-
It was first reported in 1997 that H5N1 influenza viruses were transmitted from birds to humans in Hong Kong, resulting in 18 infected cases and 6 deaths (Claas E C, et al., 1998; Subbarao K, et al., 1998). As of March 12, 2013, 15 countries have reported 371 deaths among 622 confirmed H5N1-infected human cases, a mortality rate of about 60% (WHO, 2013). Recently, two studies indicate that H5N1 highly pathogenic avian influenza viruses (HPAIV) have the potential to evolve directly to transmit between mammals and pose a risk of causing human pandemics in the future (Herfst S, et al., 2012; Imai M, et al., 2012). Therefore, is an urgent need to develop effective vaccines against H5N1 HPAIV.
Most vaccines against H5N1 HPAIV target the surface glycoprotein, hemagglutinin (HA), which is involved in receptor-binding, membrane fusion, and induction of neutralization antibodies in the host (Skehel J J, et al., 2000). It is initially synthesized as a single polypeptide precursor (HA0) and then can be cleaved into HA1 and HA2 subunits by trypsin-like proteases. The amino acids and the structure of HA0 cleavage site is a determining factor of viral pathogenicity (Chen J, et al., 1998; Senne D A, et al., 1996; Steinhauer D A, 1999). Most influenza viruses can initiate infection only in the respiratory or intestinal tracts of the host as they have a monobasic cleavage site in HA, which can only be activated by extracellular trypsin-like proteases. However, highly pathogenic influenza viruses, such as H5N1 and H7N1 HPAIV, can spread throughout the host and cause systemic infection as they have a multibasic cleavage site (MBCS) in HA, which can be cleaved by ubiquitously expressed endogenous proteases in various types of cells (Horimoto T, et al., 1994; Okumura Y, et al., 2010; Stieneke-Grober A, et al., 1992). The MBCS in the HA of H5N1 HPAIV is associated with viral infection and spread in both chickens and mammals (Horimoto T, et al., 1994; Schrauwen E J, et al., 2012; Suguitan A L, Jr., et al., 2012; Yin J, et al., 2013).
The MBCS of H5-HA is usually modified to a monobasic one in order to weaken the virulence of the recombinant virus and to generate inactivated and live attenuated vaccines against H5N1 HPAIV from chick embryos (Bresson J L, et al., 2006; Li S, et al., 1999; Lin J, et al., 2006; Stech J, et al., 2005; Subbarao K, et al., 2003; Suguitan A L, et al., 2009; Webby R J, et al., 2004). For inactivated vaccines, this modification changes the cleavability of HA, attenuates viral pathogenicity, but retains the antigenicity of HA (Subbarao K, et al., 2003). On the other hand, this modification has been shown to have a deleterious effect on the immunogenicity and efficacy of the Delta H5N1 vaccine based on investigations of the A/Ann Arbor/6/60 (H2N2) cold-adapted (ca) virus in mice (Suguitan A L, et al., 2009). However, insect-cell derived influenza virus-like particles (VLPs) containing mutant H5-HA with a mutation within the MBCS are as immunogenic as VLPs containing wild type HA (Song J M, et al., 2010). Whether such a modification affects new candidate vaccines against H5N1 HPAIV generated from mammalian cells has not been adequately investigated (Ehrlich H J, et al., 2008; Howard M K, et al., 2008; Kistner O, et al., 2007; Tambyah P A, et al., 2012; Wu C Y, et al., 2010).
Several groups have used retroviral or lentiviral vectors to generate influenza pseudotypes from mammalian cells and these pseudotypes can be used to evaluate influenza neutralization antibodies or to screen influenza inhibitory molecules (Ao Z, et al., 2008; Garcia J M, et al., 2010; Nefkens I, et al., 2007; Temperton N J, et al., 2007; Tsai C G, et al., 2009; Wang S Y, et al., 2009; Zhang S, et al., 2008). H5-HA pseudotyped lentivirus can also provide protection to mice (Zhang S, et al., 2008). Previously, we used MLV-derived retroviral vectors, pHCMV-N1, and pHCMV-wtH5, a plasmid expressing wild type HA (wtH5) of A/swine/Anhui/ca/2004 (H5N1) virus, to package wtH5N1 pseudotypes from 293T cells (Tao L, et al., 2012). Here, we generated mtH5N1 pseudotypes, which contained a mutant type (mtH5) and which contained a monobasic cleavage site with an amino acid 'T' replacing amino acids 'RRKRR' in the MCBS of wtH5 (Tao L, et al., 2012). By subcutaneously vaccinating mice with the two types of pseudotypes, we compared the specific immune responses and protective immunity induced by them in mice and found that both influenza pseudotypes could provide protective immunity to mice, and mtH5N1 pseudotypes were superior to wtH5N1 pseudotypes.
HTML
-
A rabbit polyclonal avian influenza A neuraminidase antibody was purchased from Abcam (Cambridge, UK). Alkaline phosphatase (AP)-conjugated goat anti-rabbit IgG and AP-conjugated goat anti-mouse IgG were purchased from Pierce (Thermo Scientific, Rockford, IL, USA). Rabbit polyclonal anti-HA1 serum and rabbit polyclonal anti-P30 serum were produced in our lab.
-
The cell lines 293T (human kidney epithelium) and Madin-Darby canine kidney (MDCK), were purchased from the China Center for Type Culture Collection (Wuhan, Hubei, China), and maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% (v/v) heat-inactivated (30 min, 56 ℃) fetal bovine serum (FBS; Gibco, Grand Island, NY, USA), 25 mmol/L HEPES, 100 U/mL penicillin and 100 μg/mL streptomycin at 37 ℃/5% CO2. The influenza virus A/Chicken/Henan/12/2004 (H5N1) was isolated in Henan province, China, and mouse-adapted virus was prepared as described by Zheng et al. (Zheng L, et al., 2009). In this study, the virus was used to challenge immunized mice.
-
The retroviral vector system, pMP71-eGFP-pre, pSVMo-MLVgagpol and pHCMV-SARS-SCΔ19, were kindly provided by Tsanan Giroglou at the Institute for Biomedical Research, Frankfurt University Medical School (Giroglou T, et al., 2004). Influenza Env-expressing plasmids pHCMV-wtH5, pHCMV-N1 and pHCMV-mtH5 were constructed and purified in our lab (Tao et al., 2012).
-
Influenza pseudotypes, named as wtH5N1 pseudotypes, were produced as previously described (Tao L, et al., 2012). To produce mtH5N1 pseudotypes, 293T cells (5 × 106 cells per 100 mm tissue culture plate) were transfected with 7.5 μg of pMP71-eGFP-pre, 12.5 μg of pSV-Mo-MLVgagpol, 2 μg of pHCMV-mtH5, and 1 μg of pHCMV-N1 using the ProFection Mammalian Transfection System (Promega, Madison, WI, USA). Cell culture medium was replaced with 7 mL DMEM/2% FBS at 6 h post-transfection. Supernatants containing pseudotypes were harvested at 24 h and 48 h post-transfection, and then cleared by spinning at 3500 rpm for 10 min at 4 ℃, filtered through 0.45-μm pore size filters (Millipore, Jersey City, NJ, USA). They were stored at 4 ℃ overnight before ultracentrifugation or at -80℃ in aliquots before carrying out transduction and titration assays as follows.
-
To verify the existence of influenza pseudotypes, the collected supernatants were used for transduction and titration assays. MDCK cells were seeded at a concentration of 1.5×104 cells/well into 96-well tissue culture plates, and on the next day, they were transduced with 100 μL supernatant containing influenza pseudotypes in the presence or absence of L-(tosylamido-2-phenyl) ethyl chloromethyl ketone (TPCK)-trypsin (5 μg/mL, Worthington Biochemicals Corp., NJ, USA) at 37 ℃ for 1 h. After incubation at 37 ℃ for 48 h, cells were inspected for fluorescence under an inverted fluorescence microscope. Titers of influenza pseudotypes were measured by end-point dilution assay and calculated by the Reed-Muench method. Supernatants of equal volumes or supernatants containing equal quality of HA (wtH5N1 pseudotypes) or HA0 (mtH5N1 pseudotypes) were used in the titration assay. The HA content was quantified by an H5N1 (Avian Flu) Hemagglutinin ELISA kit (Sino Biological Inc, Beijing, China) following the manufacturer's instructions. MDCK cells were seeded as mentioned above and transduced with ten-fold serial dilutions of supernatants containing influenza pseudotypes (equal in volume or in HA content) in the presence of 5 μg/mL TPCK-trypsin. After incubation at 37 ℃ for 48 h, cells were examined, and GFP-positive clusters were counted and expressed as transduction units (TU). Titers of influenza pseudotypes were calculated and displayed as TU/mL or TU/ng.
-
As previously described, influenza pseudotypes were purified (Zhang S, et al., 2008) and the total protein was measured (Tao L, et al., 2012). The HA/HA0 contents of wtH5N1/mtH5N1 pseudotypes were estimated to be 15% of the total proteins by densitometric measurement of target protein bands in repeated Western blotting results (data not shown). Purified pseudotypes were aliquoted and stored at -80 ℃ until needed. Aliquots of purified influenza pseudotypes (about 5 μg) were treated by 5 μg/mL TPCK-trypsin, or digested by 750 U of peptide N-glycosidase F (PNGase F; New England Biolabs, MA, USA) as specified by the supplier, or mock treated at 37 ℃ for 1 h. Then, the reaction was stopped by the addition of 6× protein sample buffer and heating at 95 ℃ for 10 min. Then, the samples were separated by 10% SDS-PAGE, transferred to nitrocellulose and analyzed by Western blotting using rabbit anti-HA1 serum (1:1 000), avian influenza A neuraminidase antibody (1:1 000, Abcam), or rabbit anti-P30 serum (1:1 000) as the first antibody, and AP-conjugated goat anti-rabbit IgG (1:10 000, Pierce) as the second antibody. By densitometric measurement of protein bands of the mock treated samples using the Quantity One software package (Bio-Rad), the HA content of purified wtH5N1 pseudotypes was calculated from the HA1 band, and the HA0 content of purified mtH5N1 pseudotypes was determined (Lu X, et al., 1999).
-
Purified pseudotypes were also subjected to transmission electron microscopy (TEM) analysis. In brief, pseudotypes were absorbed onto Formvar and carbon-coated copper grids (200 mesh), negatively stained with 2% phosphotungstic acid (PTA, pH 7.0), and examined at 200 kV in a Tecnai G2 Spirit Transmission Electron Microscope (TEM; FEI, Hillsboro, OR, USA).
-
In order to investigate the immunogenicity of influenza pseudotypes, female Balb/c mice, 6 to 8 weeks old, were purchased from the Center for Disease Control and Prevention (Changsha, Hunan, China), maintained in a specific pathogen free (SPF) environment, and provided with SPF water and food. Mice were randomly divided into three groups (5 mice per group), and each mouse was vaccinated by subcutaneous injection for three times at 2-week intervals. In two groups, each mouse was immunized with wtH5N1 pseudotypes or mtH5N1 pseudotypes, comprising about either 3 μg HA or HA0 and diluted into 100 μL PBS. The third group was immunized with 100 μL PBS per mouse and used as a control. Before each immunization and on the 10th day after the final boost, blood samples were collected from the lateral canthus of mice. Serum samples were collected and stored at -20 ℃ in aliquots. The serum level of anti-HA antibodies (IgG) was assessed by an indirect ELISA as previously described (Tao L, et al., 2012). Briefly, purified prokaryotic recombinant HA1 protein (amino acids 1 to 329 of wtH5) with optimized concentration (1 μg per well in 100 μL coating buffer) were coated onto 96-well ELISA plates overnight at 4 ℃. After blocking with PBS/1% BSA and 1-hour incubation with 1:100 diluted mouse sera at 37 ℃, plates were washed and bound antibodies were detected by AP-conjugated goat anti-mouse IgG (1:3 000). The reaction was visualized by adding paranitrophenyl phosphate (pNPP) substrate, and the absorbance at 405 nm (A405) was measured in a Synergy HT Multi-Mode Microplate Reader (Bio-TEK, Winooski, VT, USA).
To evaluate the cellular immune response, three mice from each group were sacrificed by cervical dislocation and spleens were isolated on the 10th day after the final immunization. Single-cell suspensions of mouse splenocytes were prepared as previously described (Lu B, et al., 2009). ELISPOT assays were carried out using the Mouse IFN-γ ELISPOT kit (U-CyTech, Utrecht, Netherlands) following the manufacturer's instructions, with slight modifications as previously described (Zheng L, et al., 2009). Briefly, mouse splenocytes (2.5×105 per well) were stimulated with 10 μg/mL purified recombinant HA1 protein for 30 h and all stimulation conditions were tested in quadruplicate, with controls, in parallel. The spots were scanned and counted in an ELISPOT BioReader 4000 Pro-X (Bio-Sys Limited Germany, Hamburg, Germany). The number of spot forming cells (SFC) per 106 splenocytes cells was used to express ELISPOT results.
-
Mice were randomly divided into three groups (6 mice each group) and immunized with PBS, wtH5N1 pseudotypes, or mtH5N1 pseudotypes subcutaneously for three times, with boosting on the 14th day and on the 44th day. Six weeks after the final boost, mice were intranasally (i.n.) infected with 5 x 50% mouse lethal doses (MLD50) of mouse-adapted A/Chicken/Henan/12/2004 (H5N1) viruses in a Biosafety Level 3 (BSL-3) Lab. Mice were anesthetized by intraperitoneal injection with the appropriate volume of 0.5% sodium pentobarbital and infected by intranasal drip with mouse-adapted A/Chicken/Henan/12/2004 (H5N1) viruses (5 × MLD50 in 20 μL PBS per mouse), and then observed to record the survival rate and body weight change for 2 weeks.
-
Statistical analysis was performed with SPSS 13.0 for Windows and assessed by the Student's t-test. P-values less than 0.05 were considered statistically significant.
Antibodies and sera
Cell lines and viruses
Plasmids
Influenza pseudotypes production
Transduction and titration
Western blotting analysis
Electron microscopy analysis
Immunization of mice
Challenge experiment
Statistical analysis
-
Influenza pseudotypes were packaged by co-transfection of 293T cells with retroviral vector plasmids, pMP71-eGFP-pre, pSV-Mo-MLVgagpol and Env-expressing plasmids, pHCMVN1 and pHCMV-wtH5 or pHCMV-mtH5. To investigate the transduction efficacies of influenza pseudotypes, MDCK cells were transduced with supernatants containing wtH5N1 pseudotypes or mtH5N1 pseudotypes from transiently transfected 293T cells. Bright fluorescence was observed in MDCK cells transduced with wtH5N1 pseudotypes in the absence or presence of TPCK-trypsin (Fig. 1A and B) and in those transduced with mtH5N1 pseudotypes in the presence of TPCK-trypsin (Fig. 1D), but no detectable fluorescence was observed in those transduced with mtH5N1 pseudotypes in the absence of TPCK-trypsin (Fig. 1C). Thus, activation of mtH5N1 pseudotypes was dependent on exogenous TPCK-trypsin.
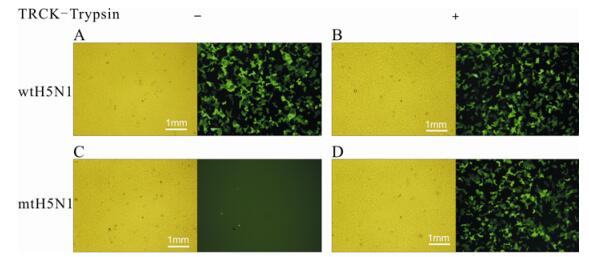
Figure 1. Transduction of MDCK cells with influenza pseudotypes. MDCK cells (1 × 104 cells per well) were seeded into 96-well plates and on the following day, they were transduced with 100 μL supernatants containing either wtH5N1 pseudotypes (A & B) or mtH5N1 pseudotypes (C & D) in the absence (A & C) or presence (B & D) of TPCK-trypsin (5 μg/mL). The fluorescence of cells was inspected under a fluorescent microscope (100×) after incubation at 37 ℃/5% CO2 for 48 h. Phase-contrast and fluorescence images are shown.
Titers of influenza pseudotypes in the supernatants were also detected, as shown in Fig. 2A and B. From comparison to wtH5N1 pseudotypes, an approximate 2-fold increase of titers could be observed in mtH5N1 pseudotypes, indicating that particles of mtH5N1 pseudotypes in the supernatants were more than that of wtH5N1 pseudotypes.

Figure 2. Titration of influenza pseudotypes on MDCK cells. Ten-fold dilutions of supernatants containing either wtH5N1 pseudotypes or mtH5N1 pseudotypes, equal in volume or in HA/HA0 content, quantified by an H5N1 (Avian Flu) Hemagglutinin ELISA kit, were used to transduce MDCK cells in the presence of TPCK-tyrpsin (5 μg/mL). After incubation for 48 h, the fluorescence of cells was examined. Titers were calculated and displayed as TU per mL (TU/mL, A) or TU per ng HA (TU/ng, B). Results are the mean ± SD of three stocks of either type of influenza pseudotypes conducted in duplicate experiments. TU, transduction units.
To further characterize influenza pseudotypes, aliquots of purified pseudotypes were treated with TPCK-trypsin, or PNGase F, or mock-treated and then subjected to Western blotting analysis. After treatment by TPCK-trypsin and PNGase F, the NA bands of both pseudotypes migrated faster (Fig. 3A and B). In the TPCK-trypsin-treated mtH5N1 pseudotypes, the HA0 band was barely visible and a band similar to the HA1 band of wtH5N1 pseudotypes appeared (Fig. 3A). After treatment by PNGase F, the HA1 band of wtH5N1 pseudotypes and the HA0 band of mtH5N1 pseudotypes also had faster mobility (Fig. 3B). This indicated that both influenza pseudotypes contained glycoproteins, but HA was cleaved into HA1 and HA2 in wtH5N1 pseudotypes, while the precursor HA0 existed in mtH5N1 pseudotypes.
Figure 3. Examination of the main proteins in influenza pseudotypes. Purified influenza pseudotypes were aliquoted to 5 μg per tube, and treated with TPCK-trypsin (A), or PNGase F (B) at 37 ℃ for 1 h. Samples were separated by 10% SDS-PAGE and then subjected to Western blotting analysis using the rabbit anti-HA1 serum (1:1 000, top), the avian influenza A neuraminidase antibody (1:1 000, middle) or the rabbit anti-P30 serum (1:1 000, bottom). (#) indicates positions of proteins treated by TPCK-trypsin, (*) indicates positions of proteins treated by PNGase F, and the prestained protein markers are indicated on the left side.
We also used TEM analysis to inspect the morphology of influenza pseudotypes. As shown in Fig. 4, influenza pseudotypes all had thick, spike-like layers but with exterior similar to wild-type influenza virus.
-
To investigate the HA specific humoral immune responses induced in mice by the influenza pseudotypes, the sera from immunized mice were collected before each immunization and on the 10th day after the final boost and HA specific IgG antibodies were detected by indirect ELISA. Compared with the control group, both groups immunized with influenza pseudotypes elicited HA specific serum IgG after boost immunization (P < 0.05, Fig. 5A), and the IgG level of those immunized with Fig. 5A). Therefore, both influenza pseudotypes could induce an HA specific humoral immune response, and the mtH5N1 pseudotypes enhanced the humoral immune response after boost immunization.
Figure 5. HA specific humoral and cellular immune responses were enhanced by mtH5N1 pseudotypes. Mice were divided into three groups (n=5 per group) and immunized subcutaneously three times at 2-week intervals. One group of PBS-immunized mice served as a control. Each mouse of the other two groups was immunized with either wtH5N1 pseudotypes or mtH5N1 pseudotypes containing 3 μg HA or HA0. A: Sera were collected before each immunization and on the 10th day after the final boost. HA-specific IgG in the serum was detected by an indirect ELISA with duplicate replications. Results are expressed as mean ± SD (n = 5). B: On the 10th day after the final boost, spleens were separated from three sacrificed mice of each group. Splenocytes were isolated and stimulated with recombinant HA1 protein in ELISPOT assay to score IFN-γ producing cells. The results are expressed as the number of spot forming cells (SFC) per 106 cells. The values represent the average of triplicate wells of three mice, and are displayed as mean ± SD (n = 3). Compared with the group immunized with wtH5N1 pseudotypes, the levels of serum IgG after each boost and the secretion of IFN-γ after the final boost were significantly higher in the group immunized with mtH5N1 pseudotypes (* P < 0.05).
The cellular immune response was also evaluated in these immunized mice after the second boost by measuring the secretion of IFN-γ (Fig. 5B) and IL-4 (data not shown). Mice immunized with influenza pseudotypes induced HA specific IFN-γ secretion relative to controls, and the level of IFN-γ secretion in the group immunized with mtH5N1 pseudotypes was about six times of that induced by wtH5N1 pseudotypes (P < 0.05, Fig. 5B), strongly supporting the conclusion that both influenza pseudotypes could induce an HA specific cellular immune response, and the mtH5N1 pseudotypes enhanced the cellular immune response in immunized mice.
-
To evaluate the protective efficiency of influenza pseudotypes against an influenza virus challenge, a challenge experiment was carried out in a BSL-3 facility. Compared to the control group, mice immunized with influenza pseudotypes showed a higher survival rate (Fig. 6A) and less body weight loss (Fig. 6B). Mice in the control group showed the most obvious weight loss and the average weight on day 3, 7, 10 and 14 was 101.2%, 86.82%, 79.85% and 79.17% of the original weight on day 0 post-challenge. While mice immunized with wtH5N1 pseudotypes showed little of weight loss and the average weight on day 3, 7, 10 and 14 was 97.33%, 95.96%, 98.17% and 98.84% of the original weight. In contrast, mice immunized with mtH5N1 pseudotypes displayed no weight loss and their average weight was 100.40%, 104.85%, 103.90% and 105.72% of the original weight on day 3, 7, 10 and 14, respectively. The survival rate of the control group, wtH5N1 pseudotypes group and mtH5N1 pseudotypes group was 16.7%, 83.3% and 100%, respectively. The results demonstrated that both influenza pseudotypes could provide protective immunity to mice, and mtH5N1 pseudotypes were superior to wtH5N1 pseudotypes.
Figure 6. Protective efficacies of influenza pseudotypes in mice. Groups of mice (n = 6) were immunized as described in Fig. 3. Six weeks after the final boost, mice were challenged with 5 × MLD50 of mouse-adapted A/Chicken/Henan/12/2004(H5N1) viruses and observed for 14 days after infection to record the body weight change (A) and survival rate (B). Mean body weight change is expressed as a percentage of original body weight and displayed as mean ± SD (n = 6).
Generation and characterization of influenza pseudotypes
Enhancement of immune responses by mtH5N1 pseudotypes
Superior protection efficacy of mtH5N1 pseudotypes
-
The highly pathogenic H5N1 influenza viruses have a characteristic multibasic signature at the HA1/HA2 cleavage site of the HA protein, and this is thought to be related to the cleavability of HA and viral pathogenicity (Steinhauer D A, 1999). In this study, we generated two influenza pseudotypes, wtH5N1 pseudotypes and mtH5N1 pseudotypes, which had different numbers of basic amino acids within the H5-HA cleavage site. In particular, the multibasic cleavage site of wtH5 was modified to be monobasic in mtH5. We found that these mtH5N1 pseudotypes, packaging mtH5 with a monobasic cleavage site, showed transduction ability only in the presence of exogenous trypsin-like proteases, displaying the HA0 precursor, inducing stronger humoral and cellular immune responses than wtH5N1 pseudotypes in immunized mice, and providing more effective protective immunity. This study demonstrates that modification of the H5-HA MBCS may be beneficial to the development of new candidate vaccines from mammalian cells to combat H5N1 HPAIV.
Influenza pseudotypes can be used to elicit antibodies in immunized mice, and can be employed in the study of HA and in the detection of HA antibodies, which is important for vaccine evaluation and sera surveillance (Garcia J M, et al., 2010; Haynes J R, et al., 2009; Ho J W, et al., 2008; Kong W P, et al., 2006; Lin A H, et al., 2002; Nefkens I, et al., 2007; Tsai C G, et al., 2009; Wang S Y, et al., 2009; Wang W, et al., 2010; Wang W, et al., 2008; Wu Q, et al., 2009; Zhang S, et al., 2008). Here, we generated influenza pseudotypes packaging mammalian expressed HA (wtH5 or mtH5) and N1 proteins around the core of MLV using a strategy similar to that previously described by Szecsi et al (Szecsi J, et al., 2006). We observed that the transduction ability of wtH5N1 pseudotypes was independent of the presence of additional TPCK-trypsin, while mtH5N1 pseudotypes showed transduction activity only in the existence of exogenous TPCK-trypsin, similar to recombinant influenza A virus vaccines reported by Li et al. (Li S, et al., 1999). As indicated by the results of Western blotting analysis, wtH5N1 pseudotypes display cleaved HA, while mtH5N1 pseudotypes carry the precursor HA0. We speculate the difference between wtH5N1 pseudotypes and mtH5N1 pseudotypes is mainly caused by the different forms of HA packaged by them.
By evaluating HA specific immune responses of mice immunized with influenza pseudotypes, we observed that mtH5N1 pseudotypes, as well as wtH5N1 pseudotypes, could induce humoral and cellular immune responses in mice. Additionally, mtH5N1 pseudotypes elicited a stronger humoral immune response and Th1 type cellular immune response than wtH5N1 pseudotypes, as sera IgG and IFN-γ secretion were enhanced. We also found mtH5N1 pseudotypes were superior to wtH5N1 pseudotypes in providing protection to mice against an H5N1 influenza virus challenge. This is partly consistent with a previous study, in which modification of the multi-basic cleavage site of H5-HA to a monobasic one did not alter the antigenicity of H5-HA (Li S, et al., 1999). But our results were different to those from another study, based on a live attenuated influenza A H5N1 cold-adapted vaccine virus which demonstrated modification of the multibasic cleavage site of H5-HA to a monobasic one had a deleterious effect on the immunogenicity and efficacy of the ΔH5N1 vaccine in mice (Suguitan A L, et al., 2009). These differences may be a consequence of differences in administration routes and variations in experimental details, especially the choice of specific immunogens employed in these experiments. The live attenuated influenza virus with a monobasic cleavage site in H5-HA can only replicate in the cooler upper respiratory tract and its action is restricted in the warmer temperature of the lung. Furthermore, the cleavage of H5-HA with a monobasic cleavage site and related virus activation is dependent on extracellular trypsin-like protease, while the live attenuated influenza virus with a MBCS in H5-HA replicates better in several tissue cells since it is independent of extracellular protease but dependent on intracellular endogenous protease. Thus, the former is less immunogenic than the latter and induces weaker responses afterwards in mice. Song et al. reported that influenza VLPs containing mutant HA without the multi-basic cleavage site (ΔVLP) displayed an uncleaved precursor conformation, and elicited immune responses comparable to VLPs containing wild type HA (wtVLP) produced from baculovirus-insect cell system (Song J M, et al., 2010). The authors speculated that ΔVLP possibly provided superior protection versus wtVLP as evidenced by effective viral clearance. The present study shows results analogous to what Song et al. reported, and our conclusion is more reliable to support the hypothesis that the H5-HA with a monobasic cleavage site stimulates stronger immune responses in mice than the H5-HA with MBCS. As immunogens such as VLPs and pseudotypes cannot replicate in immunized mice, the immune responses are induced only once upon vaccination and are different from live attenuated vaccines which can reproduce immunogens until they are cleared by the host.
Although we found the mtH5N1 pseudotypes could enhance immune responses in immunized mice and provide more effective immunity to them than wtH5N1 pseudotypes, we are still unclear regarding the mechanisms leading to these differences. Observations of the cellular immune response to influenza A virus suggest that there are two distinct pathways of protein antigen presentation to T lymphocytes (Braciale T J, et al., 1987). We suspect that the form of HA may affect the antigen presentation pathway of influenza pseudotypes as an earlier report showed that cleavage of HA0 results in structural rearrangements to facilitate conformation change for fusion (Chen J, et al., 1998). While wtH5N1 pseudotypes with cleaved HA1 and HA2 could fuse with antigenpresenting cells (APCs) and facilitate their entry, mtH5N1 pseudotypes remain inactivated and may not fuse with APCs in the absence of exogenous trypsin-like proteases. Another reason may be that mtH5N1 pseudotypes are superior to wtH5N1 pseudotypes in quantity, or stability, or both. However, definitive causes have to be investigated more extensively by additional experiments.














 DownLoad:
DownLoad: