viral cDNA synthesis was performed according to a standard protocol using random hexamer primers (Takara, Dalian, China). The templates were amplified from viral cDNA with the following specific PCR primers: 5'-CCCGGCGGAA AGAAAAAG-3' and 5'-CGCCTGAAATCCTGGCA CTAC-3'. Products (461bp) were ligated into PMD-20T vector (Takara). The recombinant plasmid PMD-gag461 was transformed into DH5α E.coli Strain and the inserted sequence and direction were verified by sequencing. The recombinant plasmid was purified and linearized with EcoR I (Takara) and then was used in vitro transcription as described (6). RNA transcript (544bp) was purified with RNeasy mini kit (Qiagen) and the optical density (OD) was measured to determine the concentration. The RNA was then 10-fold serially diluted in diethylpyrocarbonate (DEPC)-treated water containing carrier tRNA (transfer RNA from Escherichia coli, 30ug/mL; Sigma, St. Louis, MO). The standard RNA dilutions were immediately frozen in 10 μL aliquots at -80 ℃.
-
To avoid major mismatches due to SIV variability, two specific oligonucleotides that recognize specific and conserved sequence on the gag region were chosen: 5'-AAATACTTTCGGTCTTAGC-3' and 5'-GGGTAATTTCCTCCTCTG-3'. The amplification of this pair of oligonucleotides yielded a 232bp gag fragment.
SYBR green real-time PCR assay was carried out in 50μL PCR mixture volume consisting of 25 μL of 2×Quantitect SYBR green RT-PCR Master Mix (Qiagen), containing HotStarTaq DNA polymerase, 2.5μL of 10 μmol/L of each oligonucleotide primer, 0.5μL of 100×QuantiTect RT Mix (containing Omniscript and Sensiscript reverse transcriptases) and 2μL of RNA extracted from samples or 2μL from ten-folds serial diluted RNA standard (from 5×107 to 5 copies/μL). SIVmac251 gag gene amplification was carried out as follows: reverse transcription at 50 ℃for 30 min; initial activation of HotStar Taq DNA Polymerase at 95 ℃ for 15 min; 45 cycles in four steps: 94 ℃ for 10 s, 56 ℃ for 30 s, 72 ℃ for 30 s. At the end of the amplification cycles, melting temperature analysis was carried out by a slow increase in temperature (0.1 ℃ /s) up to 95 ℃.
-
RNA standard (from 1×106 to 1×101 copies/μL)was amplified as described above and was repeated four times to confirm the repeatability in different runs.
The specificity of PCR was confirmed through dissociation curve analysis where different PCR products are reflected in the number of first derivative melting peaks (14) and confirmed by sequencing.
In addition, the amplification efficiency of PCR is evaluated by using the ten-fold serial diluted RNA from transcripts and infected cell supernatants as templates. The amplification efficiencies of serial dilutions between RNA standard and viral RNA should be approximately equal; in other words the difference between the slopes (Δs) of amplification curves should be smaller than 0.1 for the purpose of reliable quantification. The amplification efficiencies (E) were calculated as 101/-s -1 (6).
Samples preparation
RNA standard preparation
PCR program establishment
Repeatability, specificity and amplification efficiencies analysis
-
The 461bp amplified products from the gag gene of SIVmac251 was inserted into PMD-20T plasmid and linearized with EcoRI as the transcript template. Electrophoresis demonstrated the absence of plasmid DNA contamination in the purified RNA transcripts (544bp) (Fig. 1).
-
SYBR green real-time RT-PCR products from plasma RNA showed 100% homology with SIV-mac251 on sequencing (data not shown). The tenfold diluted RNA standards containing 1×108 copies to 10 copies were amplified to determine the sensitivity of this method. The result (Fig. 2A) indicated that the lower limit of detection was 10 copies per reaction in 50μL PCR system.
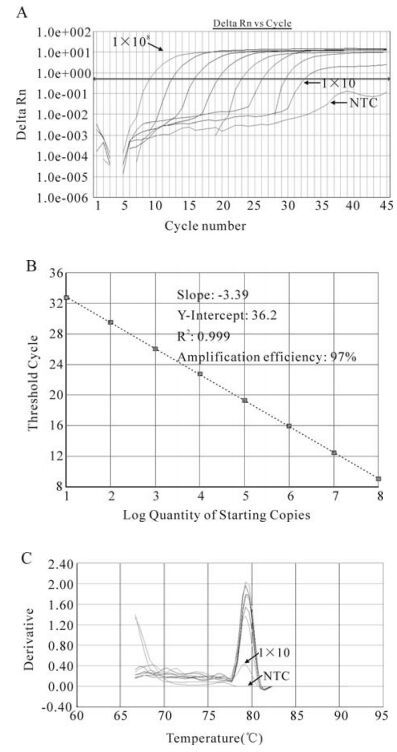
Figure 2. Data analysis of SYBR green Real-time RT-PCR. The threshold limit, R2 value and dissociation curve were determined with the SDSv 1.3.1 software package on the 7300 ABI Prism. A: RT-PCR was conducted with a serial 10-fold dilution of RNA transcript (108-10copies), and with no template controls (NTC). Delta Rn is normalized via the fluorescence of the passive internal dye, ROX. B: Standard curve: Starting quality of target template versus threshold cycle (CT). Linearity was observed over an 8-log magnitude. C: Dissociation curve: Derivative displays a plot of the first derivative of the rate of change in fluorescence as a function of temperature. The specific PCR products with a Tm 79.4 ℃ were determined in this figure, whereas for NTC no special curve was detected.
Standard curve and dissociation graphs were generated by using the SDSv1.3.1 software package (Fig 2B and 2C). The standard curve indicated a high correlation coefficient (R2= 0.999) and amplification efficiency. The dissociation curve plot displaying the single amplification peaks with a Tm of 79.4℃.
The variation within a run was determined by perform each RNA copy number 3 times at one PCR reaction and repeatability of this method was confirmed by four times' separate running with each RNA copy number (Table 1). Even in the lowest template concentration (20 copies per reaction), the coefficient of variation within runs and between runs was 0.15% and 0.39%.

Table 1. Precision of SYBR green Real-Time RT-PCR Approach
Furthermore, the amplification efficiency between different samples and the RNA standard was compared. Samples and RNA standards were serially diluted and amplified in 50μL system. The slopes of three curves (-3.39, -3.35 and -3.34 respectively for RNA standard, SIVmac251 and SIVmac239 viral RNA) were comparable (Δs < 0.1), which demonstrated that the amplification efficiencies of SIVmac251viral RNA, SIVmac239 viral RNA and RNA standard were approximately equal.
-
To evaluate the utility of this method, we analyzed samples from a rhesus macaque which was inoculated with SIVmac251 virus. As shown in Fig. 3, the monkey showed a typical course of primary viremia following intravenous challenge with SIVmac251. The virus was first detected at as early as first week post-challenge of virus and plasma SIV RNA value reached a peak (3.6 ×106 copies/mL) a week later.Subsequently however, viral load declined to approximately 1×104 copies/mL by 8 weeks postchallenge, and keep steady around the level in the chronic phase.
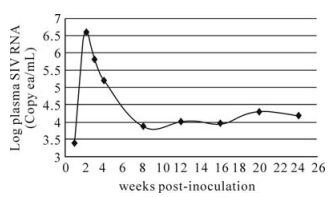
Figure 3. Viral load profile for Rhesus macaque (03047) which was SIV-naÏve at the time of intravenous challenge with SIVmac251. Similar with typical plasma viremia profile for SIV naÏve animals inoculated with SIVmac251, 03047 had a peak of plasma viral RNA value (peak 3.6 ×106 copies/mL) at 2 weeks post-challenge. Subsequently however, viral load declined to 1×104 copies/mL by 8 weeks post-challenge, and keep steady around this level in chronic phase.
The standard SIV RNA
Sensitivity, specificity and amplification efficiency of SYBR green RT-PCR
Effect for the detection of rhesus macaque samples
-
The levels of viral loads in plasma and CD4+ T cell count have been shown to be the most important indicators for evaluating prognosis of SIV infection, effects of vaccine and antiviral drug therapy (1, 4). Since most SIV infections are restricted in laboratory studies, the commercial kits to detect SIV viral load are lacking in their suitability and expensive. Here we have succeeded in establishing a relatively cheap but highly sensitive SYBR green-based RT-PCR method to quantify the SIV viral load in plasma.
In a previous report, the SYBR green Real Time RT-PCR for SIVmac251 and SIVmac239 viral RNA detection had a limit as low as 1 000 copies per analysis (3) and the assay based on TaqMan probe can reach a lower limit of 4 copies per reaction in SIV detection (5). This assay based on SYBR green as detection dye showed a sensitivity comparable to the TaqMan method: the detection limit was as low as 10 copies per reaction and 215 copies/mL of plasma. Moreover, the primers targeted a conserved sequence on the SIVmac251 gag region and so can also exactly match with other SIV isolates including SIVmac239, SIVmac17E-Cl, SIVmac17E-Fr and Simian-Human immunodeficiency virus (SHIV) strain 89.6. We did not perform a test on 'one-copy' sensitivities since this is often not realistic and unreliable since there are limitations inherent in distribution of target sequences in the volume of sample aliquots taken for testing.
Plasma viral load of one Chinese Rhesus was determined by our assay at different times after inoculation of SIVmac251. The dynamics of plasma viral load was highly consistent with previous published results quantified by commercial branched DNA amplification assay in the same animal model (8, 9, 13). Furthermore, by using PMD-gag461 plasmids as DNA standards, the pair of primers in this assay has also been employed in quantification of the proviral DNA of SIVmac251. The detection threshold is as little as 10 copies per reaction, and 50 copies per 106 peripheral blood mononuclear cells (data not shown).
Some experimental interventions such as vaccination and drug treatment may lead to plasma viral load to drop below the threshold of this assay (215copies/mL). To further increase the sensitivity of plasma virus detection, concentration of virus from plasma is required (16) and the method for viral RNA extraction needs to be modified.
In conclusion, the Real-Time RT-PCR based on SYBR green as detected dye is less expensive and more convenient than the assay based on labeled probes (7, 16) and b-DNA assay. The method has application in the accurate determination of SIV RNA viral loads and can aid in furthering our understanding of viral dynamics in animal models.














 DownLoad:
DownLoad: