-
Foamy viruses (FVs; also known as spumaviruses or spumaretroviruses) were once classified as one of the genera of the Retroviridae. However, they possess different replication and gene regulation strategies from other retroviruses and consequently these characteristics led to the creation of a distinct viral subfamily known as the Spumaretrovinae by the International Committee on Virus Taxonomy in 2002. In fact, their replication may bridge the gap between retroviruses and hepadnaviruses (1). However, they share no commonalities in their pathology, because FVs have not been found to cause any apparent diseases (5, 7). So research concerned with their distinct gene regulation strategies and interaction with host cells has generated much interest. Furthermore, their distinct characteristics may contribute to the construction of gene vector based on FVs and benefit work on disease therapy.
Tas, the transactivator of FVs, plays a key role in viral replication and gene regulation by binding with LTR and IP (4). Recently, it has been reported that the interaction of Tas and some cellular proteins, such as the promyelocytic leukemia protein, could affect both viral gene expression and infectivity (6). The 249aa Borf1 (or BTas, the Tas equivalent of PFV) is encoded by the first open reading frame (ORF). Previously, we had demonstrated that Borf1 was the transactivator of BFV (2) and could regulate viral gene expression by binding to LTR and IP (3, 8). To further investigate its structural localization and function, we raised a hightiter anti-Borf1 serum against the recombinant soluble Borf1 protein. Western blot analysis and immnofluoresence assay showed that this antiserum could recognize the eukaryotic Borf1 with high specificity. Also, the nuclear and cytoplasmic localization of Borf1 was confirmed in HeLa cells that were transfected with Borf1. Furthermore, with this specific antibody, we showed the nuclear and cytoplasmic localization of Borf1 in Fetal Bovine Lung (FBL) cells that were infected by BFV. These results further confirmed our findings about the transactivator role of Brof1 in BFV infection, and provided an effective way to monitor the activities of Borf1 with this specific antibody, as well as a way to further investigate the molecular mechanism of this key process in infection.
HTML
-
Fetal Bovine Lung (FBL), HeLa and 293T cells (conserved by our lab) were grown in DMEM (Gibco) supplemented with 10% (v/v) fetal bovine serum (HyClone) and antibiotics.
-
The complete coding region of Borf1 was amplified by PCR from Hirt DNA of BFV. The following two primers, which incorporate the EcoR Ⅰ or Sal Ⅰ site, were designed for PCR: Forward, 5'-cag gaa ttc gag atg gct agc gg-3'; Reverse, 5'-cag gtc gac cgc aac aca tat c-3'. The resulting PCR product was cloned into pGEX-6p-1 to construct prokaryotic expression plasmid pGEX-Borf1, and also cloned into pCMV-Tag2B to construce eukaryotic expression plasmid PcmvTag-Borf1. The resultant plasmids were checked by restriction enzyme analysis and sequencing.
-
The prokaryotic expression plasmid pGEX-Borf1 was transformed into E. coli BL21 (DE3) (Invitrogen), and the positive colonies were selected by antibiotics. Briefly, the desired clone was grown overnight at 37 ℃, inoculated into fresh media and grown for an additional 3 h (OD600 = ~ 0.7) and then induced by IPTG at a final concentration of 1 mmol/L in culture medium for 4 h. The expressed fusion proteins were subsequently purified by Glutathione Sepharose 4B (Amersham) with PreScission Protease according to the manufacturer's instructions. In addition, an anion exchange column and molecular sieve chromatography were used to purify this protein.
-
The purified Borf1 protein was used as the antigen to raise antiserum in mice. Five BALB/C mice were firstly immunized with 50 μg purified protein that was emulsified in an equal volume of Freund's complete adjuvant. The inoculations were performed by subcutaneous injection in the belly. Freund's incomplete adjuvant and 30 μg purified protein were used in subsequent injections. Three booster injections were given every other week after the first injection. Three days after the last immunization, the mice were sacrificed to prepare the antiserum.
-
First, purified Borf1 was incorporated in a 96-well plate (0.04 μg per well). Then, each well was incubated with different dilutions of the anti-Borf1 serum. After washing with PBS, a 1:3 000 dilution of goat anti-mouse peroxidase-labelled secondary antibody was added. Finally, the wells were treated with TMB and subsequently analyzed by a Precision Microplate Reader.
-
293T cells were transfected with the pCMVTagBorf1. After 48 h, the cells lysates were electrophoretically separated on the SDS-PAGE and electrically transferred to the PVDF Membranes (Amersham). Nonspecific protein banding was blocked by treating the membranes at room temperature for 45 min with the phosphate buffered saline (PBS) containing 5% skimmed milk powder. The membranes were washed five times with the PBS containing 0.05% Tween 20 (PBST) and incubated at room temperature for 1 h with a 1:3 000 dilution of the anti-Borf1 serum or the mouse anti-Flag antibody in PBS. After washing five times with PBST, the membranes were incubated with a 1:3 000 dilution of the goat anti-mouse peroxidaselabelled secondary antibody at room temperature for 45 min. The membrane was then washed five times with PBST, treated with Western Blot Luminal Reagent (Santa Cruz), and exposed to an X-Omat BT Film (Kodak).
-
HeLa cells were grown on coverslips and transfected with the pCMVTag-Borf1 or pCMVTag2B (Stratagene). After 48 h, cells were fixed in 4% paraformaldehyde and permeabilized with 0.2% Triton X-100. Coverslips were blocked with 3% BSA to reduce the non-specific background. The cells were reacted at 37℃ for 30 min with the anti-Borf1 serum diluted 1:400 in PBS, washed in excess PBST and then reacted with a 1:100 dilution of the Cy2-conjucted AffiniPure Goat Anti-Mouse IgG (Jackson ImmunoResearch). Cell nuclei were stained with 4, 6-diamidino-2-phenylindole (DAPI), and the fluorescent images were taken on a confocal microscopy.
Cells and virus
Plasmid Construction
Expression and purification of Borf1
Generation of Antibody
ELISA
Western blot analysis
Immuno-fluorescence assay
-
The prokaryotic expression plasmid pGEX-Borf1 was constructed and checked by the three restriction enzymes Pst Ⅰ, EcoR Ⅰ/Sal Ⅰ and Pvu Ⅰ (Fig. 1). The plasmid was then transformed into E. coli BL21 (DE3). Using pGEX-6p-1 transformed E. coli BL21 (DE3) as the control, SDS-PAGE analysis showed that there was a specific band associated with the pGEXBorf1 transformed E. coli BL21 (DE3), which was consistent with the predicted molecular weight of 54 kDa (Fig. 2).
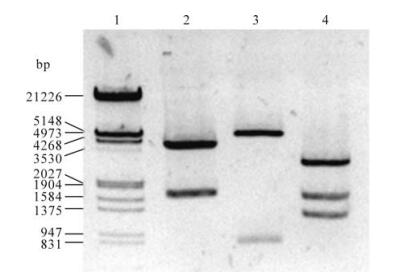
Figure 1. Identification of construct pGEX-6p-Borf1. 1, DNA marker; 2, Pst Ⅰ: 4.0kb, 1.7kb; 3, EcoR Ⅰ/Sal Ⅰ: 4.9kb, 0.8kb; 4, Pvu Ⅰ: 2.9kb, 1.6kb, 1.2kb.

Figure 2. SDS-PAGE analysis of pGEX-6P-Borf1 expression in E.coli BL21 (DE3). 1, Protein Marker; 2, BL21 (DE3); 3, BL21 (DE3) + IPTG; 4, BL21 (DE3) transformed with pGEX-6P-1; 5, BL21 (DE3) transformed with pGEX-6P-1 + IPTG; 6, BL21 (DE3) transformed with pGEX-6P-Borf1; 7, BL21 (DE3) transformed with pGEX-6P-Borf1 + IPTG..
-
To obtain purified Borf1, we used three steps in the purification process. First, the GST-Borf1 was purified from the bacterial lysates by the affinity chromatography using Glutathione Sepharose 4B. The cleavage of the Borf1 protein from GST was achieved by using PreScission Protease. Later, we purified the Borf1 by an anion exchange column Resource Q and the molecular sieve chromatography (120 mL superdex G200). Then the superdex elutes was analyzed with SDS-PAGE. The final purification of the Borf1 exceeded 98% (Fig. 3).
-
Purified Borf1 protein was immunized in 5 BALB/ C mice. After four immunizations, the titer of antiserum was tested with ELISA. The five samples had a titer of 1:4×104, 1:16×104, 1:4×104, 1:8×104 and 1:16×104, respectively. The results showed that the antiserum could be used in western blot analysis. Then the antiserum were harvested and stored in -20℃.
-
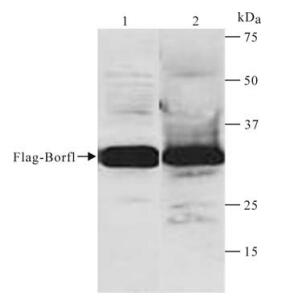
To test the specificity of the antiserum, pCMVTagBorf1 was transfected into 293T cells. After 48 h, the Flag-Borf1 protein was expressed in 293T cells. Then hese cells were lysated and immunoblotted with antiBorf1 serum and anti-Flag antibody, respectively. The Fig. 3. SDS-PAGE analysis of Superdex elutes. 1-4, Elutes sample, elution volume is 12.5 mL, 13 mL, 13.5 mL, 14 mL; 5, The condensed sample; 6, Marker. result showed that there is a unique band of ~ 30 kDa in both lanes (Fig. 4). This demonstrated that the antiorf1 serum can recognize the Borf1 protein expressed in eukaryotic cells and has a similar specificity with the commercial anti-Flag antibody.
-
Using the antiserum, we next tested the intracellular localization of Borf1. First, we tested the localization of Borf1 in transfected cells. HeLa cells were transfected with the pCMVTag-Borf1 or pCMVTag-2B (negative control). Then, the indirect immunofluorescence staining was used to confirm the localization of Borf1. The result in Fig. 5A shows that Borf1 was located in both the cytoplasm and the nu-cleus, but predominantly in the nucleus. In the negative control, there was no green fluorescence (Fig. 5B).

Figure 5. Intracellular localization of Borf1. A: The HeLa cells were transfected with the pCMVTag-Borf1. B: The HeLa cells were transfected with the pCMV-Tag2B. C: The FBL cells were infected with BFV. D: The FBL cells were not infected with BFV.
Next, we tested the localization of Borf1 during BFV infection. Because BFV can infect the FBL cells and lead to the rapid syncytium formation, we chose these cells as a suitable target. Forty-eight hours after infection by BFV, the infected FBL cells were used to perform the immunofluorescence assay with antiBorf1 serum. The result showed that Borf1 located in both the nucleus and cytoplasm (Fig. 5C), but no green fluorescence was shown in the negative control (Fig. 5D). All these results show that the anti-Borf1 serum can correctly recognize the natural Borf1 and can be used for immunofluoresence assay. In addition, the localization of Borf1 was identical in both transfected cells and infected cells.
Gene expression in E.coli
Purification of Borf1 protein
Preparation of anti-Borf1 serum
Specificity analysis of anti-Borf1 serum
Intracellular localization of Borf1
-
Previously, we had demonstrated that Borf1 showed its transactivation role by activating both the LTR and IP of BFV in the reporter gene assay. We also revealed that this activation process was completed by the direct interaction of Borf1 with LTR or IP (3, 8). In this study, the results about the subcellular localization of Borf1 contribute to our knowledge of the activities of Borf1 during the BFV infection. Borf1 is expressed in the cytoplasm of infected cells and commences activation of viral gene expression by entering the nucleus of the infected cells; with the activation of viral gene expression, the infected cells begin to present the typical syncytia signature at this stage of the BFV infection.
Combined, these results provide a sketch of the Borf1 activities during BFV infection, but the molecular mechanism of the Borf1 subcellular localization remains unclear. Although computer based analysis failed to predict any sequence which could serve as a possible nuclear localization signal (NLS), there must nevertheless be some sequences that are responsible for this crucial activity of Borf-1 during the infection of BFV. Deleting the Borf-1 proteins may help to identify this mechanism. On the other hand, there might be other proteins that can interact with Borf1 and contribute to the localization switch of Borf1 from cytoplasm to the nucleus during the infection process.















 DownLoad:
DownLoad: