-
The rice gall dwarf disease, caused by Rice gall dwarf virus (RGDV), is a severe virus disease of rice in Southeast Asia and China [24]. This disease was first reported in central Thailand in 1980 [15, 18], and then occurred successively in Malaysia [16] and the rice-producing areas of China [3]. In recent years, it has been occurring severely and frequently in parts of Guangdong and Guangxi provinces of China, and is distributed sporadically in such areas as Yunxiao, Zhaoan and Zhangpu of Fujian province, and is exhibiting a tendency of spreading further [1]. RGDV is a member of the genus Phytoreovirus of the family Reoviridae, and is mainly transmitted in a persistent manner by Nephotettix cincticeps and Recilia dorsalis [6, 7]. Its virions (particles) exhibit a spherical icosahedron shape with a diameter of 65~70 nm, and have double shells. The viral genome is composed of 12 double-stranded RNA segments, designated S1 to S12 in ascending order of their mobility during SDS-PAGE gel electrophoresis [6]. So far, the complete sequences of RGDV isolates from Thailand [13] and Guangxi of China have been determined [23], laying the foundation for the research of the function of various segments.
RGDV virions have a total of six structural proteins. The minor core protein P1 encoded by S1 is an RNA polymerase, S2 encodes the minor outer capsid protein P2, P3 protein encoded by S3 is a major inner capsid protein, the minor core protein P5 encoded by S5 has guanylyltransferase activity, the minor core protein P6 encoded by S6 is a nucleic acid binding protein, and the S8-encoded P8 protein is a major outer capsid protein [13]. The structural protein of RGDV, especially the outer capsid, not only protects the viral nucleic acid from being impacted and damaged by external factors, acting as the primary antigen component, but also has such important biological functions as adsorbing to, penetrating and entering the host cell during virus infection [14]. Obtaining the recombinant protein by in vitro expression can facilitate the research of related functions of these proteins. The authors have tried to express the RGDV S8 gene in E.coli using the prokaryotic expression system, but the recombinant proteins existed in the form of inclusion body [4], and lacked the biologic activity they should have. Accordingly, this research, using the Bac-to-Bac baculovirus-insect expression system, constructs the RGDV outer coat protein gene S8 that has been cloned and identified in the baculovirus shuttle vector pFastBacTM1, and explores its expression characteristics in the Spodoptera frugiperda insect cell line (Sf9).
HTML
-
The baculovirus expression vector pFastBacTM1 was the product of the Invitrogen Company. The recombinant plasmid pMD-S8 was provided by our laboratory [2]. TaKaRa LA Taq DNA polymerase was purchased from TaKaRa Biotechnology (Dalian) Co., Ltd, and DNA restriction enzymes, T4DNA ligase and protein molecular weight standards were purchased from MBI Fermentas. Cellfectin transfection reagent was purchased from Invitrogen, and Grace's insect cell culture medium, fetal calf serum (FBS), streptomycin and penicillin were all products of GibcoBRL, USA. Alkaline phosphatase-goat anti-rabbit was purchased from Sigma Company, and RGDV outer coat protein polyclonal antibody was prepared in our laboratory [4].
-
E. coli DH5α and E. coli DH10Bac (containing bacmids and helper plasmids) were preserved in our laboratory. And Spodoptera frugiperda insect cell line (Sf9) was purchased from the China Center for Type Culture Collection (CCTCC).
-
The restriction enzymes, BamH Ⅰ and Xho Ⅰ, were used to digest the recombinant plasmid pMD-S8 that contained the RGDV outer coat protein gene S8. The target gene fragment was subcloned into the corresponding restriction site of pFastBacTM1, and the recombinant baculovirus transfer vector pFB-S8 was obtained and subjected to confirmatory sequencing.
-
The recombinant baculovirus transfer vector (pFB-S8) was transformed into E.coli DH10Bac competent cells. Two rounds of screening were performed on an LB plate containing 40 μg/mL IPTG, 100 μg/mL X-gal, 50 μg/mL kanamycin, 7 μg/mL gentamicin and 10 μg/mL tetracycline, and the recombinant virus DNA, i.e. rbpFB-S8, was extracted by alkaline process. PCR identification was performed using the pUC/M13 forward and reverse primers (M13F: 5′-GTTTTCCC AGTCACGAC-3′, M13R: 5′-CAGGAAACAGCTAT GA-3′), with the following reaction conditions: an initial denaturation at 93℃ for 3 min, followed by 35 cycles of 94℃ for 45 sec, 55℃ for 45 sec and 72℃ for 5 min, and an extension at 72℃ for 7 min.
-
Sf9 cells were cultured in Grace's medium (containing 10% FBS) until the cells almost covered the bottom of the bottle, inoculated on 6-well plates at a concentration of 9 ×105 cells/mL medium, and then transfected as described by Spector et al [20]The recombinant bacmid rbpFB-S8 was used to transfect Sf9 cells, mediated by Cellfectin. Three days later, the cellular supernatant exhibiting obvious symptoms of virus infection was collected to superinfect Sf9 cells for viral genome amplification. After four generations of infection, the cells were harvested, and the viral genomic DNA was extracted for PCR identification using pUC/M13 primers. The viral titer was determined by the viral plaque assay according to Invitrogen's instructions.
-
Sf9 cells in the supernatant were infected with MOI of 4, 6 and 8, respectively, and cultured in the complete medium at 27℃ for 1-4 days, harvested after 24 h, 48 h, 72 h and 96 h. The bacmid-transfected Sf9 cells were taken as negative control to examine the P8 protein expression.
-
The infected Sf9 cells were centrifuged at 2 000 ×g for 5 min, and the supernatant was discarded. After being washed with 1 ×PBS (pH 7.3) buffer for 3 times, 6 ×SDS loading buffer was added. The proteins were separated by 10% SDS-PAGE electrophoresis, and observed by Coomassie Brilliant Blue staining.
-
Method of Sambrook [19] with minor improvements was used. After SDS-PAGE electrophoresis, the proteins were transferred to PVDF membrane by electrophoretic transfer. The membrane was incubated overnight at 4℃in blocking buffer, added with the prepared RGDV P8 protein antiserum (1:300 diluted in TTBS containing 5% skim milk powder) as the primary antibody, and kept at 37℃ for 1h. Then the membrane was washed, and alkaline phosphatase-goat anti-rabbit IgG (IgG-AP) (1:10 000) was added, followed by warm bath at 37℃ for 1h. After sufficient rinsing, the membrane was immersed in BCIP/NBT solution until distinct bands could be observed, and then put into distilled water to terminate the reaction. Photos were taken after air drying.
-
At different times after inoculation, Sf9 cells on coverslips were washed twice with 0.01 mol/L PB (pH 7.3) that contained 0.15 mol/L NaCl (PBS) and fixed in cold acetone for 2 to 3 min. The fixed cells were washed twice with PBS and incubated for 40 to 50 min at 37℃ with rabbit antibodies against RGDV P8 protein which had been appropriately diluted with PBS. The cells were then washed twice with PBS, incubated with fluorescein isothiocyanate-conjugated (FITC-conjugated) goat antibodies against rabbit IgG, and diluted 1:120 in PBS (pH 7.3) that contained 3% bovine serum albumin. Excess staining was removed by dipping the cells in fresh PBS twelve times in succession. The coverslips were mounted in PBS that contained 50% glycerol on glass slides and examined under a fluorescence microscope (AH2-FL; Olympus, Tokyo, Japan). Coverslips containing mock-infected cells were included in every experiment and processed in parallelwith infected cells.
Plasmids and reagents
Isolates and cells
Construction of the baculovirus transfer vector
Acquisition and identification of the recombinant baculovirus DNA
Insect cell transfection and recombinant virus identification
Expression of recombinant virus in Sf9 cells
SDS-PAGE analysis
Western blotting analysis
Immunofluorescence microscopy
-
The S8 gene that has been cloned into the pMD-18T vector was ligated into pFastBacTM1 after restriction enzyme digestion, and the ligation product was transformed into DH5α competent cells. White colonies were selected to verify and screen positive clones by BamH Ⅰ and Xho Ⅰ digestion. Digestion fragments of about 4 775 bp and 1 278 bp were observed, which were the pFastBacTM1 vector and the target gene, respectively. The sequencing result indicated that the insertion direction and reading frame of S8 gene were correct, and the recombinant baculovirus transfer vector pFB-S8 was constructed successfully (Fig. 1A).
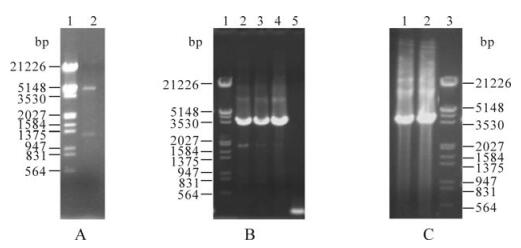
Figure 1. The construction of recombinant virus. A: The restriction enzyme digestion of the recombinant transfer plasmid pFB-S8. 1, λDNA/EcoR Ⅰ + Hind Ⅲ Marker; 2, Plasmid pFB-S8 digested with BamH Ⅰ and Xho Ⅰ. B: PCR analysis of recombinant bacmids. 1, λDNA/EcoR Ⅰ+Hind Ⅲ Marker; 2, 3 and 4, rbpFB-S8 PCR product; 5, Bacmid in blue bacteria PCR product. C: PCR analysis of recombinant viral clones. 1 and 2, rvpFB-S8; 3, λDNA/EcoR Ⅰ+Hind Ⅲ Marker.
-
The recombinant baculovirus transfer vector pFB-S8 after PCR identification was transformed into E. coli DH10Bac (containing bacmids and helper plasmids). White colonies were selected to extract the recombinant bacmid DNA for PCR detection with pUC/M13 primers. There was a specific band at about 3600 bp, which was the sum of the 2 300 bp inner transposon fragment and the 1 278 bp outer coat protein gene fragment. The fragment amplified from the non-recombinant bacmids (extracted from blue colonies) was about 300 bp (Fig. 1B), indicating that the recombinant bacmid rbpFB-S8 had been constructed successfully.
-
Under serum-free conditions, Cellfectin reagent was used to mediate the transfection of Sf9 cells in the logarithmic growth phase by the recombinant bacmid DNA with the right size and structure, and the cellular supernatant was collected 3 days later. The cellular supernatant was used to superinfect Sf9 cells in the logarithmic growth phase to amplify recombinant virus, and the viral genomic DNA was extracted from cells of the infected recombinant viruses. PCR amplification using pUC/M13 primers produced a specific fragment of about 3600 bp, which was consistent with the size of the target fragment, proving that the recombinant baculovirus rvpFB-S8 with the RGDV outer coat protein gene had been amplified in the insect cells (Fig. 1C).
-
The recombinant viruses were inoculated at different MOIs, the cells were collected at different time points, and the total protein was extracted for SDS-PAGE electrophoresis and stained with Coomassie Brilliant Blue R-250 staining. Preliminary test results showed that the protein was not sufficiently expressed within a short time of infection (24 h, 48 h). But if the infection time was too long (96 h), the infected cells might stop growing and even be lysed, so that the expressed protein might be destroyed and lost into the medium. Accordingly, cells collected after infection for 72 h were used for protein expression analysis. SDS-PAGE electrophoresis results showed that, compared with the negative control, a specific band of about 47 kDa was observed in the rvpFB-S8-infected cells, which was consistent with the size of the expected protein. And there were specific expression bands observed when Sf9 cells were infected with the MOI of 4, 6 and 8, but the expression level was highest at MOI of 8, indicating that the outer coat protein gene S8 had been expressed in Sf9 cells (Fig. 2A).
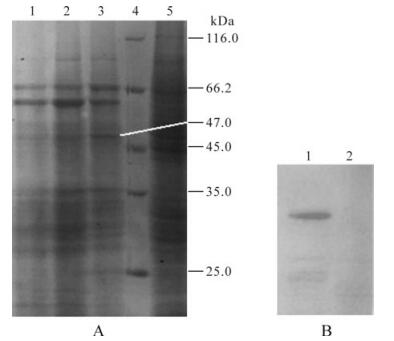
Figure 2. SDS-PAGE and Western blotting analysis of the expression of P8 protein in Sf9 cells. A: SDS-PAGE analysis of the expression of rvpFB-S8 in Sf9 cells. 1, Sf9 cells infected with MOI of 4; 2, Sf9 cells infected with MOI of 6; 3, Sf9 cells infected with MOI of 8; 4, Protein molecular weight marker; 5, Uninfected Sf9 cells (control). B: Western blotting analysis of the expressed protein. 1, Sf9 cells infected with rvpFB-S8; 2, Sf9 cells (control).
-
To further confirm whether the obtained protein was the expression product of the target gene, the polyclonal rabbit antiserum prepared with prokaryotic expression and the alkaline phosphatase-goat anti-rabbit IgG were used as the primary antibody and secondary antibody, respectively, to perform Western blotting for the analysis of the expression product of the recombinant baculovirus rvpFB-S8 in Sf9 cells. Results showed that one band was detected in cells infected with recombinant virus, but none in the control lane, indicating that the recombinant virus expressed the inserted foreign gene successfully (Fig. 2B).
-
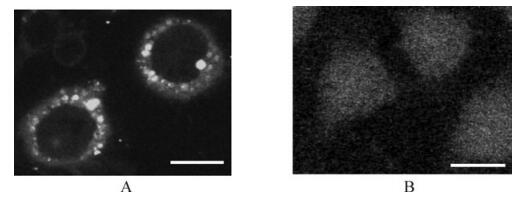
To investigate the subcellular localization of RGDV P8 protein, Sf9 cells were inoculated with recombinant baculovirus and incubated for various periods. Then the cells were examined by immunoblotting with P8-specific antibodies. Immunofluorescence staining of the cells grown on coverslips revealed that, at 72 h p.i., P8 protein of RGDV formed distinct punctate structures in the cytoplasm of cells (Fig. 3).
Construction of the baculovirus transfer vector
Acquisition and identification of the recombinant baculovirus DNA
Insect cell transfection and recombinant virus amplification
SDS-PAGE electrophoresis analysis of the recom-binant baculovirus expression product
Western blotting analysis
P8 protein of RGDV can form inclusions alone in vivo
-
Bac-to-Bac system is a new baculovirus-insect expression system developed in recent years. Insect cells acting as the host undergo post-translational chemical modifications of many higher eukaryotic cells, which is different from the bacterial system that doesn't experience these modifications. We have tried to express the RGDV outer coat protein gene in E.coli using the prokaryotic expression system, and the expression products existed in the form of insoluble inclusion body [4], losing the biologic activity they should have. This is not only closely related to the physical and chemical properties and structure of the expressed protein itself, but is also related to the fact that many proteins synthesized by bacterial cells may not undergo proper folding. Proteins expressed in insect cells are more likely to retain their antigenicity and solubility by correct folding because of their extensive and more accurate post-translational modifications [20], thus providing the possibility for the subsequent research of protein structure and function.
The virus major capsid protein has the characteristics of self-assembly into virus-like particles (VLPs). Most viruses can achieve self-assembly effectively in eukaryotic expression systems. For example, Orthoreovirus [10], Rotavirus [22] and Bluetongue virus [9] in the Reoviridae family can assemble into single-, double-or triple-layered virus-like particles in the presence of different proteins. Research on P3 and P8 proteins of Rice dwarf virus (RDV), a member of the genus Phytoreovirus, also showed that double-shelled virus-like particles can be obtained by co-expression of RDV P3 and P8 proteins in transgenic rice and baculovirus expression systems [5, 25]. Assembly of viruses is an important step in viral replication and proliferation. Although there are few researches on the functions of RGDV gene fragments encoding proteins, favorable progress has been achieved by predicting the functions with the method of comparative genomics and then verifying the result with relevant experiments, such as S1 fragment encoding RNA-dependent RNA polymerases (RdRp) [13], S5-encoded minor core protein having the guanylyltransferase activity [8], and S11 encoding an RNA silencing suppressor [11], etc. In the genus Phytoreovirus, the structure and assembly mechanism of RDV particles have been most clearly described. The core particles and particles in the outer shell of RGDV and RDV can exchange with each other for heterologous recombination [21], and co-expression of the RDV core protein P3 and RGDV outer coat protein P8 in insect cells can produce chimeric virus-like particles [12], indicating that RGDV particles and RDV particles have a similar configuration, and employ a similar assembly mechanism. In this experiment, P8 protein of RGDV was successfully expressed in Sf9 cells, and immunofluorescence microscopy showed that P8 protein of RGDV formed punctate structures in the cytoplasm of Sf9 cells, laying a foundation for research of the specific function of a single protein and the interaction between various proteins.
Bioinformatics analysis of RGDV showed that the 427 amino acid residues of RGDV P8 protein could constitute two functional domains. The domain composed of highly conservative region that is located on the surface of P8 trimer and dominated by α-helix participated in the molecular interaction between P8-P8 and between P8-P3. Especially, the 1st, 29~55th, 77th, and 95~112th amino acids at N-terminal might be the functional region that generated virus-like particles [12]. Therefore, with the use of the baculovirus expression system and the amino acid deletion mutation technology, performing deletion mutation analysis of the possible functional region of P8 and conducting research on in vivo and in vitro recombination of virus-like particles will help to reveal the effect of interactions between homogeneous (P3-P3 and P8-P8) or heterogeneous (P3-P8) protein subunits on the assembly of virus-like particles.














 DownLoad:
DownLoad: