HTML
-
The first reports of coronavirus disease 2019 (COVID-19) occurred in China on 31st December 2019 (Fanelli and Piazza 2020; Lu et al. 2020b; Remuzzi and Remuzzi 2020; Wang C et al. 2020; Zhu et al. 2020); globally, as of 23 November, 2020, 58, 425, 681 cases of COVID-19 have been confirmed, including 1, 385, 218 deaths, as reported to the World Health Organisation (WHO) (https://covid19.who.int/). The COVID-19 pandemic has had serious impacts around the world (Alwan et al. 2020; Paoli et al. 2020; Remuzzi and Remuzzi 2020; The Lancet 2020; The Lancet Infectious Diseases 2020). Convenient and reliable COVID-19 detection assays and equipment are urgently needed for efficient management of this pandemic. With effective prevention and control strategies and sufficient detection kits, China won a significant early victory against COVID-19, and is now focused on preventing the transmission of imported COVID-19 (Li Z et al. 2020; Wang J et al. 2020). Notably, in the early stage, nucleic acid detection kits for SARS-CoV and other coronaviruses were used for COVID-19 diagnosis, with varying specificity and sensitivity (Zhou et al. 2020; Zhu et al. 2020). Meanwhile, when detection kits were not available, imaging examinations, especially chest computed tomography (CT), played an important role in the diagnosis of COVID-19 (Zhou et al. 2020; Zhu et al. 2020), although these examinations are not specific to COVID-19. Soon after the genome of the pathogen underlying COVID-19, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), previously named as 2019 novel coronavirus (2019-nCoV), was identified (Lu et al. 2020b; Zhou et al. 2020) Novel quantitative real-time reverse-transcription polymerase chain reaction (rRT-PCR) assays were rapidly developed in response to the emerging pandemic of COVID-19 (Niu et al. 2020), described in national technical guidelines for China (Center for Disease Control and Prevention 2020), and shared globally. Since then, these assays have been widely adopted for laboratory detection (Chen et al. 2020; Pan et al. 2020; Wang W et al. 2020). Subsequently, multiple detection kits for SARS-CoV-2 have been developed and utilised in the battle against COVID-19. As of November 20, 2020, a total of 51 detection kits for SARS-CoV-2 nucleic acids (24 kits), antibodies (25 kits) and antigens (2 kits) have been approved by the National Medical Products Administration of China (NMPA), some of which have been registered and employed worldwide.
Currently, commercially available COVID-19 detection kits approved for use in China can be divided into three categories. The first category includes molecular assays of SARS-CoV-2 RNA based on real-time reverse transcription polymerase chain reaction (rRT-PCR) techniques, isothermal amplification and genome sequencing. The second category includes serological assays that detect antibodies produced by individuals exposed to SARS-CoV-2 based on lateral flow immunoassay (LFIA), chemiluminescence immunoassay (CLIA) or enzyme-linked immunosorbent assay (ELISA). The third category includes LFIA-based antigen detection kits. These three categories of detection kits play complementary roles in the management of the COVID-19 pandemic. SARS-CoV-2 viral RNA and antigens can be identified in SARS-CoV-2-infected individuals during the acute phase of infection, while serological tests subsequently identify individuals who have developed antibodies to the virus, which can be used for close contact tracing and monitoring of the immune status of individuals and groups over time (Arun Krishnan et al. 2020; Hu et al. 2020; James and Alwneh 2020; Meo et al. 2020; Oliveira et al. 2020; Priyadarshi et al. 2020; Shen et al. 2020; Wang H et al. 2020; Xu T et al. 2020).
-
Currently, the most widely approved tests for the diagnosis of COVID-19 are based on PCR (Arun Krishnan et al. 2020; Hu et al. 2020; James and Alwneh 2020; Meo et al. 2020; Oliveira et al. 2020; Priyadarshi et al. 2020; Shen et al. 2020; Wang H et al. 2020; Xu T et al. 2020; Xu Y et al. 2020). Two different strategies are used for PCR-based assays: rRT-PCR and isothermal amplification PCR. By amplifying the viral genetic material, rRT-PCR achieves high sensitivity and specificity for COVID-19 diagnosis, and is quantitative in nature, whereas isothermal amplification PCR is qualitative (Arun Krishnan et al. 2020; Li and Ren 2020; Shen et al. 2020). Compared to rRT-PCR, the latter method is expected to be more cost effective and less time consuming. However, the accuracy of isothermal amplification remains to be determined, which has prevented its widespread adoption. Although rRT-PCR was the first RNA detection method used for SARS-CoV-2, improper sample collection, handling and transportation may lead to false negative results, markedly reducing the sensitivity of the assay.
-
After infecting a human, SARS-CoV-2 amplifies quickly. Therefore, nucleic acids of the virus can be detected from the early stage in samples such as nasopharynx swabs, oropharynx swabs, sputum, and stool (Li and Ren 2020; Paoli et al. 2020; Petrillo et al. 2020; Yan et al. 2020). Among all approved nucleic acid detection kits, the novel rRT-PCR techniques were developed in rapid response to the emergence of COVID-19 in China. The rRT-PCR reaction system includes a pair of specific primers and a TaqMan probe for the target nucleic acid. The probe is a specific oligonucleotide fragment complementary to a template sequence, and both ends of the probe are labelled with a reporter fluorescence group and fluorescence quenching group. The complete probe aligns perfectly with the template during PCR and the fluorescence signal emitted by the reporter group is absorbed by the quenching group; when PCR is performed and the target gene is amplified in the reaction system, DNA polymerase exerts its exonuclease activity, cleaving and degrading the probe, and then the reporting group and the quenched group separate, allowing fluorescence to be emitted. Each time a DNA strand is amplified, a fluorescent molecule is produced. The number of the threshold cycle (Ct value) is detected when the fluorescence generated reaches a fluorescence or signal threshold. The Ct value is related to the concentration of viral nucleic acid in the specimens tested, with higher concentrations of viral nucleic acid associated with smaller Ct values. The Ct value and the logarithm of the copy number of nucleic acids in the specimens have a linear relationship (Niu et al. 2020; Xu T et al. 2020). Initially, three gene targets, namely open reading frame 1ab (ORF1ab), nucleoprotein (N) and envelope (E), were evaluated via rRT-PCR to achieve high specificity and sensitivity (Fig. 1A). The results showed that the ORF1ab and N gene-based assays were specific, exactly matching the target genes in SARS-CoV-2. However, E gene-based rRT-PCR showed cross-reactivity with other beta-coronaviruses, such as SARS-CoV. Therefore, E gene-based rRT-PCR was suggested as a universal screening tool for B lineages of betacoronavirus, including SARS-CoV, SARS-CoV-2, bat SARS-like coronavirus, and others (Niu et al. 2020). To avoid false negatives resulting from gene mutation, two gene targets of ORF1ab and N were adopted. Furthermore, ORF1ab and N gene-based rRT-PCR were confirmed to be the standards recommended and described in the Technical Guidelines for COVID-19 Laboratory Testing in China (Center for Disease Control and Prevention 2020), which have been widely applied. Although each manufacturer determines the criteria used by their product, most amplification plots of rRT-PCR kits approved by NMPA target ORF1ab and N (Table 1). Laboratory confirmation of positive cases requires either positive RT-PCR results for both ORF1ab and N in the same specimen, a positive RT-PCR result for only one target (ORF1ab or N) in two types of specimens, or two positive results from the same type of sample. Fortunately, the detection time could be shortened greatly by combining the detection kits with a special fluorescent PCR device, for example in the detection tests from Coyote Bioscience Co., Ltd., Sansure Biotech Inc., and Daan Gene Co., Ltd. of Sun Yat-Sen University (Table 1). Notably, negative nucleic acid detection results cannot rule out SARS-CoV-2 infection. Therefore, interference factors should be considered carefully (Chinese Center for Disease Control and Prevention 2020).
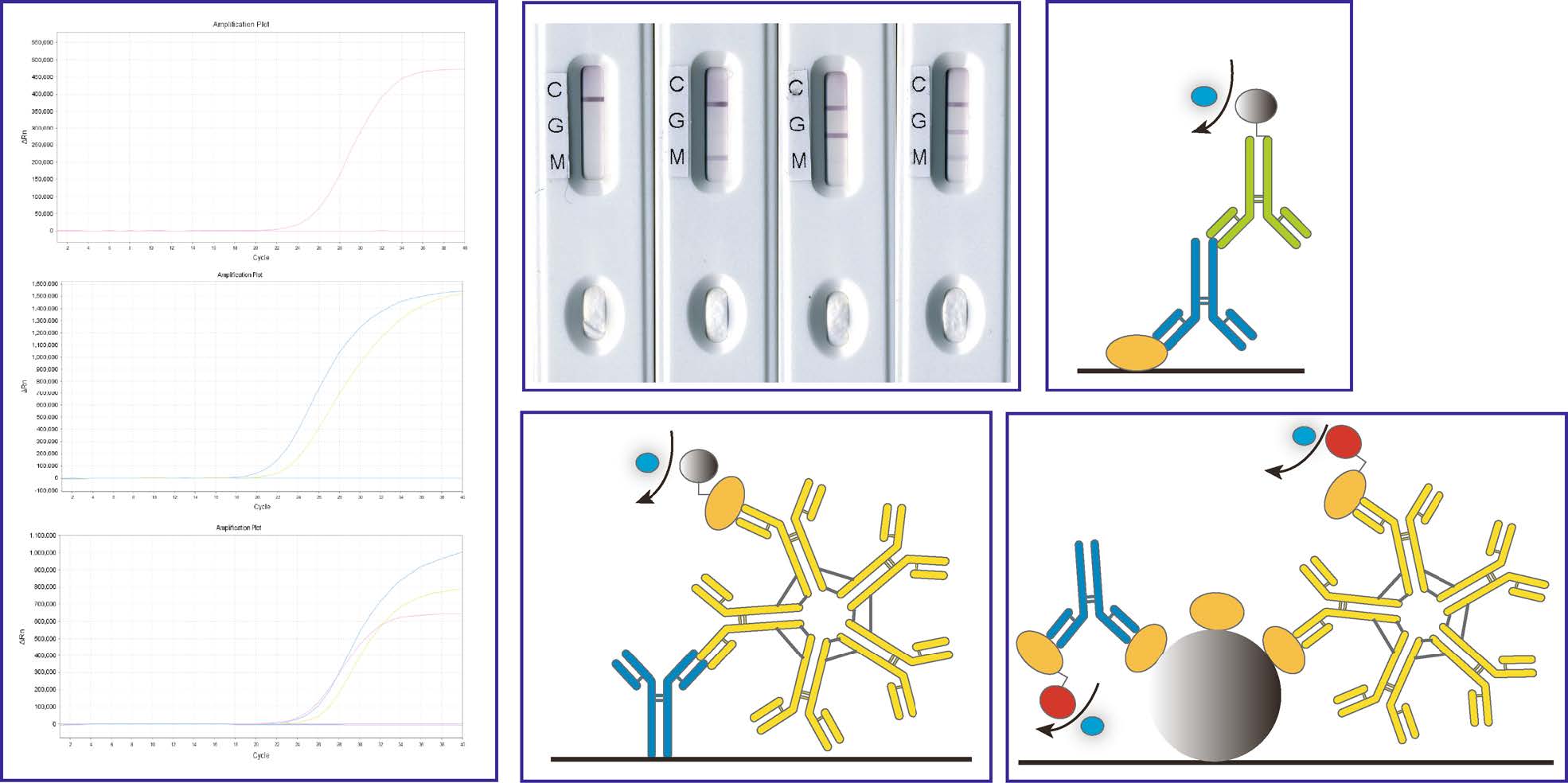
Figure 1. Representative detection methods developed for SARS-CoV-2. A Representative amplification plots of approved SARS-CoV-2 rRT-PCR kits targeting one (top), two (middle) or three (bottom) regions of the 2019-CoV genome. B Typical results of LFIA approved for 2019-nCoV antibody detection in China. Four lanes show negative, IgM positive, IgG positive, and both IgM and IgG positive results, respectively. C–E Schematic diagram of indirect ELISA for IgG detection; IgM-capture ELISA for IgM detection, and CLIA based on magnetic particle detection for all antibodies, including IgM and IgG.
No. Manufacture, organization name Test name Test type Gene or region detected Sample source Limits of detection Test result time/additional information Throughput information Country of approval 1 Shanghai ZJ Bio-Tech Co., Ltd. Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real-time RT-PCR ORF1ab, E and N gene Throat swab, sputum, and BALF 1000 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400057) 1/26/2020, WHO, CE 2 Shanghai GeneoDX Biotech Co., LTD Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real-time RT-PCR ORF1ab and N gene Nasopharyngeal swab and BALF 500 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400058) 1/26/2020 3 BGI Biotechnology (Wuhan) CO., LTD Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real-time RT-PCR ORF1ab Throat swab and Bronchoalveolar Lavage Fluid (BALF) samples 100 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400060) 1/26/2020/CE Marked/FDA Authorized/PMDA Approved 4 Daan Gene Co., Ltd. of Sun Yat-Sen University 2019 Novel Coronavirus (2019-nCOV) RNA Detection Kit Real-time RT-PCR ORF1ab and N gene Throat swab, sputum, nasopharyngeal swab 500 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400063) 1/28/2020, CE 5 Sansure Biotech Inc. Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-Fluorescence probing) Real-time RT-PCR ORF1ab and N gene Throat swab and Bronchoalveolar Lavage Fluid (BALF) samples 200 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400064) 1/28/2020, CE, FDA EUA 6 Shanghai BioGerm Medical Biotechnology Co., Ltd. Novel Coronavirus (2019-nCoV) Nucleic Acid Detection Kit Real-time RT-PCR ORF1ab and N gene Oropharyngeal, nasopharyngeal, and sputum 500 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400065) 1/31/2020 7 Beijing Applied Biological Technologies Co., Ltd. Multiple Real-time PCR kit for Detection of 2019-nCoV Real-time RT-PCR ORF1ab, E and N gene Sputum and throat swab 200 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400179) 2/27/2020, CE 8 Maccura Biotechnology Co., Ltd. SARS-CoV-2 Fluorescent PCR Kit (for the COVID-19 Coronavirus) Real-time RT-PCR ORF1ab, E and N gene Throat swab and Sputum 1000 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400184) 3/1/2020 9 Wuhan EasyDiagnosis Biomedicine Co. Ltd COVID-19 (SARS-CoV-2) Nucleic Acid test Kit Real-time RT-PCR ORF1ab and N gene Oropharyngeal, nasopharyngeal, and sputum 137 copies/mL Results in ~ 3 h after extraction Depended on the real-time instrument China NMPA (20203400212) 3/12/2020, CE, TGA, Brazil, South Africa, South East Asia 10 Shanghai Fosun Long March Medical Science Co., Ltd. Novel Coronavirus (2019-nCoV) RT-PCR Detection Kit Real-time RT-PCR ORF1ab, E and N gene Throat swab and Sputum - Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400299) 3/24/2020 11 Beijng Kinghawk Pharmaceutical Co., Ltd. Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real time RT-PCR ORF1ab and N gene Throat swab and Sputum 500 copies/mL Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400322) 4/3/2020 12 Jiangsu Bioperfectus Technologies Co., Ltd COVID-19 Coronavirus Real Time PCR Kit Real time RT-PCR ORF1ab and N gene Oropharyngeal, nasopharyngeal, and sputum - Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400384) 4/16/2020 13 Zhejiang Oriental genetic biological products Co., Ltd Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real time RT-PCR ORF1ab and N gene Throat swab and Sputum 500 copies/mL Results in 50-75 min Depended on the real-time instrument China NMPA (20203400520) 5/21/2020, PEUA 14 Shenzhen United Medical Science and Technology Co., Ltd. Real Time PCR Kit for Novel Coronavirus 2019-nCoV (ORF1ab, N) Real time RT-PCR ORF1ab gene Throat swab and Sputum 200 copies/mL Results in ~ 1.5 h Depended on the real-time instrument China NMPA (20203400535) 6/5/2020 15 Beijing NaGene Diagnosis Reagent Co., Ltd Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-fluorescence probing) Real time RT-PCR ORF1ab and N gene Oropharyngeal swab and sputum specimens - Results in ~ 2 h after extraction Depended on the real-time instrument China NMPA (20203400537) 6/9/2020 16 Coyote Bioscience Co., Ltd. DirectDetect™COVID-19 Detection Kit Real time RT-PCR ORF1ab and N gene Oropharyngeal swab and sputum specimens 400 copies/mL Results in 30 min ~ 90 min with the instrument of Mini8 plus 4 samples/run China NMPA (20203400644) 7/13/2020, CE, Mexico, Columbia, Indonesia, the Philippines, Aaudi Arabia, Australia 17 Daan Gene Co., Ltd. of Sun Yat-Sen University Detection Kit for SARS-CoV-2 RNA (Fast PCR-Fluorescence Probing) Real time RT-PCR ORF1ab and N gene Throat swab or sputum 500 copies/mL Results in 50-62 min, with the instrument of AGS4800 8 samples/run China NMPA (20203400749) 9/21/2020, CE 18 BGI Biotechnology (Wuhan) CO., LTD Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (Combined probe anchored polymerization sequencing) Sequencing SARS-CoV-2 Throat swab and Bronchoalveolar Lavage Fluid (BALF) samples 100 copies/mL Gene sequencer DNBSEQ-T7, and Automatic sample loading instrument MGIDL-T7 - China NMPA (20203400059) 1/16/2020/CE Marked/FDA Authorized/PMDA Approved 19 Chengdu CapitalBio Jingxin Biotechnology Co., Ltd. Nucleic Acid Detection Kits for Six Kinds of Respiratory Virus (isothermal amplification based on chip) Isothermal Amplification based on Disk Chip SARS-CoV-2 S and N gene, Influenza A, new Influenza A H1N1 Virus (2009) Influenza A H3N2, Influenza B, RSV Throat swab - - - China NMPA (20203400178) 2/22/2020 20 Ustar Biotechnologies (Hangzhou), Ltd. EasyNAT Diagnostic Kit for 2019-nCoV RNA (Isothermal Amplification Real Time Florescence Assasy) Isothermal Amplification-rt-PCR ORF1ab and N gene Throat swab and sputum specimens 1000 copies/mL Results in 79 min 2 samples/run China NMPA (20203400241) 3/16/2020, CE 21 Anbio (Xiamen) Biotechnology Co., Ltd Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (RNA Hybrid Capture-Immunofluorescence Assay) Hybrid capture-immunofluorescence assay ORF1ab, N and E gene Throat swab and sputum specimens 500 copies/mL Results in 45 min 120 samples in 1 h, instantaneous measurement China NMPA (20203400298) 3/24/2020 22 Rendu (Shanghai) Biotechnology Co., Ltd Novel Coronavirus (2019-nCoV) Nucleic Acid Diagnostic Kit (RNA Hybrid Capture-Immunofluorescence Assay) Magnetic based target RNA capturing technology, Isothermal Amplification and real-time Immunofluorescence assay ORF1ab gene Oropharyngeal swab and sputum 250 copies/mL Results in 90 min 80 samples/run China NMPA (20203400300) 3/26/2020 23 Wuhan Zhongzhi Biotechnologies Inc. Novel Coronavirus 2019-nCoV Nucleic Acid Detection Kit (RNA Amplification Lateral Flow Assay) Isothermal Amplification and gold probe-based Chromatography ORF1ab and E gene Throat swab, sputum specimens, nasopharyngeal swab and Bronchoalveolar Lavage Fluid (BALF) samples 1000 copies/mL Results in 1 h 1 sample/run, continuously detected China NMPA (20203400301) 3/31/2020, CE 24 Wuhan Zhongzhi Biotechnologies Inc. Novel Coronavirus 2019-nCoV Nucleic Acid Detection Kit (Dual amplification) Isothermal Amplification based on reverse amplification and T7 RNA polymerase, and chemiluminescence ORF1ab and E gene Throat swab, sputum specimens, nasopharyngeal swab and Bronchoalveolar Lavage Fluid (BALF) samples 100 copies/mL Results in ~ 3 h Highthroughput, 192 samples in 1 run in 3 h China NMPA (20203400302) 3/31/2020 Note: "-" means information was not available Table 1. Molecular diagnostic tests used to detect viral genetic material in SARS-CoV-2 approved by the NMPA.
-
An alternative technology to rRT-PCR is urgently needed to support real-time detection of SARS-CoV-2 (Augustine et al. 2020; Subsoontorn et al. 2020). This technology should serve as a point-of-care testing (POCT) method, providing results in less than 30 min and with low operating costs. IAT amplifies nucleic acids at a constant temperature in a streamlined and exponential manner for detection, and does not require thermocycling like rRT-PCR. Multiple strategies exist for amplifying target genes, including reverse transcriptase and loop-mediated isothermal amplification (RT-LAMP), recombinase polymerase amplification (RPA), helicase-dependant amplification (HDA), strand displacement amplification (SDA), and nucleic acid sequence-based amplification (NASBA) (Obande and Singh 2020; Shen et al. 2020). In these strategies, primers first bind to a template, and then amplification is performed at a constant temperature using a polymerase with strand-displacement activity that separates the strand annealed to the target sequence for detection. Amplified gene products can be detected through photometry, chemiluminescence, immunofluorescence due to hybridisation capture and lateral flow immunoassay (Annamalai et al. 2020; Arun Krishnan et al. 2020; Li and Ren 2020). The precise strategy used in the IAT for detection of SARS-CoV-2 approved by the China NMPA is not available, as it is a business secret, and the efficiency must be further validated. Notably, not all IAT tests have been demonstrated in the POCT context, which requires results in about 30 min (Table 1). Therefore, IAT as a diagnostic tool for COVID-19 is expected to be much faster, particularly at the point of care. However, isothermal amplification tests have recently emerged as potential technologies for use in airports, community clinics and hospitals to identify both symptomatic and asymptomatic individuals rapidly, potentially reducing the spread of COVID-19 (Arun Krishnan et al. 2020; Carter et al. 2020).
-
Most specific pathogens can be detected through PCR, but trace pathogens may not be detected. Metagenomics next-generation sequencing (mNGS) is used to identify pathogens through sequencing of nucleic acid fragments, followed by analysing and comparing biological information and databases with high accuracy. Theoretically, mNGS can amplify most nucleic acid components in clinical samples and thereby obtain sequence information for pathogens in the samples. First, DNA or RNA in the sample is extracted and a library is constructed for analysis by the sequencer. Then, sequencing is performed, and bioinformatics analysis is conducted to obtain the necessary information. By pretreatment and enrichment of the samples, the most likely pathogen can be identified through gene sequencing, which provides essential information for clinical diagnosis. To identify the pathogen driving the outbreak of pneumonia in Wuhan, Hubei Province, in late December, 2019, next-generation sequencing of samples from bronchoalveolar lavage fluid (BALF) and cultured isolates from nine inpatients was conducted, and a new human-infecting betacoronavirus, SARS-CoV-2 was identified (Lu et al. 2020b). In China, a novel coronavirus SARS-CoV-2 nucleic acid detection kit employing the probe anchoring polymerisation sequencing method produced by BGI Biotechnology (Wuhan) CO., LTD was approved on January 26, 2020, which could be used as a supplement to rRT-PCR. mNGS is generally used for genetic mapping rather than diagnostic testing due to its high cost and long detection time. With the reduction of sequencing cost and optimisation of this technology, high-throughput sequencing is expected to be more widely used for laboratory detection.
rRT-PCR
Isothermal Amplification Test (IAT)
Genetic Sequencing
-
"Although rRT-PCR is considered the 'gold standard' for the diagnosis of COVID-19, it has limitations, including a short positive time of SARS-CoV-2 RNA in most infected individuals" (Carinci et al. 2020; Oliveira et al. 2020). The limitations of rRT-PCR increase the importance of serological assays, which can be utilised for a long time after infection. Infection with SARS-CoV-2 can be detected indirectly by measuring patients' antibodies, which appear 1–2 weeks after nucleic acids (Sethuraman et al. 2020). The plaque reduction neutralisation test (PRNT) and pseudovirus particle neutralisation test (ppNT) are not widely available due to their requirements for specific facilities, such as a Biological Safety Level (BSL) 2 or 3 laboratory. Therefore, immunoassays have been developed by companies in China for detection of COVID-19 infection in serum, plasma and whole blood (Table 2). Among these strategies, lateral flow immunoassays (LFIA) based on gold particles, up-converting phosphor, or quantum dot fluorescence, along with CLIA and ELISA, are the most promising approaches.
No. Manufacture, organization name Test name Test type Ab type Sample source Test result time/additional information Country of approval 1 Guangzhou Wondfo Biotech CO., Ltd. Wondfo SARS-CoV-2 Antibody Test (Lateral Flow Method) LFIA based on gold particle IgM/IgG Serum, plasm, and blood Results in 15 min China NMPA (20203400176) 2/22/2020 2 Innovita (Tangshan) Biological Technology Co., Ltd COVID-19 IgM/IgG Antibody Test Kit (colloidal gold method) LFIA based on gold particle IgM/IgG Serum and plasm Results in 15 min China NMPA (20203400177) 2/22/2020 3 Guangdong Hexin Health Technology Co., Ltd COVID-19 IgM Antibody Test Kit (colloidal gold method) LFIA based on gold particle IgM Serum and plasm Results in 15 min China NMPA (20203400199) 3/11/2020 4 Vazyme (Nanjing) Biotech Co., Ltd 2019-nCoV IgG/IgM Detection Kit (Colloidal Gold-Based) LFIA based on gold particle IgM/IgG Serum and plasm Results in 10 min China NMPA (20203400239) 3/13/2020 5 Zhuhai Livzon Diagnostics Inc The Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2) (Lateral Flow) LFIA based on gold particle IgM/IgG Serum, plasm, and blood Results in 15 min China NMPA (20203400240) 3/14/2020, CE mark 6 Shanghai Outdo Biotech Co., Ltd. Novel coronavirus (SARS-CoV-2) antibody (IgM/IgG) test LFIA based on gold particle IgM/IgG Serum, plasm, and blood - China NMPA (20203400367) 4/10/2020 7 Beijing Zinxing Sihuan Biotech Co., Ltd 2019-nCoV antibody IgM test (Colloidal Gold-Based) LFIA based on gold particle IgM Serum and plasm Results in 10-15 min China NMPA (20203400457) 5/8/2020 8 Bioscience (Chongqing) Diagnostic Technology Co., Ltd Diagnostic kit for novel coronavirus (2019-nCoV) IgM antibody (Magnetic particle CLIA) CLIA based on magnetic particle IgM Serum Chemiluminescence immunoassay Axceed 260 China NMPA (20203400182) 2/29/2020 9 Bioscience (Chongqing) Diagnostic Technology Co., Ltd Diagnostic kit for novel coronavirus (2019-nCoV) IgG antibody (Magnetic particle CLIA) CLIA based on magnetic particle IgG Serum Chemiluminescence immunoassay Axceed 260 China NMPA (20203400183) 2/29/2020 10 Xiamen InnodxBiotech Co. Ltd. Diagnostic kit for novel coronavirus (2019-nCoV) IgM/IgG antibody (Magnetic particle CLIA) CLIA based on magnetic particle IgM/IgG Serum and plasm - China NMPA (20203400198) 3/6/2020 11 Dynamiker Biotechnology (Tianjin) Co., Ltd. Diagnostic kit for novel coronavirus (2019-nCoV) IgG antibody (Magnetic particle CLIA) CLIA based on magnetic particle IgG Serum and plasm - China NMPA (20203400365) 4/10/2020 12 Dynamiker Biotechnology (Tianjin) Co., Ltd. Diagnostic kit for novel coronavirus (2019-nCoV) IgM antibody (Magnetic particle CLIA) CLIA based on magnetic particle IgM Serum and plasm - China NMPA (20203400366) 4/10/2020 13 Zhengzhou Autobio Diagnostics Co., Ltd 2019-nCoV IgM CLIA chemiluminescence CLIA based on magnetic particle IgM Serum and plasm AutoLumo A2000Plus China NMPA (20203400494) 5/15/2020 14 Zhengzhou Autobio Diagnostics Co., Ltd 2019-nCoV IgG CLIA chemiluminescence CLIA based on magnetic particle IgG Serum and plasm AutoLumo A2000Plus China NMPA (20203400495) 5/15/2020 15 Maccura Biotechnology Co., Ltd. SARS-CoV-2 IgG (CLIA) CLIA IgG Serum and plasm 20 min, sensitivity: 96.24%; specificity: 98.13%; total coincidence rate: 97.15% China NMPA (20203400496) 5/18/2020 16 Maccura Biotechnology Co., Ltd. SARS-CoV-2 IgM (CLIA) CLIA IgM Serum and plasm 20 min, sensitivity: 86.99%; specificity: 100.00%; total coincidence rate:93.25% China NMPA (20203400497) 5/18/2020 17 Bioscience (Tianjin) Diagnostic Technology Co., Ltd Diagnostic kit for novel coronavirus (2019-nCoV) IgG antibody (CLIA) CLIA IgG Serum Chemiluminescence immunoassay Axceed 260 China NMPA (20203400498) 5/19/2020 18 Bioscience (Tianjin) Diagnostic Technology Co., Ltd Diagnostic kit for novel coronavirus (2019-nCoV) IgM antibody (CLIA) CLIA IgM Serum Chemiluminescence immunoassay Axceed 260 China NMPA (20203400499) 5/19/2020 19 Beijing Hotgen Biotech Co., Ltd. Novel Coronavirus 2019-nCoV Antibody Test (Up-converting Phosphor Immunochromatographic Technology) Up-converting Phosphor Immunochromatographic Technology IgM/IgG Serum and plasm Results in 15-20 min, hand-held UPT, UPT-3A-1200, UPT-3A-1800 China NMPA (20203400523) 5/25/2020 20 Beijng Kinghawk Pharmaceutical Co., Ltd. Novel Coronavirus 2019-nCoV Antibody Test (Quantum dot fluorescence immunochromatography) Quantum dot fluorescence immunochromatography IgM/IgG Serum, plasm, and blood Results in 15-20 min China NMPA (20203400536) 6/9/2020 21 BGI Biotechnology (Beijing) CO., LTD Novel Coronavirus 2019-nCoV Antibody Test (ELISA) ELISA IgM/IgG Serum and plasm Results in 2 h China NMPA (20203400567) 6/17/2020 22 Shenzhen YHLO Biotech Co., Ltd. iFLASH-SARS-CoV-2-IgM CLIA IgM Serum and plasm 300 tests/h with iFLASH-3000 China NMPA (20203400769) 9/27/2020 23 Shenzhen YHLO Biotech Co., Ltd. iFLASH-SARS-CoV-2-IgG CLIA IgG Serum and plasm 300 tests/h with iFLASH-3000 China NMPA (20203400770) 9/27/2020 24 Xiamen Aode Biotechnology Co., Ltd. Novel Coronavirus 2019-nCoV Antibody Test (Rare Earth Materials-based Nanofluorescence immunochromatography) Rare Earth Materials-based Nanofluorescence immunochromatography IgM/IgG Serum Results in 15 min China NMPA (20203400776) 9/29/2020 25 Beijing Zinxing Sihuan Biotech Co., Ltd Novel Coronavirus 2019-nCoV Antibody Test (colloidal gold method) LFIA based on gold particle IgG Serum and plasm Results in 15 min China NMPA (20203400796) 10/12/2020 Note: "-" means information was not available Table 2. Serological tests used to detect antibodies to SARS-CoV-2 approved by NMPA.
-
The design of the LFIA relies on a strip or dipstick containing immobilised test reagents, which is enclosed in a cassette. For example, for the detection of SARS-CoV-2 IgG antibody using gold particle-based LFIA, the cassette is usually composed of a sample pad (region 1), gold conjugate release pad (region 2), nitrocellulose (NC) membrane containing antibodies or antigens (region 3 for the test line and region 4 for the control line), and an absorbent pad. When the SARS-CoV-2 IgG antibody is detected through the indirect immunoassay, gold-labelled SARS-CoV-2-specific antigens along with gold-labelled rabbit IgG, anti-human IgG, and anti-rabbit IgG polyclonal antibodies were immobilised in regions 2, 3 and 4, respectively. When deposited into the sampling well (region 1), the sample pad receives the sample and acts as a filter to aid flow, the sample rehydrates the gold-conjugated SARS-CoV-2 antigens or rabbit IgG on the conjugate pad (region 2) and the specific IgG for SARS-CoV-2 binds to SARS-CoV-2 antigens. The sample continues to flow along the NC membrane through capillary action, while test lines (T line) immobilised with anti-human IgG and control lines (C line) immobilised with anti-rabbit IgG indicate the result. The labels provide visible colour for the C and T lines. In this case, a lack of binding at the T line indicates a negative result. The appearance of the C line indicates that the test has run correctly (Koczula and Gallotta 2016; Arun Krishnan et al. 2020; Carter et al. 2020; Demey et al. 2020; Santiago 2020). When IgM is detected, anti-human IgM antibody is immobilised on the NC membrane (region 3). Typical detection results are shown in Fig. 1B. A positive antibody result indicates binding between the coating antigen and antibodies as well as binding by the secondary antibody.
LFIA can be utilised as a preliminary testing tool for COVID-19 in case of community or mass population screening (Koczula and Gallotta 2016; Arun Krishnan et al. 2020; Demey et al. 2020; Santiago 2020). Currently, SARS-CoV-2 IgG/IgM antibody detection kits based on LFIA are widely available in China (Wang D et al. 2020; Zhang et al. 2020). Upon SARS-CoV-2 infection, IgM and IgG antibodies are induced, which develop from 7 to 10 days and 14–20 days after infection, respectively. IgM can be detected earlier but decreases and disappears very soon, whereas IgG appears slightly later but can persist for a long time after infection (Al Kahlout et al. 2019; Lijia et al. 2020; Liu et al. 2020; Norman et al. 2020; Wolff et al. 2020; Xia et al. 2020; Xiang et al. 2020). Although antibody test kits are reliable for preliminary screening, they may not be appropriate as a confirmatory test due to nonspecific interactions of antibodies and other proteins in the blood with the capture and detector molecules in the membrane, which may result in false positive or false negative results. The LFIA kit is characterised by easy standardisation and rapid detection. The main advantage of LFIA is that it can be used for rapid POCT.
-
SARS-CoV-2 N, S, or fragments of N or S are adsorbed onto the surface of a 96-well plate as the capture molecule, and anti-human IgG/IgM antibody conjugated with horseradish peroxidase is used as the detector molecule for ELISA (Fig. 1C, D). Manual ELISA kits for detection of COVID-19 were developed by BGI Biotechnology (Beijing) CO., LTD in China (Table 2). ELISA-based COVID-19 antibody and antigen detection kits are currently under development by many manufacturers.
Generally, several reagents are prepared for CLIA based on magnetic particles, including reagent 0 (magnetic particles-anti-FITC antibody), reagent 1 (FITC-labelled novel coronavirus recombinant antigen), reagent 2 (alkaline phosphatase-labelled mouse anti-human IgG monoclonal antibody), negative control, positive control, and other necessary auxiliary reagents. CLIA is designed on the principle of indirect detection of the anti-SARS-CoV-2 IgG antibody in human serum (Fig. 1E). Reagent 0, reagent 1 and the sample are first mixed in the reaction tube. Anti-SARS-CoV-2 IgG in the samples forms a complex with the recombinant antigen and simultaneously binds to the magnetic particles, and then the unbound components in the tube are washed away. Subsequently, reagent 2 is added to the reaction tube. The alkaline phosphatase-labelled antibody acts as a secondary antibody and binds to the IgG antibody in the sample, forming an alkaline phosphatase-labelled mouse anti-human Ab-human IgG-recombinant antigen-magnetic particle complex, and then the unbound components are washed away. The luminescence value (RLU) of each sample tube is determined through addition of a substrate solution to the automatic immunoassay system, followed by catalysis of luminescence from the substrate solution by alkaline phosphatase. The luminescence value of the sample was positively correlated with the IgG antibody concentration of the novel coronavirus, allowing detection of the IgG antibody of SARS-CoV-2 in human serum. The main advantage of automated CLIA analysers over rapid LFIA tests is the very high throughput of samples. CLIA platforms are widely used as serological techniques for the quantitative detection of specific antigens or antibodies in samples (Johnson et al. 2020; Lijia et al. 2020; Liu et al. 2020; Wan et al. 2020; Xia et al. 2020). Various detection kits have been developed in China since the outbreak of SARS-CoV-2 (Lijia et al. 2020; Liu et al. 2020; Wan et al. 2020; Wang P et al. 2020; Xia et al. 2020).
Antibody detection results can help to determine the infection stage of a patient. This method is usually used for supplementary testing of cases with negative SARS-CoV-2 nucleic acid tests, used in conjunction with nucleic acid tests for the diagnosis of suspected cases, or used in serological surveys and past exposure surveys of high-risk population groups (Arun Krishnan et al. 2020; Li H et al. 2020; Liu et al. 2020). According to the Technical Guidelines for COVID-19 Laboratory Testing of China, laboratory-confirmed positive cases must meet one of the following two conditions: 1. Serum IgM antibodies and IgG antibodies to SARS-CoV-2 are positive; 2. Serum IgG antibodies to SARS-CoV-2 turn from negative to positive or IgG antibody titres during the recovery period are four times the level in the acute phase or higher. Suggested rules for these judgements are provided in Table 3.
No. Nucleic acid IgM IgG Interpretation Treatment measures 1 + – – Patients may be during the "window period" of SARS-CoV-2 infection, typically within 2 weeks after infection Isolation, observation or clinical treatment 2 + + – May be at early infection phage of SARS-CoV-2 3 + – + May be during the mid and late infection stage or recurrent infection. When the IgG antibody in the recovery period increases by 4 times or more compared with the acute phase, a recurrent infection can be diagnosed 4 + + + The patient is in the active infection phase, a certain immunity to SARS-CoV-2 has already been developed 5 – + – One is likely to be in the acute phase of SARS-CoV-2 infection. Nucleic acid testing results should be confirmed first. Other factors such as rheumatoid factors have been found to cause weak IgM positive or positive tests. The result may suggest that one might have been vaccinated recently Vaccination should be ruled out firstly. Observe, exclude the possibility of false negative of nucleic acid, detect the nucleic acids in different kind of samples once more every 3-5 days, and recheck the antibody level about 7-14 days later to confirm whether elevation appeared. One with both IgM and IgG positive could be diagnosed as a patient. Someone would be isolated according to clinical manifestation and epidemiological history 6 – – + One might have recovered, and the virus has been cleared. The IgG could be detected for a long time in the blood. The result may suggest that one might have been vaccinated previously 7 – ± – One experience the first infection, during an early stage. Thus, the viral load is lower than the lower limit of nucleic acid detection. A small amount of IgM has been produced while IgG has not; a false positive result might be caused by rheumatoid factor. The result may suggest that one might have been vaccinated recently 8 – + + One might be recently infected with SARS-CoV-2 and is during the recovery period. The virus has been cleared, but the IgM has not been reduced to the lower limit of detection; or the nucleic acid test result might be false negative and the patient is indeed in the active infection stage. The result may suggest that one might have been vaccinated recently Note: "+", positive; "-", negative; "±", weak positive Table 3. Interpretation of the clinical status of individuals based on nucleic acid and antibody detection results.
Lateral Flow Immunoassay (LFIA)
ELISA and CLIA
-
Two antigen-based kits for rapid SARS-CoV-2 detection have recently been approved by the NMPA of China. These antigen-based detection kits were developed based on LFIA using the double antibody sandwich method. Conjugated mouse IgG (antibody 1) specific to SARS-CoV-2 antigen, the paired mouse antibody (antibody 2) against SARS-CoV-2 antigen and goat anti-mouse IgG are immobilised on regions 2, 3 and 4, respectively. In contrast to the serum, plasma and blood samples used for serological detection, the samples used for antigen detection are pharyngeal and nasopharyngeal swabs, which must be treated with protein extraction liquor provided in the kits prior to detection. Antigen detection kits were used solely during the acute stage of SARS-CoV-2 infection, and nucleic acid detection, imaging examination, epidemiological history, and other diagnostic techniques should also be considered when making an ultimate diagnosis.
-
rRT-PCR has played an important role in early detection, and is considered the 'gold standard' for COVID-19 testing. However, false negative results due to inappropriate sample collection timing, sample type, sampling technique or other problems have limited its usage and thus increased the importance of serological testing (Coupeau et al. 2020; Lee et al. 2020; Li and Ren 2020; Li H et al. 2020; Petrillo et al. 2020; Wolff et al. 2020). Serological assays can detect previous SARS-CoV-2 infection and provide information on the progression of the disease, but the tests must be performed within the correct time frame after the onset of the disease (Arun Krishnan et al. 2020; Carinci et al. 2020; Lee et al. 2020; Xu Y et al. 2020). For the antibody detection assay, both IgG and IgM antibodies against SARS-CoV-2 are target analytes, with IgM appearing in the blood within a week after infection, while IgG expression requires more than 10 days (Demey et al. 2020; Lou et al. 2020; Xiang et al. 2020). The combined utilisation of both molecular and serological tests can help to clarify the progression of disease and the proper response (Table 3). In the seventh edition of the Technical Guidelines for COVID-19 Laboratory Testing delivered by the National Health Commission, in addition to nucleic acid detection results, antibody IgM and IgG positivity have been added as some of the criteria for identifying positive COVID-19 cases. Different results determine the most appropriate treatment measures (Table 3). More importantly, the diagnosis of a case might not be based solely on the nucleic acid and antibody tests but should also involve comprehensive consideration of the clinical manifestation, biochemical tests, and imaging examination. In particular, when nucleic acid detection tests are unavailable, clinical observation plays an essential role.
Nucleic acid-based diagnostic methods, serological assays, and antigen detection techniques provide strong tools for COVID-19 diagnosis. Currently, quality control of the diagnostic tests being developed is very important. Notably, although serological diagnosis of COVID-19 has developed rapidly, many concerns regarding the sensitivity and specificity of the assays remain (Norman et al. 2020; Xia et al. 2020; Xu Y et al. 2020). The development of innovative, well standardised, highly sensitive and specific, and low-cost serological assays for COVID-19 diagnosis has been extremely rapid due to the complexity, cost and limitations of nucleic acid-based diagnostic tools. Meanwhile, the battle against COVID-19 is likely to support the development of accurate and efficient POCT for nucleic acid detection (Augustine et al. 2020; Lu et al. 2020a; Subsoontorn et al. 2020).
-
This work was supported by the National Key Program for Infectious Disease of China (2018ZX10101002), and the National Key Research and Development Program of China (2016YFD0500301, 2020YFC0840900).
-
The authors declare that they have no conflict of interest.
-
This article does not contain any studies with human or animal subjects performed by any of the authors.














 DownLoad:
DownLoad: