HTML
-
Infectious retroviruses are exogenous retroviruses that have invaded into the host cell, whereby the genomic RNA becomes reverse-transcribed into double-stranded DNA and ultimately inserted into the host cell genome. Such exogenous retrovirus invasion into the germ cells of the host has resulted in a large number of stable retroviral sequences integrated into the host genome that have become part of the host genome(Boeke and Stoye, 1997; Nakaya and Miyazawa, 2015). These retroviruses are collectively referred to as endogenous retroviruses(ERVs). Almost all animal species have ERV sequences in their genomes; for example, ERVs constitute 8% of the human genome(Lander et al., 2001) and 18% of the bovine genome(Adelson et al., 2009).
The complete ERV genomic structure is similar to the exogenous retroviral genomic structure, generally consisting of 5′-and 3′-long terminal repeats and four structural genes: gag, pro, pol, and env(Herniou et al., 1998; Lokossou et al., 2014). Most ERVs are destined toward annihilation if their expression has detrimental effects for the host. As a consequence, the accumulation of mutations, deletions, rearrangements, and methylation usually result in a replication defect, which has been fine-tuned throughout their evolution in the host genome(Boeke and Stoye, 1997; Black et al., 2010). Although most ERVs are inactivated, some of the ERV genes are also expressed in the host cells(Lokossou et al., 2014; Nakaya and Miyazawa, 2015).
ERVs show an extraordinary 'affinity' to the placenta based on evidence that ERV particles and mRNAs are often detected in the placenta(Johnson et al., 1990; Harris, 1998; Rote et al., 2004) and that several genes of the host use retroviral promoters to induce placenta-specific transcripts(Rote et al., 2004). Moreover, some envelope genes of ERVs have been found to be essential for the development and formation of the placenta in humans(Mi et al., 2000; Esnault et al., 2008), rabbits(Heidmann et al., 2009), cattle(Adelson et al., 2009), and mice(Dupressoir et al., 2005). Extensive research has confirmed that ERV envelope proteins(termed 'syncytins')promote the cellto-cell fusion of trophoblast cells, and play an important role in the process of syncytiotrophoblast formation in the placenta in primates, rodents, ruminants, lagomorphs, and carnivores(Mi et al., 2000; Frendo et al., 2003; Dupressoir et al., 2009; Dupressoir et al., 2011; Haig, 2012).
In this review, we focus on the biological roles of ERVs envelope proteins in the development of the placenta. We further discuss the existing controversy in the field and highlight areas of future research and the value of knowledge gained from ERV studies.
-
A retrovirus is an RNA virus whose genome contains at least four genes(gag, pro, pol, and env)(Vogt, 1997). The gag gene encodes the major structural polyprotein Gag, including nucleocapsid(NC), matrix(MA), and capsid(CA), and all elements required to form the viral particle core. The pro gene encodes the viral protease, and is essential for promoting the ripeness of viral particles. The pol gene encodes several products, including reverse transcriptase, RNase H, and integrase. The env gene is essential for the viral surface protein(SU) and transmembrane protein(TM)of the envelope protein(Env), which is responsible for the binding to cellular receptors and membrane fusion. Phylogenetic analyses have classified all known retroviruses into three broad groups(Tristem, 2000; Gifford and Tristem, 2003; Henzy and Johnson, 2013). Here, we focus on ERVs of class Ⅰ(epsilonretroviruses, betaretroviruses, and their associated ERVs) and class Ⅱ(alpharetroviruses, betaretroviruses, lentiviruses, and their associated ERVs), as well as on the structure and function of Env proteins(Benit et al., 2001; Katzourakis et al., 2009; Henzy and Johnson, 2013).
The Env proteins of endogenous and exogenous retroviruses are similar. The retroviral Env protein is translated in the host cell where it presents as a trimer precursor in the endoplasmic reticulum. After being transported to the Golgi apparatus, the trimer precursor is cleaved into two subunits, SU and TM, by the host's protease. They combine through non-covalent binding, as in the case of betaretroviruses and lentiviruses, or by virtue of a single intersubunit disulfide bond, as observed in gamma-, delta-and alpha-retroviruses(Leamnson and Halpern, 1976; Pinter et al., 1978; Kowalski et al., 1987; Smith and Cunningham, 2007; Li et al., 2008; Henzy and Coffin, 2013; Henzy and Johnson, 2013). The Env trimers become fixed on the cellular membrane via the TM subunit(Swanstrom and Wills, 1997), whereas the majority of the SU is exposed to the host immune system. The SU includes a signal peptide and a receptor-binding domain. There is a cleavage site between the SU and TM subunits comprising a polybasic motif(K / R-X -K / R -R)(Hosaka et al., 1991); this sequence also marks the beginning of the TM region(Henzy and Johnson, 2013). There are two hydrophobic domains in the TM subunit: the first domain forms the fusion peptide at or near the N-terminus, and the second is the membrane-spanning region. The ectodomain contains two heptad-repeat regions(hr1 and hr2), which are essential components for trimer formation and the dynamic rearrangement during the process of fusion, and form the highly conserved coiled-coil structure that is observed in many viral envelope proteins(Li et al., 2008). The ectodomain sequence of some retroviruses contains a recognized immunosuppressive domain(ISD)(Benit et al., 2001). The transmembrane region of the C-terminal is the cytoplasmic tail region, which is located in the cytoplasm of the host cell membrane before virus particle assembly and on the inner side of the membrane after assembly.
Within the Env trimer at the surface of the viral particle, the SU maintains the TM in a metastable conformation that is similar to a 'spring-loaded' model(Henzy and Coffin, 2013). Therefore, during the invasion process, the virion of retroviruses makes contact with the host cell via the SU, which binds to its receptor on the target cell(Goff, 2001; Hofacre and Fan, 2010). The virus particles make contact with the host's cell surface-specific receptors via the SU subunit of the Env protein when the retrovirus invades the host cell. Binding of the SU subunit to the receptor induces a conformational change that enables SU to be exposed to the TM subunit so that the fusion peptide is inserted into the cell membrane. This causes the TM subunits to fold into a highly stable trimer-shaped 'hairpin' structure, thereby causing the alpha-helical coiled-coils of hr1 and hr2 to make contact(Henzy and Johnson, 2013). As Env trimers transition from a metastable state to a stable state, the released energy drives the fusion of the cellular and viral membranes(Swanstrom and Wills, 1997). The envelope protein of ERVs uses the same mechanism to promote the fusion of trophoblast cells for formation of the placental syncytial layer. The ISD within the TM subunit allows for the virus particles to escape from the host immune system. Specifically, several studies have indicated that the ISD in the TM subunit of the envelope protein of ERVs can inhibit the mother's immune system from attacking against a fetus(Hosaka et al., 1991; Singh et al., 1999; Hofacre and Fan, 2010; Haig, 2013).
The envelope of ERVs have been frequentlydetected in the reproductive tract and placenta, and appear to show a specific preference to the placenta(Bierwolf et al., 1975; Kalter et al., 1975; Imamura et al., 1976; Smith and Moore, 1988; Harris, 1991; DeHaven et al., 1998; Haig, 2013). This affinity has piqued the interest of many researchers into discovering the possible function of active endogenous retroviruses in the placenta. Some researchers have suggested that retroviral promoters play a significant role in placental development(Harris, 1991; DeHaven et al., 1998; Harris, 1998). In particular, it has been proposed that placental trophoblastic cells create a comfortable and suitable environment for ERV activity, and the placenta has become 'addicted' to taking advantage of ERV-derived promoters in the course of its developmental process(Chuong, 2013; Haig, 2013). Some of other scientists insist that retroviral genes have been adopted for several placental functions(Dolei et al., 2015). In fact, independent capture events have been confirmed in primates, rodents, ruminants, carnivores, and lagomorphs, showing the participation of retroviral genes in the formation of the syncytium layer via trophoblast cellcell fusion at the fetomaternal interface(Cornelis et al., 2013). In the following sections we will present the current status of research on ERV envelope genes that have been integrated into the placenta of different species, and their potential function in reproductive biology.
-
The human placenta shows the highest levels of human endogenous retroviruses(HERVs)among all normal tissues(Tolosa et al., 2012). The env genes of the HERVs HERV-W(ERVWE1), HERV-FRD, and ERV-3 were found to be abundantly expressed in the human placenta(Venables et al., 1995; Blond et al., 2000; de Parseval et al., 2003; Black et al., 2010; Tolosa et al., 2012). In 1999, Blond et al. first reported that the ERVWE1 gene was expressed in syncytiotrophoblast cells, which was subsequently given the name syncytin-1 by Mi et al. in 2000(Blond et al., 1999; Mi et al., 2000). Syncytin-1 is an ERVWE1 envelope gene located on human chromosome 7. ERVWE1 belongs to the HERV-W lineage that is estimated to have invaded the genomes of higher primates about 25 to 40 million years ago based on phylogenetic analyses(Voisset et al., 1999; Blond et al., 2000). The syncytiotrophoblast of the mammalian placenta is a true syncytium, derived from the fusion between progenitor cytotrophoblasts cells. The syncytiotrophoblast acts as an epithelial barrier that makes direct contact with maternal blood and transports key nutrients and other solutes to the fetus for its development(Omata et al., 2013).
Syncytin-1 was recently demonstrated to be a highly fusogenic membrane glycoprotein with characteristics of retroviral Env protein(Figure 1) (Bjerregaard et al., 2014; Bjerregard et al., 2014; Huang et al., 2014a; Huang et al., 2014b). Syncytin-1 induces the formation of syncytia in the interaction with sodium-dependent neutral amino acid transporter 2(ASCT2)(Frendo et al., 2003; Tolosa et al., 2012). A large number of recent in vitro studies have indicated that syncytial-1 is involved in the cellular fusion of human trophoblast cells, which may play a key role in the formation of human placenta(Mi et al., 2000; Tolosa et al., 2012). Transfection of syncytin-1 of multiple cell lineages has been shown to induce cellular fusion; in addition, cells treated with an antibody against the syncytin-1 protein resulted in a significant decrease in cell fusion(Blond et al., 1999; Mi et al., 2000; Frendo et al., 2003; Black et al., 2010). Moreover, direct inhibition of the expression of syncytin-1 inhibited the fusion of trophoblastic cells(Frendo et al., 2003). Other studies have shown that the expression of syncytin-1 mRNA in pathological placentas is very low(Huang et al., 2014b; Yu et al., 2014; Zhou et al., 2014; Zhuang et al., 2014; Machnik et al., 2015; Tolosa et al., 2015).

Figure 1. Schematic diagram of the functional domains of Env glycoproteins proposed to be involved in placental morphogenesis. SU, surface subunit; TM, transmembrane subunit; SP, signal peptide; RBD, receptor binding domain; FP, fusion peptide; ISD, immunosuppressive domain; MSR, membrane-spanning region; CT, cytoplasmic tail; gray and blue color parts, linker regions.
Syncytin-2 is the envelope gene of HERV-FRD, and is also abundantly expressed in the human placenta(Esnault et al., 2008; Cornelis et al., 2015; Nakaya and Miyazawa, 2015; Toufaily et al., 2015). Similar to syncytin-1, transient transfection of syncytin-2 induced cell fusion in a variety of cell lines(Knerr et al., 2002; Black et al., 2010). The interaction of syncytin-2 and the major facilitator superfamily domain-containing 2 receptor promoted the cell fusion of human trophoblast cells(Machnik et al., 2015). Syncytin-2 is similar to syncytin-1 in structure, but they have different immunosuppressive properties(Figure 1) (Mangeney et al., 2007; Black et al., 2010).
ERV-3 is also expressed in trophoblast cells and syncytiotrophoblasts, but a premature stop codon in its long open reading frame has resulted in loss of the transmembrane region and other functional areas(Cohen et al., 1985; Boyd et al., 1993). The Env protein of ERV-3 does not elicit cell fusion(Lin et al., 1999). However, ERV-3 is stably expressed in trophoblast cells, and was shown to induce trophoblast differentiation and increase the levels of chorionic gonadotropin(Harris, 1998). Furthermore, ERV3 Env is also expressed in many normal tissues, especially in hormone-producing organs, including the adrenal and sebaceous glands and testis(Harris, 1998; Black et al., 2010). These findings suggest that it may play a critical role in the process of hormone secretion.
-
In 2005, Dupressoir et al.(2005) conducted a systematic in silico search of the entire mouse genome database and identified two fully coding envelope genes that were unique and independent of any known murine endogenous retrovirus, which were named syncytin-A and -B(Figure 1). Syncytin-A and -B are orthologous genes whose expression is conserved in all muridae tested(mouse, rat, gerbil, vole, and hamster), and are estimated to have entered the rodent lineage at least 40 million years ago(Dupressoir et al., 2005; Black et al., 2010; Vernochet et al., 2014; Cornelis et al., 2015). Quantitative reverse transcription-polymerase chain reaction analyses con firmed that the two genes are specifically expressed in the placenta, and in situ hybridization of placenta cryosections revealed that the two genes are specifically expressed in trophoblast cells and the syncytia layer(Dupressoir et al., 2005). In vitro transfection experiments showed that both genes can induce cell–cell fusion, but that they use different receptors(Dupressoir et al., 2005). Another study by the same group showed that homozygous syncytin-A null mouse embryos of syncytin-A knock-out mice died in utero between 11.5 and 13.5 days of gestation(Dupressoir et al., 2009). In another experiment, use of a specific antibody and antisense oligonucleotides to inhibit the expression of syncytin-A, caused marked reduction in the formation of syncytiotrophoblast cells(Gong et al., 2007). In addition, trophoblast cells cultured with syncytin-A in vitro promoted syncytiotrophoblast cell development within 24 h, strongly suggesting that syncytin-A is a fusogenic protein that plays an important regulatory role(Henke et al., 2013). These results indicate that syncytin-A is essential to the process of rodent trophoblast cell differentiation and syncytiotrophoblast formation. Both syncytin-A and -B contain a recognized immunosuppressive region, leading some researchers to suggest that they may also play a role in maternal-fetal tolerance, although there is no strong confirmatory evidence(Black et al., 2010). In 2011, Dupressoir et al. knocked out the syncytin-B gene in the mouse genome, and found that these syncytial-B-deficient mice could complete a pregnancy, but fetal growth was retarded and the number of newborns was reduced compared to wild-type mice(Dupressoir et al., 2011).
-
In 2009, Heidmann et al. conducted a systematic in silico search of full-length env genes with an uninterrupted open reading frame within the rabbit genome, and found an env gene of retroviral origin with the characteristic features of syncytin, which was named syncytin-Ory1(Figure 1). They further confirmed that the syncytin-Ory1 gene was specifically expressed in and had entered the Lagomorpha order approximately 12–30 million years ago(Heidmann et al., 2009). The results of in situ hybridization showed that syncytin-Ory1 mRNA was specifically expressed in the rabbit placenta junctional zone, where fetal tissue invades the maternal uterus decidua to form the labyrinth trophoblast. Surprisingly, the Syncytin-Ory1 receptor was found to be ASCT2, the same receptor for syncytin-1, as mentioned above(Black et al., 2010). These results demonstrated that syncytin-Ory1 likely plays an important role in formation of the rabbit placenta(Heidmann et al., 2009; Black et al., 2010).
-
Recent studies have shown that the ERV env gene is expressed in the bovine placenta(Nakagawa et al., 2013). bERVE-A possesses a sequence that is similar to the sequence of human syncytin-1, and is specifically expressed in the bovine placental trophoblast, but does not have the entire open reading frame(Henzy and Coffin, 2013; Tolosa et al., 2015). In order to determine the derived function of ERV env genes in bovine placenta development, Nakagawa et al.(2013) used a high-throughput sequencer to examine the transcription status of ENV derived env genes during different cattle pregnancy implantation periods. The results showed that about 18% of the total expression of the ERV env gene was expressed in the process of bovine placenta formation(Nakagawa et al., 2013). Finally, they succeeded in finding three env-derived genes with a long open reading frame that were highly expressed in trophoblast cell lines. However, the biological function of these ERV genes in the bovine placenta has not yet been reported. To date, only the env genes of the bovine ERVs BERV-K1 and BERV-K2 have been well studied, which were found to be abundantly expressed in the bovine placenta and bovine trophoblast cell lines cultivated in vitro(Baba et al., 2011; Koshi et al., 2012; Nakaya et al., 2013). Here, we focus on the BERV-K1 envelope glycoprotein Fematrin-1, which is specifically expressed in bovine trophoblast cells. Fematrin-1 was shown to induce cell fusion of both trophoblast and bovine endometrial cells in vitro(Nakaya et al., 2013). Fematrin-1 was found to have integrated into intron 18 of bovine FAT tumor suppressor homolog 2(FAT2)approximately 18.3–25.4 million years ago(Nakaya et al., 2013; Tolosa et al., 2015).
-
The domestic sheep genome includes at least 27 copies of endogenous beta-retroviruses, which are closely related to the exogenous and pathogenic JSRV, and were thus named enJSRVs(Palmarini et al., 2004; Arnaud et al., 2007; Arnaud et al., 2010; Black et al., 2010; Armezzani et al., 2011). JSRV is the causative agent of ovine pulmonary adenocarcinoma, an infectious lung tumor disease in sheep(Hofacre and Fan, 2010). JSRV is distinct from other oncogenic retroviruses, because its envelope protein can induce cell carcinogenesis both in vivo and vitro(Maeda et al., ; Allen et al., 2002; Danilkovitch-Miagkova et al., 2003; Liu and Miller, 2005; Wootton et al., 2005; Hofacre and Fan, 2010; Zhang et al., 2014). However, the mechanism of JSRV Env-induced cell transformation is not entirely clear. Interestingly, many enJSRVs encode functional viral proteins, and many enJSRVs Env glycoproteins are quite similar to JSRV Env glycoprotein(Black et al., 2010; Spencer and Palmarini, 2012). However, enJSRVs Env glycoprotein(Figure 1)does not cause cancer and it does not induce the transformation of cells(Hull et al., 2012; Armezzani et al., 2014).
Interestingly, enJSRV env and hyaluronoglucosaminidase 2(HYAL2), a cellular receptor for both JSRV and enJSRVs Env, are abundantly expressed in the trophoblast giant binucleate cells(BNCs) and multinucleated plaques of the syncytiotrophoblast within the placentomes(Figure 2)during pregnancy(Wooding, 1984; Wooding, 1992; Guillomot, 1995; Roberts et al., 1999; Spencer et al., 2004; Dunlap et al., 2005; Black et al., 2010; Spencer and Palmarini, 2012; Qi et al., 2012). These findings have piqued interest in enJSRV env, given that it may play an important physiological role in conceptus elongation and the onset of the differentiation of trophoblast giant BNCs. In one study, morpholino was injected into the ewe uterus on day 8 of pregnancy, which inhibited the translation of enJSRV envelope protein(Dunlap et al., 2006; Black et al., 2010; Spencer and Palmarini, 2012). The results showed that through the injection of morpholino in ewes at 16 days of gestation, the growth of trophectoderm cells was inhibited and the number of trophoblast giant BNCs was reduced. These results led to the hypothesis that en-JSRVs and its receptor HYAL2 promote the growth and differentiation of sheep placental syncytium trophoblastic cells(Spencer et al., 2007; Black et al., 2010; Spencer and Palmarini, 2012).
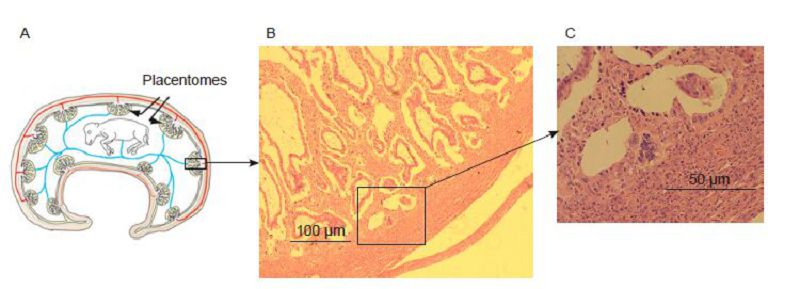
Figure 2. Structure of the synepitheliochorial placenta of sheep. (A) Schematic drawing of the sheep fetus and placenta in utero. (B, C) Hematoxylin-and-eosin stained placenta sections of sheep.
However, Cornelis et al.(2013) identified an envelope gene from the genome of sheep and cattle that shows specific expression in the Ovis aries and the Bos taurus placenta, which was named syncytin-Rum1(Figure 1). Syncytin-Rum1 is estimated to have integrated into the genome of ruminants approximately 30 Mya. The syncytin-Rum1 gene exhibits all of the typical characteristics of a syncytin gene: (ⅰ) it is specifically expressed in the placenta; and (ⅱ) it exhibits fusogenic activity by mediating cell–cell fusion (Cornelis et al., 2013). This discovery led to speculation about the veracity of the hypothesis on the effect of enJSRVs and HYAL2 on the growth and differentiation of sheep placental syncytium trophoblastic cells, because enJSRV Env is expressed in the placentabut not specifically, and there is no conclusive evidence that it shows cell–cell fusion activity. Therefore, further research on the enJSRV Env protein function and its function in reproductive biology is required. Our research group has cultured sheep trophoblast cells and cloned the enJSRV env gene. We have further established an RNA interference plasmid of targeted enJSRV env and measured its interference efficiency (Wang et al., 2014). We are next planning to up-regulate and inhibit the expression of enJSRV env gene in sheep trophoblast cells in vitro to evaluate the enJSRV Env protein function and its effect on reproductive biology. These results should help to verify the authenticity of the hypothesis.
-
The findings described above clearly demonstrate that capturing of ERV-derived genes has repeatedly occurred throughout the evolution of placental mammals, suggesting that new syncytin genes will be found in the syncytiotrophoblast organization of other placental mammals(Heidmann et al., 2009). Indeed, recent investigations in other animals such as Marmota(woodchuck) and Carnivora(dogs and cats)strongly support this hypothesis. Cornelis et al.(2012) searched for full-length coding envelope protein genes within the dog and cat genomes, and found a common gene in both species named syncytin-Car1 that displayed placenta-specific expression(Figure 1). The syncytin-Car1 gene was cloned into a cytomegalovirus-driven expression vector, and a transient transfection experiment showed that the gene displays characteristic cell fusion activity. This gene is the oldest syncytin gene identified to date, and is estimated to have integrated into the carnivorous animal genome between approximately 60 and 85 Mya(Cornelis et al., 2012). Recently, Redelsperger et al.(2014) discovered an ERV envelope gene with all the characteristic features of a syncytin. This gene is specifically expressed in the woodchuck placenta and was named syncytin-Mar1, and was shown to induce cell-cell fusion in vivo, indicating an essential role in its host's physiology. Syncytin-Mar1(Figure 1)has gone through about 25 Mya of evolution, but is not related to any of the other syncytins identified to date. In addition, Cornelis et al.(2015) also found a similar syncytin gene in the placenta of marsupials, which was named syncytin-Opol. Syncytin-Opo1 could also induce cell fusion in vitro, but does not show high expression in the placenta of marsupials.
-
Most scientists believe that the placenta evolved into an environment where retroviral genes were 'tamed' to serve functions that benefit the host, including the manipulation of maternal physiology for protecting the fetus (Haig, 2012). Chuong (Chuong, 2013) suggested that ERV-mediated regulatory evolution is, and will continue to be, an important mechanism of the evolution of placental development. The evolutionary interplay between retroviruses and host defense may contribute to the significant differences observed among mammalian placentas. Many recent studies have focused on the capture of ERVderived genes for specific functional adaptations in the placenta. Syncytin genes within the mammalian placenta(e.g., syncytin-1 and syncytin-2 in humans, syncytin-A and syncytin-B in Muroidea, syncytin-Ory1 in Leporidae, syncytin-Car1 in Carnivora, and the syncytin-Rum1 in ruminants)illustrate this point nicely. However, the exact mechanism by which the ERV env genes promote placenta differentiation and trophoblast cell fusion remains unclear. In addition, as an important protein for sheep placenta development, the specific function of enJSRV Env is also unclear and requires much more research effort. Overall, studies with enJSRVs and JSRV as well as other ERVs expressed in the placenta have helped us to comprehend how ERVs evolved from infectious elements to essential genes.
-
This report was funded by the National Natural Science Foundation of China(31360597 and 31160493), the doctoral fund project of Ministry of Education(20111515110008) and Inner Mongolia technology application research project(20130224).
-
The authors declare that they have no conflict of interinterest. This article does not contain any studies with human or animal subjects performed by any of the authors.














 DownLoad:
DownLoad: