-
Rabies is one of the major zoonotic diseases caused by the rabies virus, and is almost always fatal in 100% of cases, once symptoms occur. Pre-exposure or post-exposure prophylaxis of rabies is the only effective way to prevent human rabies. In China, more than 1000 human rabies cases have occurred in recent years, and 87.5% of these cases were caused by dog bites (Yin C P, et al., 2011). Moreover, over 12 million people in China are inoculated every year with the rabies vaccine for pre-exposure and post-exposure prophylaxis.
The titer of the rabies virus-neutralizing antibody (RVNA) is an important parameter for evaluating the efficacy of the immunization vaccine for either pre-exposure or post-exposure prophylaxis for rabies. Detection of RVNA levels is also very important for laboratory diagnosis of human rabies. The World Health Organization (WHO) recommends that booster immunizations should be carried out with reference to the titer of RVNA when it is < 0.5 international units (IU) per mL (World Health Organization, 2005).
In October 2005, a WHO expert consultation on rabies recommended that evaluation of RVNA levels should be carried out using the rapid fluorescent focus inhibition test (RFFIT) or the fluorescent antibody virus neutralization test (FAVN) (World Health Organization, 2005). RFFIT is also one of the standard methods specified in the Pharmacopoeia of the People's Republic of China for evaluation of the efficacy of human rabies vaccines.
The RFFIT method is a neutralization test. When a fixed amount of rabies virus and serial dilutions of serum is neutralized, the remaining virus in each dilution is inoculated into cultured cells, and assessed using a specific anti-rabies nucleoprotein antibody labeled with fluorescein. The quantity of the remaining virus is calculated according to the number of infection foci produced by the remaining virus. The titer of RVNA in the samples is calculated according to the number of virus infection foci and the quantity of the reference serum, and expressed as IU/mL.
In our laboratory, we established an RFFIT method according to the above procedure and preliminary work, in order to evaluate the efficacy of human rabies vaccination, and we present the results of this work below.
HTML
-
We used the WHO second international standard for rabies immunoglobulin (WHO STD), prepared by the WHO international laboratory, as the biological reference standard, with the titer of 30 IU/mL.
We also used an anti-rabies nucleoprotein antibody labeled with fluorescein (Rabies DFA Reagent; Millipore Corp., Billerica, MA, USA).
-
Two groups of human sera were used. Group A comprised 85 human sera (Guangzhou Center for Disease Control and Prevention (Guangzhou CDC), Guangzhou, China) which were used for testing the specificity of our RFFIT procedure: 40 human sera from individuals who had never been vaccinated with rabies vaccine, and 45 from individuals vaccinated by domestic PVRV using the Essen five-dose regimen. To test the repeatability of RFFIT, a second group of sera was used: group B comprised 15 human sera (Guangzhou CDC) from rabies-vaccinated individuals, which were tested triple using our RFFIT method.
-
We assessed whether different vaccination regimens would have an effect on efficacy. To evaluate the Zagreb 2-1-1 vaccination regimen and the Essen regimen, we recruited 112 healthy volunteers (sex and age were neglected) who were informed consent to receive post-exposure prophylaxis, and divided them into three groups. Group C comprised 50 subjects who received a domestic purified Vero cell rabies vaccine (SPEEDA PVRV) using the Zagreb 2-1-1 regimen; group D contained 32 subjects who received imported purified chick embryo cell rabies vaccine (PCECV) using the Zagreb 2-1-1 regimen; and group E consisted of 30 subjects who received PVRV (SPEEDA) using the Essen regimen (Table 1). From each subject, 5 mL of venous blood was collected on each of days 0 (before vaccination), 7, 14, and 45 (D0, D7, D14 and D45, respectively). The blood samples were centrifuged, and the isolated sera were frozen at -70 ℃ until tested.
Group C Group D Group E Subjects, n 50 32 30 Vaccine PVRV PCECV PVRV Regimen Zagreb Zagreb Essen Injection D0 (2 doses), D7, D21 D0 (2 doses), D7, D21 D0, D3, D7, D14, D28 D, day; PCECV, purified chick embryo cell vaccine; PVRV, purified Vero cell rabies vaccine. Group C: PVRV, Zagreb regimen; group D: PCECV, Zagreb regimen; group E: PVRV, Essen regimen. Table 1. Comparison between different vaccines and regimens.
-
A BSR cell monolayer was grown for 3 days until it reached 100%. The monolayer was digested with trypsin, and suspended in DMEM-10 medium. 1×106 cells/mL of BSR cell suspension was prepared and 2 mL suspension was inoculated into a 25 cm2 flask, and cultured in 5% CO2 in an incubator at 37 ℃.
-
The virus (CVS-11) was diluted in DMEM-10, using a set of five-fold serial dilutions (30 µL of virus added to 120 µL DMEM-10) in two adjacent lines in a 96-well plate. Then 50 µL virus suspension was removed from each well and transferred to the corresponding well of another 96-well plate, containing 100 µL DMEM-10 per well. After incubation at 37 ℃ for 1 hour, 50 µL BSR cell suspensions (1×106 cells/mL) were added to each well and incubated at 37 ℃ with 5% CO2 for 24 hours.
Direct fluorescent assay (DFA) was conducted to calculate the foci of infection from CVS-11 using the Rabies DFA Reagent. The medium was removed and the cells were rinsed with PBS and fixed with 80% cold acetone at 4 ℃ for 30 minutes, then stained with the Rabies DFA Reagent solution. Finally, 60% glycerol-PBS was added, then the virus infection foci were counted under the microscope at 10× magnification, and the titer was calculated. This procedure was repeated for calculating the titer of the whole batch of virus, which was stored at -70 ℃ until required.
-
Prepared abundant of BSR cell suspension, CVS-11 virus at MOI = 0.1 was added to the BSR cell suspension and incubated at 37 ℃ with 5% CO2 for 1 hour to adsorb adequately. The suspension was then transferred into cell culture flasks and incubated at 37 ℃ with 5% CO2 for 72 hours. The virus-cell culture medium was mixed, divided into small tubes, labeled, and stored at -70 ℃.
-
The serum samples were denatured at 56℃ for 30 minutes, each serum sample was diluted as a set of three-fold serial dilutions using DMEM-10 in a 96-well plate. The control system included the WHO reference serum, negative serum control, virus control, and cell control. The WHO reference serum and negative serum were both diluted in the same way as the sample sera. A two-fold dilution for the virus control was also prepared. CVS-11 suspension (with sufficient quantity to infect 80% of cells in a single well) was added to each well with the exception of the virus and cell control wells. After neutralization at 37 ℃ for 1 hour, 50 µL BSR cell suspensions (1×106 cells/mL) were added to each well and incubated at 37 ℃ with 5% CO2 for 24 hours.
DFA testing was conducted to measure the percentage of virus infection foci before and after 50% of cells infected, and these were used to calculate the titer of the serum samples.
Reagents
Serum samples for specificity and repeatability
Experimental groups for different vaccines and regimens
BSR cell culture
Virus titration
Culturing of CVS-11
RFFIT test procedure
-
Fluorescent foci were counted for the two wells in each line that first showed fluorescence (for example, in the first line N1, N2 (N1 > N2); the second line N1', N2' (N1' > N2')), then the virus titer was calculated as follows:
5 means the dilution factor; diluent fold was the corresponding dilution of virus in N1 and N1' wells; and the unit of virus titer was fluorescent focus units (FFU) per mL.
-
No fluorescent foci were found in the cell control wells, and > 50% of cells were infected in the first well of the negative control, while the virus control wells had 80-90% fluorescent foci (Fig. 1). Pictures were taken using a fluorescence microscope (IX51; Olympus, Tokyo, Japan).
-
The neutralizing antibody titer was calculated (Excel software; Microsoft Corp., Redmond, WA, USA) using the Reed and Muench formula (see below), which measures the 50% end-point dilution of the standard serum (STD) and the sample serum. The titer of the sample serum was calculated according to the following formula:
-
Group A sera (40 never-immunized human sera and 45 human sera immunized with the Essen regimen) were tested using the RFFIT method. The results were summarized in Table 2. The specificity of RFFIT was 100%. A titer of > 0.5 IU/mL was considered positive, and < 0.5 IU/mL was considered negative.
Unimmunized sera (n=40) Immunized sera (n=45) Negative 40 (0.01-0.1) 0 Positive 0 45 (0.63-461.67) Data are given as mean (range) in IU/mL. Table 2. Results of the specificity by RFFIT method.
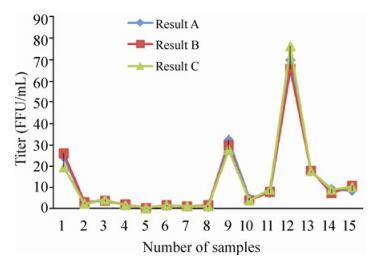
In group B, 15 rabies-vaccinated sera were tested using the RFFIT test, by three independent researchers. The three sets of results (a, b, and c) and the coefficient of variance of the three result sets were calculated (Table 3). We used the same virus stock and same serum samples almost simultaneously. The results showed the RFFIT method had good repeatability (Fig. 2).
Sample number Results, IU/mL GMT, IU/mL Standard deviation CV a b c 1 24.48 26.25 19.7 23.31 3.39 14.54 2 2.72 2.98 2.2 2.61 0.40 15.32 3 4.15 3.61 4.1 3.95 0.30 7.50 4 1.57 2.03 1.7 1.76 0.24 13.64 5 0.15 0.25 0.1 0.16 0.08 50 6 1.34 1.52 1.6 1.48 0.13 8.78 7 0.99 0.94 1 0.98 0.03 3.06 8 0.7 1.63 1.2 1.11 0.47 42.34 9 32.5 30 28 30.11 2.25 7.47 10 4.8 4 3.9 4.21 0.49 11.64 11 7.5 7.8 9 8.07 0.79 9.79 12 70 65.8 76.2 70.54 5.23 7.41 13 17.35 18 17.8 17.71 0.33 1.86 14 9.2 7.38 8.9 8.45 0.98 11.48 15 8.5 10.7 10.2 9.75 1.15 11.79 CV, coefficient of variation; GMT, geometric mean titer. Table 3. Results of the repeatability of RFFIT method.
-
Using the RFFIT method, 433 serum samples collected from 112 volunteers by different vaccines and regimens (group C: PVRV-Zagreb, group D: PCECV-Zagreb and group E: PVRV-Essen) were tested and the results analyzed (Table 4).
Day Group Subjects, n GMT, U/mL Ranges 95% CI, IU/mL N≥0.5 IU/mL Seroconversion, % 0 C 50 0.032 0.01-0.06 0.03-0.04 0 0 D 32 0.029 0.01-0.06 0.03-0.04 0 0 E 30 0.030 0.01-0.06 0.03-0.04 0 0 7 C 50 1.36 0.01-28.75 1.93-5.38 46 92 D 31 0.55 0.01-62.96 1.18-10.75 22 70.97 E 29 1.15 0.11-37.43 0.26-6.05 25 86.21 14 C 50 31.59 6.93-637.42 22.73-72.72 50 100 D 29 34.63 6.90-452.20 28.93-102.86 29 100 E 27 26.13 8.26-123.45 22.12-43.48 27 100 45 C 48 18.27 3.41-125.29 17.19-125.29 48 100 D 31 12.67 2.30-243.75 11.49-47.98 31 100 E 26 26.43 6.18-92.68 23.80-42.92 26 100 CV, coefficient of variation; GMT, geometric mean titer; PCECV, purified chick embryo cell vaccine; PVRV, purified Vero cell rabies vaccine. Group C: PVRV, Zagreb regimen; group D: PCECV, Zagreb regimen; group E: PVRV, Essen regimen. Table 4. Results of different vaccines and different regimens.
Geometric mean titer (GMT) of all the subjects at D0 ranged from 0.01 to 0.06 IU/ml, confirming that these subjects were naive to rabies vaccination. At D7, seroconversion was higher for the PVRV-Zagreb than for the PVRV-Essen regimen, indicating that antibodies were generated more rapidly by the Zagreb 2-1-1 regimen than the Essen five-dose regimen for the same vaccine. Regardless of vaccine and regimen, all 112 subjects achieved RVNA titers > 0.5 IU/mL on D14, and maintained this until D45. Interestingly, GMT was higher on D14 than D45 in the Zagreb regimen groups, indicating there was an increase in the antibody titer following the third vaccine dose on D7 than the increase after the fourth vaccine dose on D28. The rate of positive seroconversion was 100% on D14 and D45 after vaccination in all groups, with no significant difference in GMT for the two regimens on D14 and D45 suggesting that both the Zagreb and Essen regimens had a similar good immunological response, but the RVNA generated more rapidly and achieved higher titers with the Zagreb regimen.
Virus titration
Detection result of the controls
Calculation of the titer of anti-rabies neutralizing antibody
Specificity and repeatability of RFFIT
Amplification test of different vaccines and regimens
-
Currently, the methods recommended by WHO and Office of International Epizootics (OIE) for detecting RVNA are the mouse neutralization test (MNT (mice)), RFFIT, and FAVN. MNT is the gold standard for the detection of RVNA internationally, but the test operation is cumbersome, time-consuming, and detrimental to animals. In 1992, the WHO Expert Committee on Rabies recommended use of RFFIT instead of MNT to detect RVNA. Compared with MNT, the RFFIT method is simpler and faster, has good repeatability, and does not use animals. Relative to the international RFFIT, our micro-RFFIT method is based on the Pasteur RFFIT procedure, conducted in 96-well plates rather than on 8-well cell culture slides, which can reduce the cost of experiments.
The results of our tests indicated that our RFFIT method has good specificity and repeatability. The amplification test also gave good results, which provided laboratory evidence for the formulation of a new immunization program. After the establishment of our RFFIT method, we also performed other amplifications, including comparison of the RFFIT test with different standard sera and test procedures using different reference sera. We obtained significantly different results using different reference sera in our RFFIT method, showing that there is a need to unify the reference sera used worldwide (Yu P C, et al., 2012).
In recent years, there have been various improvements made to the RFFIT method. In France, Péharpré et al conducted fluorescent foci counting using computer imaging techniques and fluorescence microscopy, and found that this gave more accurate RFFIT (Péharpré D, et al., 1999), results, reducing the subjectivity of RFFIT identification. A Japanese study by Khawplod et al. using a recombinant rabies virus strain carrying a green fluorescent protein gene (rHEP-GFP) as the challenge virus for RFFIT showed that positivity could be observed directly without the need for a DFA test, which can reduce experimental expense and time.
In conclusion, we have established a new RFFIT protocol for RVNA, which can be used for detection of both human and animal RVNA. This test could be useful for investigations such as clinical diagnosis of rabies, detection of RVNA after rabies vaccination, preparation of immunoglobulin, evaluation of the effectiveness of new vaccines, and establishment of new immunization procedures. We hope it will enhance the comprehensive detection ability of institutions involved in rabies surveillance in China.
-
This work was supported by the National Department Public Benefit Research Foundation (201103032).
-
Conceived and designed the experiments: P Yu, Q Tang, and GD Liang; performed the experiments: P Yu; X Lv and X Shen helped with cell culture; P Yu and Q Tang wrote the paper.














 DownLoad:
DownLoad: