HTML
-
Enterovirus 71 (EV71) is an important human pathogen that may cause severe neurological complications and death in children under 6 years old (Chen S P, et al., 2010). There have been several outbreaks in the Asia-Pacific region during the past two decades and the virus has been considered a significant public health problem in the post-poliovirus eradication era (Solomon T, et al., 2010). In contrast to poliovirus, there are no effective vaccines or approved antiviral drugs available for use against EV71 (Wu K X, et al., 2010).
Supportive therapy is still the primary management strategy in severe cases of EV71 infection (Schezenmeir J D, et al., 2001; Vasiljevic T, et al., 2007). In addition to symptomatic treatment, intravenous immunoglobulin (IVIG) is used clinically to neutralize the virus and to suppress inflammation nonspecifically. Nevertheless, although this treatment has been applied routinely in severe cases of EV71 infection, its efficacy requires further evaluation (Nolan M A, et al., 2003). Therefore, the development of specific antiviral strategies against EV71 has become an urgent issue for the protection of children from the hazards of EV71 infection.
Plant polysaccharides are often identified as biological response modifiers (BRMs). Recently, greatly increased attention has been paid to plant polysaccharides, on account of their wide range of biological activities and their potential as sources of new 'functional foods', healthcare products and medicines (Chen L R, et al., 2009; Qiao D L, et al., 2009; Sun Y X, et al., 2008; Sun Y X, et al., 2009). Angelica sinensis (Oliv.) Diels (Umbelliferae), known as Dang-gui in Chinese and used widely as a tonic agent, is one of the most important traditional Chinese herbs (Hsu H Y, et al., 1976) and polysaccharides are amongst its principal pharmacologically active constituents. The pharmacological properties of A. sinensis include anti-oxidative, anti-inflammatory, and immunomodulatory activities (Cho C H., et al. 2000; Mei Q B, et al., 1991).
The roots of Astragalus membranaceus (AM) are also amongst the most popular health-promoting herbs in China and their use dates back more than 2000 years. Modern pharmacological studies have shown that the root of A. membranaceus possesses therapeutic activity as an immunostimulant, a tonic (adaptogenic), a hepatoprotective, a diuretic and an anti-diabetic agent (Ma X Q, et al., 2002). In addition, clinical studies have indicated that AM polysaccharides can counteract the side effects of chemotherapeutic drugs, notably by causing a substantial reduction in the degree of myelosuppression in cancer patients (Tin M, et al., 2007).
Many studies have indicated that a combination of the polysaccharides from Angelica and Astragalus roots, Guiqi polysaccharide (GQP), can exhibit -very likely synergistically in relation to its components -a range of antioxidant, anti-inflammatory and immunostimulatory effects. However, little research has yet been undertaken to characterize the pharmacology of GQP at a fundamental level and to relate it to the extraction methods that are used in the production of this traditional Chinese medicine.
The purpose of the present work was to extract and purify GQP and to examine whether GQP has anti-EV71 activity in vitro. For this purpose, cell survival, inhibition rates and cellular morphology were used as indices to evaluate the anti-EV71 activity of GQP by methyl thiazolyl tetrazolium (MTT) and observations of cytopathic effects (CPE). To our knowledge, this is likely to be the first report of the anti-EV71 activity of GQP in vitro.
-
A. sinensis and A. membranaceus were purchased from Minxian Shunfa Medicinal Material Co. (Gansu Minxian City, China). Dimethyl sulfoxide (DMSO) and MTT were obtained from Sigma Chemical Co. (St. Louis, MO, USA). Fetal bovine serum (FBS) and Dulbecco's modified Eagle's medium (DMEM) were purchased from GIBCO BRL (Auckland, New Zealand). Cell plates (96-well) were obtained from Corning Incorporated (New York, USA). Ribavirin (110502) was purchased from Tianjin Pharmaceutical Group Xinzheng Co., Ltd, Henan, China. All other reagents were of analytical grade and commercially available.
-
EV71 was donated by the Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Science (Guangzhou, China) and was propagated in African green monkey kidney (Vero) cells at 37 ℃.
Vero cells were obtained from the Central Laboratory of the School of Life Science and Engineering, Lanzhou University of Technology, Lanzhou, China. They were grown in DMEM, supplemented with 100 units/mL penicillin, 100 mg/mL streptomycin and 10% (v/v) fetal calf serum (FCS). FCS was reduced to 2% (v/v) for the viral infection. Titers of the virus were quantified on Vero cell monolayers by determination of the 50% Tissue Culture Infective Dose (TCID50), according to the Reed-Muench method (Yin Z, et al., 1997). The TCID50 value for EV71 was assayed to be 10-4.6/0.1 mL. A virus solution of 100× the TCID50 was used for the experiment.
-
Vero cells were seeded into 96-well plates, at 3x103cells/well, and incubated at 37 ℃ in an atmosphere of 5% CO2 until a cell monolayer had been formed. The cell monolayer was then washed thoroughly twice with phosphate buffered-saline (PBS, pH 7.4), the reagents or the virus were added, and the results were quantified. In this antiviral experiment, cells treated with the agents served as a cell control group, cells infected with the virus acted as a virus control group, and cells infected with the virus plus ribavirin served as a positive control.
-
A sliced mixture of untreated Angelica and Astragalus roots (in the ratio of 1:5, by weight) was extracted by refluxing twice with 95% ethanol, for 2 h each time, to remove pigments and small lipophilic molecules. The residue was then extracted with distilled water (in the ratio of 1:10, w/v) three times at 90 ℃, for 3 h each time. All the aqueous extracts were then combined, filtered, concentrated, and precipitated with anhydrous ethanol (extract:ethanol, 1:4, v/v) at 4 ℃ for 24 h, in order to precipitate the polysaccharides. Following centrifugation at 4000 g for 20 min, the collected precipitate was first de-proteinated by the Sevag method (Sun Y X, et al., 2008) and then washed, in turn, with 95 % ethanol, 100 % ethanol, and acetone, before being dried to yield a crude preparation of GQPs. The crude preparation was re-dissolved in distilled water and applied to a DEAE-52 cellulose column, equilibrated with distilled water. The polysaccharides were fractionated and eluted with distilled water followed by a step gradient of NaCl (0.05, 0.1, 0.25 and 0.5 M). The eluted polysaccharide fractions were then concentrated, before being further fractionated by size-exclusion chromatography on a Sephadex G-100 column and eluted with distilled water at a flow rate of 0.5 ml/min. The fractions were assayed spectrophotometrically by the phenol-sulfuric acid method (Dubois M, et al., 1956) and the peak fractions were retained. Three distinct main fractions (GQP-1, GQP-2 and GQP-3) were respectively collected, concentrated, dialyzed and lyophilized. The total carbohydrate content in each of these GQP preparations was assayed by high performance liquid chromatography (HLPC) (Wang J S, 2001). The fraction with the highest total carbohydrate content was selected as the experimental GQP preparation for the study of anti-EV71 activity in vitro. Before use, the fraction was diluted in DMEM culture medium to a concentration of 2 mg/mL and filter-sterilized through a sterile 0.22 μm filter.
-
The toxicity of GQP to Vero cells was determined as described by a previous report (Mosmann T I, 1983). Vero cells were seeded into 96-well plates (2×104 cells/well; 100 μL of cells). After 24 h of incubation at 37 ℃ in an atmosphere of 5% CO2, 100 μL of a solution of GQP or ribavirin was added and incubated sequentially for a further 24 h. Ten GQP or ribavirin solutions were tested, spanning a range of dilutions (1000 μg/mL, 500 μg/mL, 250 μg/mL, 125 μg/mL, 62.5 μg/mL, 31.2 μg/mL ….1.95 μg/mL). At the end of this period, 100 μL of the medium was removed and 20 μL of MTT solution (5 mg/mL) was added to each well. The plate was then wrapped in aluminium foil and incubated for a further 3-4 h. The medium was then carefully removed and 150 μL of DMSO was added to each well in order to dissolve the formazan crystals that had been formed. The MTT formazan product was determined by enzyme-linked immunosorbent assay (ELISA), measuring absorbance at 490 nm using a plate reader. The cell survival rate was calculated using the following equation:
Cell survival rate = (Aagent/Acell control)×100%.
All experiments were repeated three times. The cytotoxicity curve was then generated by plotting cell viability percentages against GQP concentrations. The 50% cytotoxic concentration (CC50) was estimated by linear regression analysis of the cytotoxicity curve.
-
The antiviral effect of GQP at different times of viral infection was evaluated as described originally by Chiang L C et al., 2002. Briefly, GQP or ribavirin, diluted in DMEM/10% FBS (GQP concentrations: 7.8 μg/mL, 15.6 μg/mL, 31.2 μg/mL or 62.5 μg/mL; ribavirin: 31.2 μg/mL), was added (100 μL/well) to 80%-confluent Vero cells, either 2 h prior to viral infection (t = -2), at the time of infection itself (t = 0), or 2 h after infection (t = +2). The infection was then carried out by adding EV71 to the wells (100 μL/well; at 100×TCID50). The three different infection conditions (t = -2, 0, +2) weie tested independently on separate plates. The final concentrations of GQP were respectively 3.9 μg/mL, 7.8 μg/mL, 15.6 μg/mL and 31.2 μg/mL; and that of ribavirin was 15.6 μg/mL (i.e., in each case, 50% of the reagent concentrations above). The plates were finally incubated for between 24 h and 72 h at 37 ℃ in a 5% CO2 incubator, after which the degree of virus inhibition was quantified through the alteration of the CPE, as described in the following section. All experiments were repeated three times.
-
To determine the ability of GQP to inactivate EV71, Vero cells (2×104cells/well; 100 μL) were seeded into 96-well plates and incubated overnight at 37 ℃ in a 5% CO2 incubator. When 80% confluent cells had been produced, the supernatant solution in each well was discarded and it was replaced, at time zero (t = 0), with a 1:1 (v/v) mixture (100 μL/well) comprising a solution of EV71 (100×TCID50) anda solutionof either GQP or ribavirin (GQP: 7.8 μg/mL, 15.6 μg/mL, 31.2 μg/mL or 62.5 μg/mL; ribavirin: 31.2 μg/mL). After 2 h of incubation at 37 ℃ in a 5% CO2 incubator, the monolayers were washed twice with PBS in order to remove excess reagents and any unbound virus. The plates were then filled with DMEM/10% FBS solution (150 μL/well) and incubated for a further 24-72 h at 37 ℃ in a 5% CO2 incubator. The degree of inhibition of the virus was recorded by microscopic CPE observations, comparing with the normal cell controls and the virus controls until the CPE score in the virus controls had become +++ and the cell controls still remained normal. The absorbance value (A490nm) in the MTT colorimetric assay was used to evaluate the direct inactivation effect of GQP upon EV71. The cell survival rate and the degree of inhibition (inhibitory rate) of EV71 by GQP were calculated using the following equation:
Cell survival rate = (Aagent/Acell control) × 100%
Inhibitory rate = (Aagent -A Virus control/Acell control -A Virus control) × 100%
The experiment was performed in triplicate. The effects upon CPE were observed and recorded as follows: a ≤25% change in CPE was scored as +, a 25-50% change as ++, a 51-75% change as +++, and a 76-100% change as ++++ (Chong H T, et al., 2001).
-
Vero cells were seeded into 96-well plates (2×104 cells/well; 100 μL) and incubated at 37 ℃ in a 5% CO2 incubator for 24 h in order to form cell monolayers. Then 100 μL of GQP (7.8 μg/mL, 15.6 μg/mL, 31.2 μg/mL or 62.5 μg/mL) or ribavirin (31.2 μg/mL) was added, respectively, to each well and the plates were cultured at 37 ℃ in a 5% CO2 incubator for 2 h. The supernatant solution (100 μL) was then discarded and EV71 (100× TCID50; 100 pL) was added to each well (therefore, in this experiment, t = -2). The plates were then cultured in the incubator under the same conditions for a further 24-72 h (Remesar M C, et al., 1988). Virus controls and cell controls were included, in parallel. Effects on CPE were then recorded and calculations of cell viability and inhibitory rate were performed, essentially as described in the immediately preceding section. The experiment was carried out in triplicate.
-
Vero cells were seeded into 96-well plates (2×104cells/well; 100 μL) and incubated at 37 ℃ in a 5% CO2 incubator for 24 h in order to form cell monolayers. Then EV71 (100×TCID50; 100 μL) was added to each well and the plates were incubated at 37 ℃ in a 5% CO2 incubator for 2 h. The supernatant solution (100 μL)was then discarded and 100 μL of GQP (7.8 μg/mL, 15.6 μg/mL, 31.2 μg/mL or 62.5 μg/mL) or ribavirin (31.2 μg/mL) was added, respectively, to each well (therefore, in this experiment, t = +2) (Peteis C J, et al., 1986). Virus controls and cell controls were included in parallel. Effects on CPE were then recorded and calculations of cell viability and inhibitory rate were performed, essentially as described in the preceding section. The experiment was repeated three times.
-
Data were analyzed by Analysis of Variance (ANOVA) using the statistical software SPSS 18.0 for Windows®. Data were considered significantly different when P values were less than 0.05.
Medicinal herbs and reagents
Enterovirus 71 and Vero cell line
Cell seeding and infection of cell cultures
Isolation and purification of Guiqi polysaccharide (GQP)
Assay of cytotoxicity of GQP to Vero cells
Time of addition assay
Direct inactivation effect of GQP upon EV71
Preventative effect of GQP upon the adsorption of EV71 to Vero cells
The inhibition efficacy of GQP on EV71 proliferation in Vero cells
Statistical analysis
-
The total carbohydrate contents of Fractions GQP-1, GQP-2 and GQP-3 were determined by HLPC to be 93.2%, 95.4% and 92.7% respectively. GQP2 was the fraction selected for the study of antiviral activity.
-
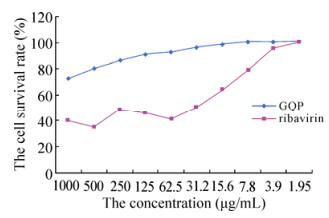
The cytotoxicity of GQP, in contrast to that of ribavirin, increased linearly in a dose-dependent manner as its concentration increased from 1.95 μg/mL to 1000 μg/mL (Fig. 1). For GQP, the concentration required to achieve 50% cytotoxicity (CC50), was greater than the highest concentration tested (> 1000 μg/mL), whereas the corresponding value for ribavirin was considerably lower -below 31.2 μg/mL. These results demonstrated that GQP was less cytotoxic than ribavirin. The highest concentration of GQP used in this experiment was 31.2 μg/mL, whereas the concentration of ribavirin used was 15.6 μg/mL.
-
GQP showed an appreciable direct inactivation effect on EV71, as indicated by the cell survival rate determined over a range of GQP concentrations. With concentrations of GQP of 3.9, 7.8, 15.6 and 31.2 μg/mL, cell survival rates of 43.4%, 51.0%, 58.1% and 65.8%, respectively, were observed; by comparison, with 15.6 μg/mL ribavirin, the cell survival rate was 63.9% and this was not significantly different from the value with 31.2 μg/mL GQP (P > 0.01). These values were significantly greater than the value obtained for cells treated with EV71 alone (28.6%) (P < 0.01).
The values for the inhibitory rate calculated for GQP at these concentrations were, respectively, 20.8%, 31.3%, 41.3% and 52.1%. The three lowest concentrations of GQP gave values for the inhibitory rate which were lower than that (49.5%) obtained with 15.6 μg/mL ribavirin (P < 0.05 for 15.6 μg/mL GQP, P < 0.01 for 3.9 and 7.8 μg/mL GQP; see Table 1 and Fig. 2C).
Groups Concentration (μg/mL) Optical density Cell survival rate Inhibitory rate GQP 31.2 0.518±0.040 65.8★★ 52.1 15.6 0.457±0.041 58.1★★▲ 41.3▲ 7.8 0.401±0.028 51.0★★▲▲ 31.3▲▲ 3.9 0.341±0.029 43.4★★▲▲ 20.8▲▲ Ribavirin 15.6 0.503±0.035 63.9★★ 49.5 Normal cell - 0.787±0.041 100★★ - Virus control - 0.225±0.012 28.6 0 GQP or ribavirin was added to Vero cells at the time of EV71 infection (t = 0) except for “Normal cell” and “Virus control” and then incubated at 37 °C in 5% CO2 incubator. When the cytopathic effect (CPE) in the ‘Virus control’ well had reached +++ (when CPE was evident in 50%-75% of the Vero cells) and the cells of the ‘Normal cell’ group were normal, the MTT assay was used to determine.A490 nm . Inhibitory rate (%) = (Aagent - AVirus control ) / (Acell control - AVirus control) ×100%. x±SD, n=3. ★★P < 0.01, compared to “Virus control”; ▲▲P < 0.01, compared to “Ribavirin”; ▲P < 0.05, compared to “Ribavirin”. Table 1. Direct inactivation effect of GQP upon EV71 (n = 3x±SD)
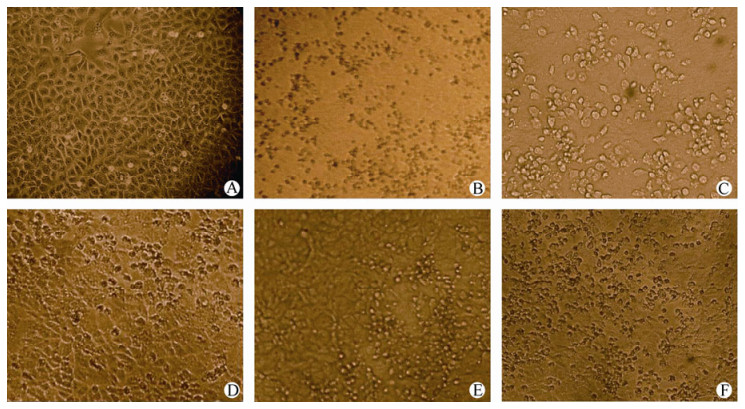
Figure 2. Effects of GQP and ribavirin upon CPE in EV71-infected Vero cells (magnified 100×). A: Normal Vero cells; B: EV71 control; C: GQP added at the point of infection (t =0); D: Prevention of viral absorption by GQP (GQP added 2 h prior to infection; t = -2); E: Inhibition of viral proliferation by GQP (GQP added 2 h post infection; t = +2); F: Inhibition of viral proliferation by ribavirin (ribavirin added 2 h post infection; t = +2). The concentration of GQP and ribavirin were 15.6 μg/mL.
-
When cells were pre-incubated with GQP prior to exposure to EV71, cell survival rates were substantially increased, compared to survival rates for controls without GQP. Thus, cell survival rates following pre-incubation with GQP at concentrations of 3.9, 7.8, 15.6 and 31.2 μg/mL were 63.8%, 75.3%, 85.8%, 94.2%, respectively -significantly higher than the cell survival rate (30.6%) for cells exposed to EV71 without pre-incubation with GQP (P < 0.01). The survival rate for each concentration of GQP was also significantly higher than that observed with 15.6 μg/mL ribavirin (P < 0.01). The virus inhibition rates with all concentrations of GQP were also significantly greater than the virus inhibition rate (26.9%) observed with 15.6 μg/mL ribavirin (P < 0.01; see Table 2 and Fig. 2D).
Groups Concentration (μg/mL) Optical density Cell survival rate(%) Inhibitory rate(%) GQP 31.2 0.747±0.082 94.2★★▲▲ 91.6▲▲ 15.6 0.681±0.058 85.8★★▲▲ 79.6▲▲ 7.8 0.597±0.043 75.3★★▲▲ 64.4▲▲ 3.9 0.506±0.045 63.8★★▲▲ 47.8▲▲ Ribavirin 15.6 0.391±0.035 49.3★★ 26.9 Normal cell - 0.793±0.041 100★★ - Virus control - 0.243±0.012 30.6 0 GQP or ribavirin was added to Vero cells 2 h prior to viral infection (t = -2) except for “Normal cell” and “Virus control”. Incubation was at 37 ℃ in 5% CO2 incubator. x±SD, n=3. ★★P < 0.01, compared to “Virus control”; ▲▲P < 0.01, compared to “Ribavirin”. Table 2. The preventative effect of GQP upon EV71 adsorption to Vero cells (n=3 x ± SD)
-
GQP was also effective in inhibiting the proliferation of EV71 in Vero cells. Thus, cell survival rates with GQP at concentrations of 3.9, 7.8, 15.6 and 31.2 μg/mL were 57.5%, 65.6%, 75.4% and 82.1%, lespectively -significantly higher than the cell survival rate (30.6%) for cells exposed to EV71 without treatment with GQP (P < 0.01). For the three highest concentrations tested, the cell survival rates with GQP were also higher than the value (60.9%) obtained with ribavirin at 15.6 μg/mL (P < 0.05 for 7.8 μg/mL GQP, P < 0.01 for 15.6 and 31.2 μg/mL GQP; see Table 3 and Fig. 2E-F).
Groups Concentration (μg/mL) Optical density Cell survival rate(%) Inhibitory rate(%) GQP 31.2 0.651±0.056 82.1★★▲▲ 74.2▲▲ 15.6 0.598±0.040 75.4★★▲▲ 64.5▲▲ 7.8 0.521±0.046 65.6★★▲ 50.5▲▲ 3.9 0.456±0.029 57.5★★ 38.7▲ Ribavirin 15.6 0.483±0.035 60.9★★ 43.6 Normal cell - 0.793±0.041 100★★ - Virus control - 0.243±0.012 30.6 0 GQP or ribavirin was added to Vero cells 2 h after EV71 infection (t = +2) except for “Normal cell” and “Virus control” and incubated at 37 °C in 5% CO2 incubator. x±SD, n=3. ★★P < 0.01, compared to “Virus control”; ▲▲P < 0.01, compared to “Ribavirin”; ▲P < 0.05, compared to “Ribavirin”. Table 3. The inhibitory effect of GQP on EV71 proliferation in Vero cells (n=3 x ± SD)
-
The time of addition assay revealed that GQP at 31.2 mg/mL consistently inhibited virus-induced cell death by more than 90% if it was added 2 h before infection (i.e. t = -2). The degree of inhibition of cell death that was observed when GQP was added at this time was greater than the degree of inhibition that was observed when it was added at the other times tested (i.e. t = 0; t = +2), and this effect was statistically significant (P < 0.01); and at t = -2 the effect with GQP was also greater than that observed with ribavirin (Tables 1, 2 and 3). GQP added 2 h after viral infection (t = +2) was more effective than GQP added at the same time as infection (t = 0); addition of GQP at the point of infection with EV71 was therefore the least effective of the three time-points for GQP addition that were tested. In contrast to the three lowest concentrations of GQP, ribavirin displayed significantly stronger (P < 0.05 for 15.6 μg/mL GQP, P < 0.01 for 3.9 and 7.8 μg/mL GQP) virus-inhibition activity when it was added either simultaneously with EV71 (t = 0). The inhibition rate for ribavirin under this condition was 63.9% -higher than the value (49.3%) observed for ribavirin addition 2 h before infection (t = -2).
Isolation and purification of Guiqi polysaccharide (GQP)
Cytotoxicity of GQP to Vero cells
The direct inactivation effect of GQP on EV71
The preventative effect of GQP upon EV71 adsorption to Vero cells
The inhibition efficacy of GQP on EV71 proliferation in Vero cells
Time of addition assay
-
In contrast to the situation with poliomyelitis, there is still no effective vaccine against EV71 available to provide immunity in children. Therefore, the development of anti-EV71 agents has become an urgent issue in epidemic-prone regions.
In this study, we have investigated the cytotoxicity and anti-EV71 activity of GQP colorimetrically using MTT, and we have observed the effects of GQP or ribavirin upon EV71-induced CPE using an inverted light microscope. Ribavirin is well known as a broad-spectrum antiviral drug, and it has been widely used in the treatment of viral diseases. Its mechanism of action is the inhibition of inosine monophosphate dehydrogenase; hence the replication of RNA and DNA viruses is inhibited (Kramer M, et al., 2007; Liu M L, et al., 2005; Liu Y W, et al., 2002).
Our investigations of anti-EV71 activity of GQP were performed in a concentration range from 3.9 to 31.2 μg/mL, in accordance with the results of cytotoxicity tests (Wang Y F, et al., 2004). Our initial objective was to obtain preliminary data relevant to the mechanism of action of GQP by exploring the effects of adding GQP at different time in vitro (t = -2, 0, +2) in relation to the time of EV71 infection.
The results showed that GQP had a weaker direct viral inactivation activity on EV71 than ribavirin; its inhibition rate (except at 31.2 μg/mL) was lower than that for ribavirin (P < 0.05 for 15.6 μg/mL GQP, P < 0.01 for 3.9 and 7.8 μg/mL GQP). The cell survival rate in the presence of GQP, however, was higher than that observed for cells in the presence of EV71 alone (P < 0.01).
GQP was more effective in blocking EV71 adsorption to Vero cells than in inhibiting EV71 replication within Vero cells; and in the case of each of these effects, it displayed a stronger activity than ribavirin. The virus inhibitory rates for GQP reached, respectively, 91.6% and 74.2% (at 31.2 μg/mL), significantly higher in each case than the corresponding value for ribavirin.
A possible explanation for these results is that GQP might have changed the structure of the Vero cell membrane in some way, thereby preventing adsorption at the cell surface and subsequent fusion of EV71 with the Vero cell. Furthermore, GQP might additionally have prevented EV71 from invading its host cell following fusion.
The inhibitory activity of GQP on EV71 proliferation was further studied by examining the CPE occurring within Vero cells. As shown in Fig. 2 C-E, the CPE within Vero cells caused by EV71 infection was clearly inhibited when: 1) GQP at 15.6 μg/mL was added 2 h before infection (t = -2; see Fig. 2D); 2) GQP was added 2 h post-EV71 infection (t = +2; see Fig. 2E); and 3) ribavirin at 15.6 μg/mL was added 2 h post-EV71 infection (t = +2; see Fig. 2F).
Although the CPE caused by EV71 infection was not obviously inhibited if GQP was added at the point of infection (t = 0), in contrast to the inhibitory effects that were observed if GQP was added to the cells either pre-or post-infection (t = -2; t = +2), nevertheless the findings we report here suggest that GQP could potentially be used in future in the prevention and treatment of diseases caused by EV71 infection. From our studies, GQP in vitro appeared to be more effective than ribavirin used at the same concentration, but this result would need to be followed up by a detailed evaluation of the antiviral effects of GQP upon EV71 in vivo.
In conclusion, GQP used at concentrations of 3.9 μg/mL-31.2 μg/mL exhibited clear antiviral effects on Vero cells infected with EV71, indicating that GQP could effectively reduce the infectivity of EV71 in vitro. Moreover, these effects were stronger than those of ribavirin. GQP may exert a major antiviral effect during EV71 infection; and since GQP is considered to be less toxic than the clinically licensed, broad-spectrum antiviral ribavirin, it may warrant further evaluation as a possible agent in the treatment of hand, foot and mouth disease (HFMD).
-
This study was supported by research grants from The National Natural Science Foundation of China (NO 81260070), The Project of Science and Technology of Lanzhou (NO 2011-1-71) and The Doctor Project of Lanzhou University of Technology (NO 0908ZXC127).
-
Pu X Y conceived and designed the study and revised the manuscript; Wang H R and Li Y performed the experiments and drafted the manuscript; Fan W B and Yu S assisted in the coordination and execution of the work, undertook the statistical analyses and assisted in drafting the manuscript.













 DownLoad:
DownLoad: