-
Dear Editor,
The pandemic of COVID-19, a disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2; previously known as 2019-nCoV), has placed an enormous burden on health authorities in 213 countries and territories. At the time of writing, over 26 million cases have been recorded across the world, with more than 870 k associated deaths (www.worldometers.info/coronavirus/). World Bank warns COVID-19 pandemic risks dramatic rise in poverty, with the majority of economies expecting to suffer from falling levels of GDP in 2020.
The main symptoms of COVID-19 are onset of a new continuous cough and/or a high temperature with symptoms ranging from mild to life threatening. Those who are elderly or have pre-existing health issues are more likely to develop severe disease. The most prevalent comorbidity is hypertension, followed by diabetes (Guan et al. 2020a, b). We reported recently that hypertension is a risk factor for severe cases of COVID-19, independent of age and other variables (Liu et al. 2020a). An important question is why patients with hypertension and diabetes yield poorer clinical outcomes than those without. Human pathogenic coronavirus SARS-CoV-2 utilizes angiotensin-converting enzyme 2 (ACE2) as a receptor for viral cell entry. Since the levels of ACE2 are substantially increased in patients with hypertension or diabetes, who are treated with ACE inhibitors (ACEIs) and angiotensin Ⅱ type-Ⅰ receptor blockers (ARBs) (Ferrario et al. 2005), Fang and colleagues hypothesized that ACE2-stimulating drugs could potentially increase the risk of developing severe COVID-19 (Fang et al. 2020). This was not supported by a recent study led by Dr. Reynolds (Reynolds et al. 2020), whose analysis showed no positive association for ACEIs or ARBs for either the risk of SARS-CoV-2 infection or severe illness (Reynolds et al. 2020). What else might explain the poorer clinical outcomes of COVID-19 patients with hypertension or diabetes?
To explore this question, we re-analysed the same cohort of 99 COVID-19 patients discharged from the general wards of Renmin Hospital of Wuhan University between 5 February 2020 and 14 March 2020 (Ethics approval No: WDRY2020-K124) (Liu et al. 2020a, b). Demographic, clinical, laboratory and radiographic findings were extracted from electronic medical records. We asked whether there were any factors associated with COVID-19 patients with either hypertension or diabetes (summarised in Table 1). All data analyses and graphs were done in R (V3.6.1) or GraphPad Prism (V8.2.1).
P value, median (ICQ) Diabetes Hypertension −
(n = 84)+
(n = 15)−
(n = 65)+
(n = 34)Demographics Age 0.2725 0.0030 Sex 0.8578 0.9846 Signs and symptoms Fever 1.0000 1.0000 Cough 0.1877 0.2292 Dyspnea 1.0000 0.7422 Anorexia and/or lethargy 1.0000 0.4944 Fatigue 0.9510 1.0000 Myalgia and/or arthralgia 1.0000 0.6506 Diarrhoea 0.7846 0.6871 Laboratory findings White blood cell count (× 109/L) 0.8224 0.0351 4.87
(3.52–5.69)4.11
(3.23–6.12)4.47
(3.32–5.46)5.50
(3.94–6.65)Neutrophil count (× 109/L) 0.6676 0.0450 2.93
(1.96–4.28)3.25
(2.21–4.67)2.81
(1.73–3.87)3.62
(2.54–5.12)Lymphocyte count (× 109/L) 0.0192 0.2595 1.08
(0.80–1.45)0.83
(0.54–1.16)1.09
(0.77–1.45)0.96
(0.70–1.31)D- dimer (μg/L) 0.0531 0.0166 0.38
(0.24–0.82)0.56
(0.44–1.11)0.36
(0.21–0.80)0.66
(0.41–0.93)Lactate dehydrogenase (U/L) 0.1337 0.0070 191.0
(157.0–242.5)215.0
(195.0–258.0)180.0
(151.5–240.0)222.5
(192.5–263.2)C- reactive protein (mg/dL) 0.9086 0.1421 1.70
(0.52–4.07)2.41
(0.34–4.78)1.37
(0.51–3.24)2.69
(0.53–5.84)Radiographic findings HRCT peak score 0.0119 0.0088 14.5
(8.0–20.3)21.0
(15.5–30.0)14.0
(8.0–20.0)19.0
(14.0–25.8)Data are P values. Medians and IQR (interquartile range) are also provided for laboratory and radiographic findings. P values were calculated by Mann–Whitney U test or Chi squared test as appropriate using R (V3.6.1) or GraphPad Prism (V8.2.1). Numbers in bold mean P values less than 0.05 and are statistically significant. Table 1. Comparisons of demographic, clinical, laboratory and radiographic findings in COVID-19 patients with diabetes or hypertension.
As expected, COVID-19 patients with health conditions like hypertension or diabetes had a longer length of hospital stay than those without, but this was at the limit of statistical significance (Fig. 1A, P = 0.07). To check the extent of pulmonary pathology in COVID-19 patients, we developed a high-resolution computed tomography (HRCT) score system considering both radiographic features and distributions (Liu et al. 2020b; Yuan et al. 2020). In brief, the HRCT findings were graded on a 3-point scale: normal attenuation (1 point), ground-glass attenuation (2 points), and consolidation (3 points). Each lung zone (6 lung zones in total for each patient) was assigned a following scale according to the distribution of the affected lung parenchyma: normal (0 point), < 25% abnormality (1 point), 25%–50% abnormality (2 points), 50%–75% abnormality (3 points), and > 75% abnormality (4 points). The 4-point scale of the lung parenchyma distribution and the radiographic scale described above were then multiplied to produce a weighted score for each zone. Points from all zones were then summed for a final total cumulative score (HRCT score), with values ranging from 0 to 72. HRCT peak scores are the highest HRCT score during disease course, which reflex maximal chest HRCT abnormalities. We found HRCT peak scores for hypertensive COVID-19 patients were higher compared to non-hypertensive ones (median: 19.0; IQR: 14.0–25.8 vs. median: 14; IQR: 8.0–20.0) (P = 0.0088) (Table 1; Fig. 1B). HRCT peak scores in COVID-19 patients with diabetes (median: 21.0; IQR, interquartile range: 15.5–30.0) were also higher compared to those without (median: 14.5; IQR: 8.0–20.3) (P = 0.0119) (Table 1; Fig. 1C). These findings confirmed an increased disease severity in COVID-19 patients with hypertension or diabetes (Liu et al. 2020a).
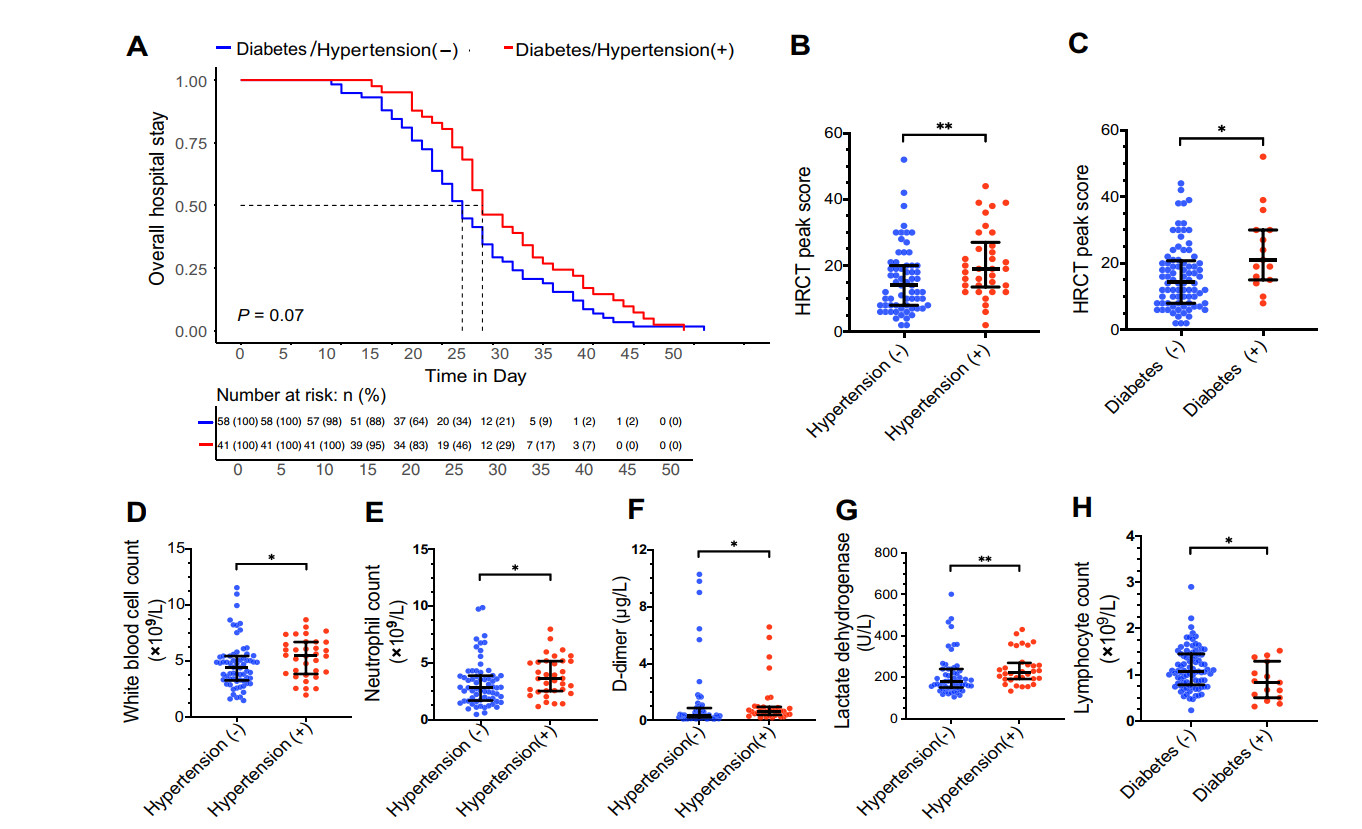
Figure 1. Impacts of hypertension or diabetes on length of hospital stay, HRCT peak scores and laboratory findings in COVID-19 patients. A Kaplan–Meier plot showing the overall hospital stay in COVID-19 patients with hypertension or diabetes and those without. Patients are separated into two groups. The control group is those patients, who are free of diabetes and hypertension (n = 58), and the other group is those with either diabetes or hypertension (n = 41). Numbers below are n (%). P value was calculated by log-rank test. B Graph showing the distributions of HRCT peak scores in COVID-19 patients with or without hypertension. C Graph showing the distributions of HRCT peak scores in COVID-19 patients with or without diabetes. D–G Graphs showing white blood cell counts (D), neutrophil counts (E), D-dimer (F) and lactate dehydrogenase (G) in COVID-19 patients with or without hypertension. H Graph showing lymphocyte counts in COVID-19 patients with or without diabetes. Data in (B–H) are median and IQR. *P < 0.05. **P < 0.01. P values in (B–H) were calculated by Mann–Whitney U test.
Most interestingly, in laboratory findings on admission (Table 1), we found white blood cell counts (Fig. 1D; median: 5.50 vs. 4.47; P = 0.035), neutrophil counts (Fig. 1E; median: 3.62 vs. 2.81; P = 0.045), D-dimer (Fig. 1F; median: 0.66 vs. 0.36; P = 0.017) and lactate dehydrogenase (LDH) (Fig. 1G; median: 222.5 vs. 180.0; P = 0.007) were all increased in hypertensive COVID-19 patients compared to non-hypertensive ones; while lymphocyte count was not significantly changed in hypertensive COVID-19 patients (median: 0.96 vs. 1.09; P = 0.260). In contrast, there was a significant decrease in lymphocyte count in COVID-19 patients with diabetes compared with those without (Fig. 1H; median: 1.08 vs. 0.83; P = 0.019); while changes in white blood cell counts, neutrophil counts, D-dimer and LDH were not significant (P > 0.05) in COVID-19 patients with diabetes. In addition, hypertensive COVID-19 patients were older than non-hypertensive ones (P = 0.003), but this was not the case for diabetic COVID-19 patients (P = 0.273). There were no significant differences in C-reactive protein, sex and common symptoms (such as fever, cough, dyspnoea, fatigue, anorexia and/or lethargy, myalgia and/or arthralgia and diarrhoea) for both hypertension and diabetes.
SARS-CoV-2 infections lead to a fast activation of innate immune cells, especially in COVID-19 patients developing severe disease (Zou et al. 2020). It was reported that circulating neutrophil numbers are consistently higher in survivors of COVID-19 than in non-survivors, and the infection also induces lymphopenia (low level of lymphocytes in the blood) (Zhou et al. 2020). In our analysis, we found on admission hypertensive COVID-19 patients had higher neutrophil numbers compared to non-hypertensive ones, while lymphopenia was more frequently observed in COVID-19 patients with diabetes. These results suggest different mechanisms exist for hypertension or diabetes as risk factors for severe cases of COVID-19. It is known that both hypertensive and diabetes patients have impaired immune response to infections (Lopez Gelston and Mitchell 2017; Zhu et al. 2020). Low-grade inflammation is known to facilitate the development of hypertension, and poor glycaemic control influences cytokine profile and T cell activation, which were reflected by the laboratory findings in this study. However, more lab work is required to confirm these in COVID-19. Established mouse models of hypertension and diabetes can be applied to the K18-hACE2 transgenic mice for this purpose.
There are several limitations in this study. (1) Interpretation of our findings might be limited by the sample size. (2) This study is limited by its retrospective nature, the non-standardised documentation resulting in the inability to remove the influence of age on laboratory findings. Despite these limitations, we were able to identify different laboratory abnormalities on admission in COVID-19 patients with hypertension or diabetes, which might shed light on future mechanistic studies.
HTML
-
We acknowledge all the patients involved in this study, and appreciate all the frontline medical and nursing staff involved in the diagnosis and treatment of patients in Wuhan. This project was jointly funded by Medical Research Council (UK) [MR/S025480/1] and a Key Project of Science and Technology on COVID-19 of Hubei Province [No. 2020FCA002]. YZ was supported by an Institute for Life Sciences (University of Southampton) PhD Studentship.
-
The authors declare that they have no conflict of interest.
-
All methods were carried out in accordance with relevant guidelines and regulations. This retrospective study was approved by the Ethics Committee of Renmin Hospital of Wuhan University, Hubei, China (No. WDRY2020-K124), and the requirement for informed consent was waived by the Ethics Committee due to a public health outbreak investigation.














 DownLoad:
DownLoad: