-
Peste des petits ruminants (PPR) is one of the most important viral diseases with serious socio-economic implications and is estimated to cause loss to the tune of 1, 800 million INR (39 million US$) in India [20]. PPR is a highly contagious, world organization for animal health (OIE) notifiable and transboundary viral disease of small ruminants, especially sheep and goats, characterized by necrotizing, erosive stomatitis, enteritis and pneumonia [1]. The causative agent, PPR virus (PPRV), has been classified under the family Paramyxoviridae, order Mononegavirales and genus Morbillivirus [19].
The virus causes severe destruction of lymphoid cells leading to immunosuppression in the host [13]. Cytokines play a major role in shaping immune response as TH-1/type-1 cytokines elicit predominantly cell-mediated immunity (CMI), whereas TH-2/type-2 cytokines elicit predominantly humoral immunity. Most of the cytokine studies were carried out on Measles Virus (MV).
Experimental PPR infection in goats can lead to marked suppression of host immune responses accompanied by severe leucopenia [13], but the mechanism is not understood. However, it can be speculated that infection interferes with host immune effector cells mediated by cytokines. A study of the relationship between cytokine expression and kinetics of antigen and antibody response in PPR affected animals has not yet been attempted. Therefore the current investigation was undertaken to correlate the cytokine profile and kinetics of antigen and antibody in PPR infected and vaccinated goats. To the best of the authors' knowledge, this is the first report on the cytokine profile in PPR infected goats.
HTML
-
The cytokine gene-specific primers used in the study were designed based on the sequence data available for the cytokine gene in the GenBank database using the sequence analysis software package DNASTAR (Lasergene6 Inc, USA) and Primer3 (an online software tool, http://frodo.wi.mit.edu/ cgi-bin/primer3/primer 3_www.cgi) and were synthesized by M/s Metabion, Planegg-Martinsried, Germany. The oligonucleotide sequences of the primers are as follows. The nucleotide position with respect to gene is indicated in the parenthesis. IL-4 F[5'-CTTAGGCGTATCTACAGGAAC-3'(240-260)] and IL-4R [5'-AGGTCTCTCAGCGTACTTG-3'(355-373)]AY648947;IFN γ F[5'-CAGGAGCTACCGATTTCAGC-3' (5-24)] and IFN γ R [5'-AGGCCCACCCTTAGCTACAT-3' (182-201)] AY603405; β-Actin–F(5'-GAGAAGCTGTGCTACGTCGC-3') and β-Actin R-(5'-CCAGACAGCACTGTGTTG GC-3').
-
Live attenuated PPR vaccine (Sungri/96 strain) (IVRI, Mukteswar) [18] was used to vaccinate the goats. Virulent virus (Izatnagar/94) maintained at the Division of Virology [15] was used to produce experimental PPR disease in goats.
-
One year old non-descriptive healthy hilly goats (both sexes) were screened for PPRV antibody prior to experiment by Competitive ELISA (c-ELISA) [16]and serum neutralization test (SNT) [15] and were randomly divided into Group-A and Group-B, with 4 animals in each group. Group-A animals were inoculated with virulent PPRV using 10% tissue suspension by subcutaneous route (s/c) (4 mL suspension, 2 mL each at two different sites). The nucleocapsid (N) protein was quantified using sandwich ELISA (s-ELISA) [17], which is equivalent to the amount of N protein present in 1 mL of PPR vaccine (titer of 105.5 TCID50/mL) [14]. Group-B was vaccinated s/c with a field dose (103TCID50) of live attenuated PPR vaccine. The goats were provided feed, fodder, and water ad libitum throughout the experiment and housed in different sheds. All the animals were observed for thermal reaction and appearance of PPR-specific clinical signs for 21 dpi. The whole blood, plasma, serum and peripheral blood mononuclear cells(PBMCs) were collected on 0, 1, 3, 5, 7, 9, 12, 15, 18 and 21st dpi from all the experimental animals. All sampwere used for detection of antigen and antibody by ELISA [16, 17], whereas, only PBMCs were used for studying the cytokine expression by real-time reverse transcription polymerase chain reaction (rt RT-PCR).
-
In order to validate the SYBR green based real-time RT-PCR assay, Con-A stimulated PBMCs were used as positive control for generation of standard curve and determination of rt RT-PCR efficiency for cytokine genes. Blood was collected from PPRV antibody negative goats and PBMCs were isolated using density gradient centrifugation [3]. PBMCs thus isolated were cultured in 24-well (2×106 cells/mL) plates in RPMI-1640 media enriched with 10% foetal bovine serum (FBS), 100 μL of 100× antibiotic and antimycotic solutions (Himedia Pvt. Ltd., India) and 2 g of sodium bicarbonate per Liter of media. The cultured PBMCs were simultaneously stimulated with Concanavalin-A (Con-A, 5 μg/mL; Sigma-Aldrich, St. Louis, USA) and harvested at hourly intervals and stored at –20℃ until use.
-
The total RNA was isolated from the harvested PBMCs using Tri®Reagent LS (Sigma-Aldrich, St. Louis, USA) as per manufacturer's protocol and the purity and concentration of RNA was checked by a NanoDrop-2000 spectrophotometer (Thermo Fisher Scientific Inc., Wilmington, DE, USA), and stored at –20℃ until use. 1×10-6g of RNA was reverse transcribed using a RevertAidTM First Strand cDNA Synthesis Kit (M/s MBI Fermentas Life Sciences, Maryland, USA) according to the manufacturer's protocol. The cDNA thus obtained was kept at –20℃ until use.
Cytokine specific-PCR assays were carried out with cDNA obtained from in vitro ConA-stimulated PBMCs. The PCR conditions were in house validated and reconfirmed for each cytokine gene and endogenous control (β-actin). In a 0.5 mL PCR tubes, 5.0 μL of 10× PCR Buffer, 1.5 μL of 50 mmol/L MgCl2, 1.0 μL of 10 mmol/L dNTPs, 1.0 μL each of forward and reverse primers (1×10-5 mol/L), 5.0 μL of cDNA, 0.5 μL of Taq DNA Polymerase (5 U/μL) was added and volume was made up to 50 μL using nuclease free water (NFW). PCR reaction was set with the following amplification conditions: initial denaturation at 94℃ for 2 min, followed by 30 cycles of denaturation at 94℃ for 30 s, annealing at 55℃ for 30 s and extension at 72℃ for 30 s with a final extension for 10 min at 72℃. The amplicon so generated were resolved on agarose (Sigma-Aldrich, St. Louis, USA) gel electrophoresis.
-
The amplicon generated by RT-PCR was gel extracted and cloned into pGEM-T Easy vector. After transformation into E. coli Top10 F' cells, the plasmid was isolated as per manufacturer's protocol using QIAprep® Spin Miniprep Kit (QIAGEN GmbH, Germany). Recombinant plasmid was confirmed by PCR, restriction enzyme digestion and sequence analysis. Efficiency and slopes of real time RT-PCR was determined using standard curves (7500 Real Time PCR System, Applied Biosystem, USA) with ten-fold serial dilution of cloned cytokine specific plasmid DNA [4].
-
Real time RT-PCR was performed using the SYBR Green-I master mix (SYBR Green chemistry), a two-step RT-PCR kit (QuantiTect®SYBR® Green PCR Master mix, Qiagen GmbH, D-40724 Hilden, Germany). Each PCR reaction mix (20 μL) consisted of 10 μL of SYBR Green PCR Master mix, 2.0 μL of cDNA, 0.5 μL each of forward and reverse primers (10 pmol/μL) and 7 μL of NFW. Each reaction was carried out in triplicate with non-template control (NTC) for each gene. Amplification and detection of specific products were performed with the following cycle profile: one cycle of 95℃ for 10 min, and 40 cycles of 94℃ for 30 s, 55℃ for 30 s, 72℃ for 30 s, with one final cycle of standard dissociation curve. The passive reference dye ROX was used for normalization of the reporter signal (SYBR Green). The zero day samples of respective animals was used as the calibrator whereas β actin was employed as the internal/endogenous control. The results were analyzed by ∆∆Ct or delta delta cycle threshold [8]method as per the formula: ΔΔCt = [(Ct target – Ct internal control) sample – (Ct target – Ct internal control) calibrator)]. The change in the gene expression was calculated as 2-ΔΔCt and the value indicated in an n-fold difference relative to the calibrator.
-
PPRV antigen detection from whole blood, serum and plasma was carried out using an s-ELISA kit [17]. Similarly, the samples were tested for anti-PPRV antibodies in terms of percentage inhibition (PI) in a monoclonal antibody (PPRV Haemagglutinin) based ELISA kit [16]. Samples showing PI > 50 were considered as positive for the presence of PPRV antibodies.
Oligonucleotide primers
PPR vaccine and virulent viruses
Experimental Design
Isolation and in vitro culture of peripheral blood mononuclear cells
RNA extraction and RT-PCR
Generation of recombinant plasmid
Real time RT–PCR for cytokine profiling
Detection of PPRV Antigen and Antibody
-
The experiment animals were seronegative to PPR virus-specific antibody in c-ELISA (PI < 50%) and in SNT (titre < 1:2). Different primers showed optimum amplification at 55℃. For detection of cytokine and β-actin genes, cDNA of 6 h (exhibited maximum expression; data not shown) post stimulated PBMCs were used. The specific Tm values, slope values and efficiency of PCR for IL-4, IFN γ and β-Actin was found to be 80℃, -3.384, and 97.5; 78℃, -3.461, 94.5; and 85℃, -3.433, 95.6, respectively. In order to check the modulation in cytokine expression following PPR vaccination and infection, in vivo relative expression was quantified with real time RT-PCR using SYBR Green chemistry.
-
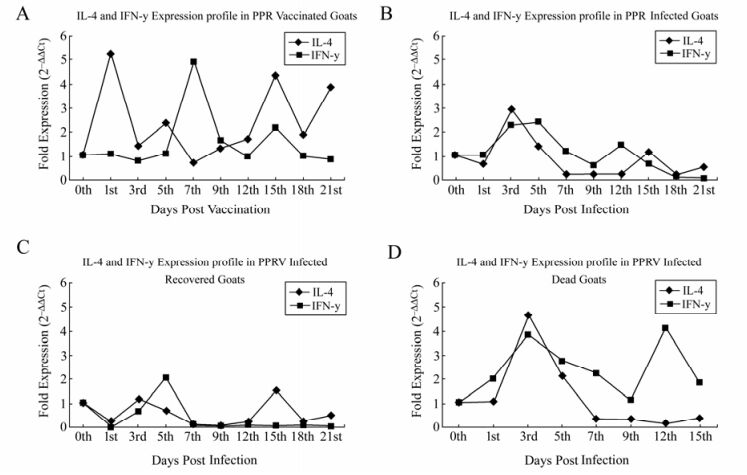
Expression of IFN-γ was more pronounced in early stages of the disease in the infected group (1 to 5 dpi) in comparison to vaccinated animals (Fig. 1A). In the infected group, two goats died and two survived until the end of the observation period. Within the infected group, infection-survived animals showed slight up regulation in early phase (3 to 5dpi) only; no expression was noticed thereafter until the end of the study (Fig. 1B). On the contrary, infected and dead animals had higher expression on 3dpi which gradually receded by 9dpi, and then there was an elevation in expression prior to death (Fig. 1D). Vaccinated animals showed uniform expression throughout the experiment with maximum levels on 7 and 15dpi (Fig. 1A).
-
Both Group A and B revealed up-regulation of IL-4 in the early stage of the disease (1 to 5dpi) and down-regulation in the intermediate phase of the disease (7to 12 dpi), the later was more pronounced in infected animals compared to vaccinated animals. In the late phase (12 to 21dpi), there was a slight up-surge in the expression in vaccinated animals as compared to infected ones (Fig. 1A). In contrary to IFN-γ, within infected groups some difference in IL-4 gene expression was noticed. The animals that recovered from the disease exhibited an almost identical expression pattern to the vaccinated animals, although the intensity was lower (Fig. 1C). Animals that succumbed to disease exhibited relatively higher IL-4 gene expression on 3dpi, and then there was gradual decline in expression until death (Fig. 1D).
-
The presence of PPRV antigen was observed in serum samples from 3dpi, with a sharp rise on 7 dpi to a maximum from 9 to 15 dpi (Fig. 2A and B). Interestingly, in infection-survived animals, the antigen titer was lower compared to infected dead animals. In the case of vaccinated animals, no antigen was detected throughout the experiment except weak positive on 9 dpi. In both groups, detectable level of antibody was noticed from 9 day onwards. There was a gradual increase in titer in infected animals until the end of the experiment; however, the kinetics of the antibody was similar in both recovered and dead animals. In the case of vaccinated animals, a plateau was maintained from 9dpi to the end of the experiment (Fig. 2A and B).
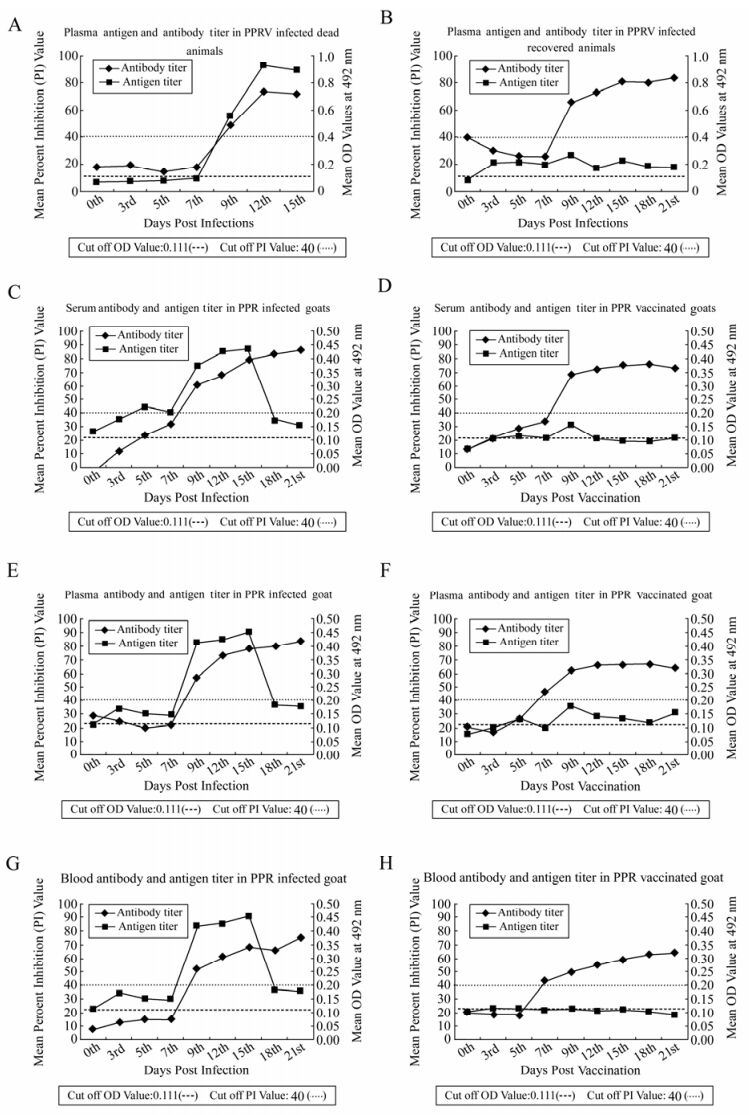
Figure 2. Peste des petits ruminants (PPR) virus antigen and antibody kinetics in infected and vaccinated goats.
The kinetics of antigen in plasma (Fig. 2C and D) was almost similar to what was observed for the serum in both the groups but overall, the plasma exhibited more virus antigen load as compared to serum. In infected animals, plasma antibody level showed a sharp rise on 9 dpi and then a gradual increase up to the end of the experiment (Fig. 2C and D). On the other hand, vaccinated animals exhibited a gradual increase in titer of antibodies from 9 dpv with a plateau response (Fig. 2E and F).
Among all the components of the blood, whole blood had low virus load. In infected animals PPR virus antigen was detected from 7 dpi and reached maximum on 9 dpi. A gradual decline in antigen was observed from 9 to 18 dpi. No PPRV antigen was detected in serum and plasma of vaccinated animals during the study period. However, in the infected group, the antibody level in blood was akin to plasma levels but with less intensity. The appearance of antibody in blood was much earlier in the vaccinated group (5 dpi), than the other two components (Fig. 2G and H).
Cytokine specific PCR and Expression profile
IFN-γ mRNA expression
IL-4 mRNA expression
Antigen and antibody kinetics in various components of blood
-
In this study, the correlation between the kinetics of PPRV antigen and antibody in various components of blood with expression of cytokines was assessed in PPR infected and vaccinated goats. PPR infected animals exhibited both TH1 and TH2 (mixed response) responses in the initial phase of the disease and the findings are in partial agreement with Howe et al. [7], who demonstrated IFN-γ and IL-4 production by PBMCs in response to MV infection in vitro. They showed that these responses are mediated primarily by CD4+ T cells with a TH-1 like phenotype and to a lesser extent by CD4+ T cells with either a TH2 or a TH0 phenotype. They further demonstrated that generation of IFN-γ and IL-4 secreting cells can be modulated in vitro, and probably in vivo, by variable antigen dose. Accordingly, optimum IFN-γ production was elicited at high MV dose and IL-4 at much lower doses. In the present study, goats were inoculated with 10% tissue suspension of virulent PPR challenge virus containing high virus titre and animals were vaccinated with only one vaccine dose (103TCID50/mL). Obviously, the virus load in vaccine is lower than that of virulent virus in the challenge inoculum. This difference in the quantity of virus might have influenced the expression of cytokine in both groups of animals.
It has been established that generation of a TH1 response is less efficient after vaccination than that of natural infection [21] and similar findings were documented for MV [12]. In the present study the infected animals also exhibited mainly TH1 response (increased IFN-γ) as observed in vaccinated goats. However, biphasic response (IL-4 expression) in vaccines is a novel finding in PPR. Both in the initial and later stages of vaccination, a TH2 response (IL-4) were predominant. However, there was an up regulation of IFN-γ in vaccinated goats on 7 dpi and this need to be investigated further. But, it can be hypothesized that as the vaccine is live, there may be periodic replication of virus in the host as evidenced by the presence of antigen in serum and plasma on 9dpv. This periodic replication might affect the cytokine production in general and IFN γ in particular.
Different cytokine expression patterns were observed among the infected animals. Animals which succumbed to PPRV infection expressed a higher level of IFN-γ and antigen than recovered animals. Furthermore, the antigen load in infected-survivor was lower than infected-succumbed animals. However, PPRV antibody titre was akin to vaccinated goats (plateau 15 dpi) and this could be due to better immune response of the animals following infection. The recovered animals might have generated an effective immune response leading to neutralizing/ lowering of circulating antigen. The switching of TH1/TH2 response in vaccinated and infected recovered animals was in accordance with past reports of natural infection of children with MV, where increased production of IL-2 and IFN-γ were observed during the acute phase of the disease, while IL-4 production coincided with antibody-mediated virus clearance [6, 9]. Such switching of TH1/TH2 was also reported in ferrets experimentally infected with canine distemper virus (CDV) [10]. In this study infected and dead animals also showed both TH1 and TH2 response followed by TH1 (IFN-γ) prior to death. This implies that a functional TH2 (IL-4) response is required to generate a specific antibody in order to neutralize the circulating viral antigen.
In this study, the IL-4 expression profile was low in the infected group, which is also concurrent with an earlier report on CDV infection, where TNF-α, IL-6, and IL-2 gene expression were down regulated [10]. Furthermore, it is to hypothesize that the shutdown of cytokine expression with virulent PPRV (Izatnagar/94) might be due to the contact between viral glycoproteins and immune cells in combination with a V protein and phosphoprotein independent inhibition of the induction of the anti-viral state in infected cells, as was reported for CDV [10].
The low PPRV antigen load in whole blood, compared to serum and plasma, could be due to masking of antigen by other intrinsic factors and these findings are in accordance with Frisk et al. [5], who demonstrated the presence of the nucleoprotein of CDV in serum, whole blood, and/or cerebrospinal fluid by using real time PCR. Likewise, Blake et al. [2] and Onishi etal. [11], detected feline immunodeficiecy virus and cytomegalovirus, respectively in plasma by real time PCR. In contrast, no PPRV antigen was detected in blood components of vaccinated animals except on 9 dpv. The antibody titer gradually rose from 3 dpv and remains at a plateau in the later stages in vaccinated animals. In contrast, the level was constant in infected animals till 7dpi. Later, the titre rose up from 7 dpi and attained a plateau afterwards. These findings are in accordance with that of Singh et al. [17].
In summary, the kinetics of PPRV antigen/antibody has a significant involvement in the profile of cytokines, but the exact mechanism is unknown. The drastic reduction in antigen load, especially in infected animals, could possibly be due to virus clearance from the body. In the late phase of the disease, the TH2 response predominates, thereby increasing the levels of IL-4 production. Presence of antigen in blood/serum/plasma could be important in determining the possible role of circulating antigen in virus sequestration/persistence. In the infected group the surge was much higher when compared to the vaccinated group. The upsurge of cytokine response in infected animals could be due to circulation of antigen in the infected animals and sequestration of antigen in vaccinated animals. An interesting correlation is the ability to mount a functional TH2 response and the absence of antigen in blood or serum or plasma. The study suggests that the ability to mount a functional TH2 response after 14 dpi could be a vital determinant in deciding the survival of the PPR infected animals.













 DownLoad:
DownLoad: