-
Dear Editor,
Arthropod-borne viruses are a group of the most important emerging pathogens. They cause a range of diseases in vertebrate hosts and threaten human health (Gan and Leo 2014). The global distribution of arboviruses is associated with the vector which is strongly affected by changes in environmental conditions. Dengue virus (DENV) and Chikungunya virus (CHIKV), which cause high annual infected cases and have an increasing geographic distribution, are transmitted by Aedes spp. mosquitoes, in particular Ae. albopictus and Ae. Aegypti (Presti et al. 2014; Higuera and Ramírez 2018). Although, the main vector of dengue virus, Ae. aegypti, was not detected in Iran, other possible important vectors such as Ae. Albopictus and Ae. unilineatus were recorded (Doosti et al. 2016; Yaghoobi-Ershadi et al. 2017). West Nile Virus (WNV), a member of the genus Flaviviruses, is one of the most widespread arboviruses (Chancey et al. 2015). The epidemiological evidence of WNV in different hosts in Iran was found (Bagheri et al. 2015), and the circulation of WNV in the main vector, Culex pipiens s.l. and Cx. pipiens, has been proved (Shahhosseini et al. 2017). Due to limited information on the situation of CHIKV, DENV and WNV in Iran, we performed a wide geographical investigation to determine the prevalence of IgG specific antibodies in human samples as well as the genome of WNV, CHIKV and DENV in mosquitoes.
From September 2017 to June 2018, a total of 1257 serum samples were collected in six provinces (south area: Bushehr, Hormozgan, Sistan & Baluchestan, Khuzestan; north area: Gilan, Mazandaran, ) (Fig. 1). Patients with previous history of occasional fever, headache, body ache, arthralgia or rash illness and age over 15 years were included. Euroimmune ELISA kits were used to detect the IgG antibodies against WNV, DENV and CHIKV (Andayi et al. 2014). Adult female mosquitoes and larvae (10, 488 adult mosquitoes and larvae) were collected from 190 pools in the above six provinces between March 2017 to March 2018 using light traps (Fig. 1). Morphological identification of mosquitoes was carried out using the keys of Becker et al. (Schaffner et al. 2001; Becker 2010). RNA was extracted and Altona Real-time PCR kits were used to detect and amplify the genome of WNV, DENV and CHIKV (see Supplementary Material for detailed methods). All statistical analyses were conducted using IBM SPSS Statistics version 22 (IBM Corp, Armonk, NY). Logistic regression analysis using single and multiple univariate analysis was used to determine the relationship between the variables and seroreactivity of WNV, DENV and CHIKV.
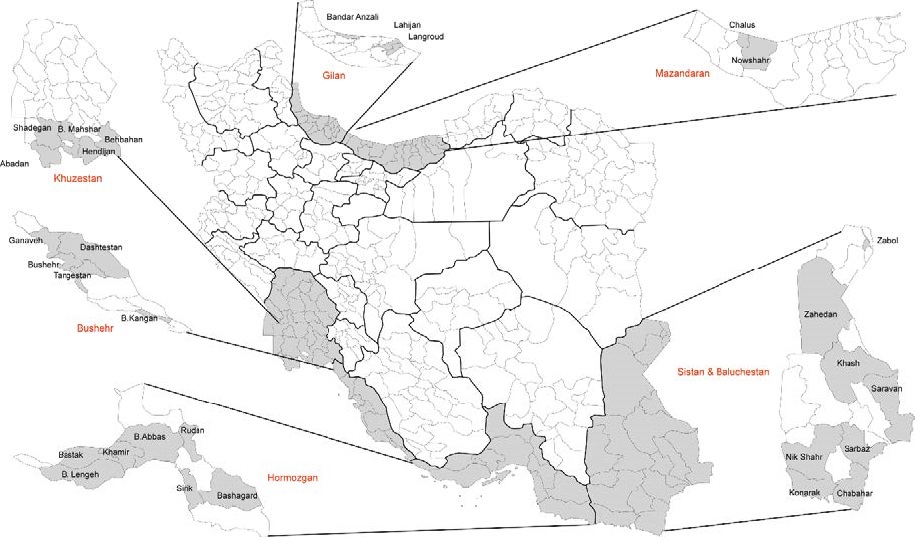
Figure 1. The map of the sampling regions in this cross-sectional study. The sampling areas are highlighted in grey.
The demographic characteristics of study participants are shown in Table 1. Results showed that 236 (18.8%) and 74 (5.9%) serum samples were reactive for WNV and DENV IgG antibodies, whereas IgG antibodies against CHIKV (22, 1.8%) were lower than WNV and DENV. According to the univariate analysis, WNV seroprevalence were significantly associated with age (45-54 vs. 1-24, OR = 1.77, 95% C.I.: 1.03-3.02, P < 0.05; ≥ 55 vs. 1-24, OR=1.93, 95% C.I.: 1.15-3.26, P < 0.05), and residential areas (Gilan vs. Bushehr; OR = 0.39, 95% C.I.: 0.12-0.71, P < 0.001). Also, DENV and CHIKV seroprevalences were significantly associated with residential areas (Hormozgan vs. Bushehr; DENV, OR = 0.09, 95% C.I.: 0.018-0.95, P < 0.05; CHIKV, OR =8.5, 95% C.I.: 2.287-33.01, P < 0.05) (Supplementary Table S1).
Characteristic Total Count Percent (%) Age (years) (n = 1257) 1-24 203 16.2 25-34 399 31.7 35-44 263 20.9 45-54 187 14.9 ≥ 55 205 16.3 Gender (n = 1207) Female 734 60.8 Male 473 39.2 Residential area (n = 1257) Bushehr 414 32.9 Hormozgan 153 12.2 Sistan & Baluchestan 230 18.3 Gilan 165 13.1 Mazandaran 95 7.6 Khuzestan 200 15.9 Travelling history (n=980) Yes 230 23.5 No 750 76.5 Seroprevalence (n = 1257) West Nile virus (WNV) 236 18.8 Dengue virus (DENV) 74 5.9 Chikungunya virus (CHIKV) 22 1.8 Coinfection (n = 1257) WNV + DENV 67 5.3 WNV + CHIK 14 1.1 DENV + CHIK 4 0.3 DENV + WNV + CHIK 4 0.3 Table 1. Study populations' demographic characteristics and WNV, DENV and CHIK IgG seroprevalence.
West Nile virus Characteristic Negative cases (N) Positive cases (N) Seropositivity (%) OR 95%CI P-value Age (years) 1-24 176 27 13.3 Ref 25-34 325 74 18.5 1.48 0.92 2.39 0.1 35-44 215 48 18.25 1.45 0.87 1.42 0.15 45-54 147 40 21.4 1.77 1.03 3.02 0.03 ≥ 55 158 47 22.9 1.93 1.15 3.26 0.01 Gender Female 590 144 19.6 Ref Male 387 86 18.2 0.91 0.67 1.2 0.53 Residential area Bushehr 330 84 20.3 Ref Hormozgan 118 35 22.9 1.16 0.74 1.8 0.5 Sistan & Baluchestan 189 41 17.8 0.85 0.56 1.28 0.44 Gilan 150 15 9.1 0.39 0.121 0.71 0.000 Mazandaran 83 12 12.6 0.56 0.29 1.08 0.08 Khuzestan 151 49 24.5 1.27 0.85 1.9 0.23 Travelling history (780) Yes 174 56 24.3 Ref No 601 149 19.9 1.29 0.91 1.8 0.14 Dengue virus Age (years) 1-24 194 9 4.4 Ref 25-34 385 14 3.5 0.78 0.33 1.68 0.57 35-44 243 20 7.6 1.77 0.79 3.98 0.16 45-54 177 10 5.3 1.2 0.48 3.8 0.67 ≥ 55 184 21 10.2 2.4 1.06 5.5 0.21 Gender Female 700 34 4.6 Ref Male 435 38 8.0 1.79 1.1 2.94 0.01 Residential area Bushehr 388 26 6.3 Ref Hormozgan 152 1 0.7 0.09 0.018 0.73 0.02 Sistan & Baluchestan 220 10 4.3 0.67 0.32 1.1 0.3 Gilan 154 11 6.7 1.06 0.51 2.2 0.86 Mazandaran 90 5 5.3 0.82 0.31 2.2 0.7 Khuzestan 179 21 10.5 1.75 0.95 3.19 0.06 Travelling history (780) Yes 213 17 7.4 Ref No 709 41 5.5 1.38 0.76 2.47 0.28 Chikungunya virus Age (years) 1-24 202 1 0.5 Ref 25-34 388 11 3.0 5.78 0.73 44.6 0.09 35-44 261 2 0.8 1.5 0.13 17.1 0.72 45-54 183 4 2.1 4.4 0.48 39.8 0.18 ≥ 55 201 4 2.0 4.02 0.44 36.2 0.21 Gender Female 720 14 1.9 Ref Male 466 7 1.5 0.77 0.3 1.9 0.58 Residential area Bushehr 411 3 0.7 Ref Hormozgan 144 9 5.9 8.5 2.287 33.01 0.001 Sistan & Baluchestan 228 2 0.9 1.2 0.199 7.245 0.841 Gilan 160 5 3.0 4.2 1.011 18.123 0.048 Mazandaran 94 1 1.1 1.4 0.150 14.167 0.74 Khuzestan 198 2 1.0 1.3 0.22 8.3 0.72 Travelling history (780) Yes 225 5 2.2 Ref No 741 9 1.2 1.83 0.6 5.5 0.28 Notes: Ref, the data was set as reference Table S1. Univariate analysis of WNV, DENV and CHIK IgG seroprevalence by age, gender, residential area and travelling history to outside of country.
Multiple univariate analysis showed significant association between WNV seroreactivity and age (45-54 vs. 1-24, OR = 1.82, 95% C.I.: 1.8-1.02, P < 0.05; ≥ 55 vs. 1-24, OR =3.52, 95% C.I.: 1.98-6.26, P < 0.01). The association was also found between WNV seroreactivity and residential areas (Gilan and Khuzestan vs. Bushehr; OR = 0.25, 95% C.I.: 0.121-0.52, P < 0.001 and OR = 1.57, 95% C.I.: 1.01-2.45, P < 0.05). Also, DENV and CHIKV seroprevalences were significantly associated with residential areas (Hormozgan vs. Bushehr; OR = 0.12, 95% C.I.: 0.18-0.95 and OR: 9.0, 95% C.I.: 2.21-36. 6, P < 0.05) (Table 2).
West Nile virus Characteristic Negative cases (N) Positive cases (N) Seropositivity (%) OR 95%CI Adjusted P-value Age (years) 1-24 176 27 13.3 Ref 25-34 325 74 18.5 1.35 0.82 2.21 0.22 35-44 215 48 18.25 1.45 0.82 2.42 0.152 45-54 147 40 21.4 1.82 1.8 1.02 0.04 ≥ 55 158 47 22.9 3.52 1.98 6.26 0.00 Gender Female 590 144 19.6 Ref Male 387 86 18.2 0.732 0.497 1.04 0.53 Residential area Bushehr 330 84 20.3 Ref Hormozgan 118 35 22.9 1.29 0.8 2.07 0.29 Sistan & Baluchestan 189 41 17.8 0.85 0.56 1.28 0.44 Gilan 150 15 9.1 0.25 0.121 0.52 0.000 Mazandaran 83 12 12.6 0.56 0.29 1.06 0.08 Khuzestan 151 49 24.5 1.57 1.00 2.45 0.04 Dengue virus Age (years) 1-24 194 9 4.4 Ref 25-34 385 14 3.5 0.63 0.26 1.5 0.3 35-44 243 20 7.6 1.15 0.49 2.7 0.73 45-54 177 10 5.3 0.65 0.24 1.7 0.4 ≥ 55 184 21 10.2 2.19 0.92 5.19 0.07 Gender Female 700 34 4.6 Ref Male 435 38 8.0 1.17 0.67 2.03 0.56 Residential area Bushehr 388 26 6.3 Ref Hormozgan 152 1 0.7 0.12 0.18 0.95 0.04 Sistan & Baluchestan 220 10 4.3 0.71 0.32 1.55 0.4 Gilan 154 11 6.7 0.77 0.32 1.8 0.54 Mazandaran 90 5 5.3 0.91 0.31 2.45 0.84 Khuzestan 179 21 10.5 1.9 0.95 3.7 0.057 Chikungunya virus Age (years) 1-24 202 1 0.5 Ref 25-34 388 11 3.0 5.4 0.68 43.2 0.1 35-44 261 2 0.8 2.07 0.18 23.7 0.55 45-54 183 4 2.1 5.7 0.54 60.3 0.14 ≥ 55 201 4 2.0 4.9 0.44 53.7 0.18 Gender Female 720 14 1.9 Ref Male 466 7 1.5 1.05 0.37 3.02 0.91 Residential area Bushehr 411 3 0.7 Ref Hormozgan 144 9 5.9 9.0 2.21 36.6 0.001 Sistan & Baluchestan 228 2 0.9 1.3 0.21 8.15 0.77 Gilan 160 5 3.0 4.2 0.8 22.86 0.08 Mazandaran 94 1 1.1 1.4 0.14 13.89 0.74 Khuzestan 198 2 1.0 1.25 0.19 8.1 0.8 Notes: Ref, the group was set as reference. Table 2. Multiple univariate analysis for the assessment of factors associated with these arboviruses seropositivity.
The mosquitoes collected in this study belonged to 4 genera and 23 species, including 13 Culex, 8 Aedes, 1 Culiseta and 1 Uranotaenia genera (Supplementary Table S2). In Sistan and Baluchestan Province, the highest detection frequency species of mosquito larvae and adults were Cx. quinquefasciatus (44%) and Ae. vexans (78%). The species of mosquito larvae with highest detection frequency in other regions were: Cs. Longiareolata in Hormozgan, Cx. pipiens complex in Khuzestan, Gilan and Bushehr. Ae. Albopictus species was only detected in Sistan and Baluchestan Province, but Ae. Caspius and Cx. pipiens complex were detected in all of the provinces. All species were screened for the presence of WNV, CHIKV and DENV, but RNA of three arboviruses were not detected.
Species Provinces and Counties Total (N/%) Assay Sistan and Baluchestan Konarak Sarbaz Khash Chabahar Saravan Zahedan Nikshahr Zabol Larvae/Adults L A L A L A L A L A L A L A L A L A Cx. perexiguus 0 ND 5 ND 0 ND 70 ND 30 ND 7 ND 9 ND 0 ND 121 (4.64) ND rRT-PCR Cx. pipiens complex 0 0 0 ND 0 ND 36 29 1 67 18 ND 0 ND 0 0 55 (2.1) 96 (5.1) Cx. theileri 0 ND 0 ND 0 ND 38 ND 0 ND 99 ND 9 ND ND ND 146 (5.6) ND Cx. laticinctus ND 0 ND ND ND ND ND 19 ND 10 ND ND ND ND ND 0 ND 29 (1.5) Cx. mimeticus 0 ND 0 ND 0 ND 4 ND 71 ND 0 ND 0 ND ND ND 75 (2.9) ND Cx. sinaiticus 0 ND 10 ND 0 ND 0 1 0 3 0 ND 0 ND ND 0 12 (0.46) 4 (0.2) Cx. sitiens 271 ND 0 ND 0 ND 113 ND 54 ND 0 ND 2 ND ND ND 438 (16.8) ND Cx. tritaeniorhynchus 5 0 22 ND 0 ND 184 5 93 2 0 ND 7 ND ND 0 311 (11.9) 7 (2.4) Cx. quinquefasciatus 0 ND 300 ND 0 ND 270 ND 410 ND 94 ND 71 ND ND ND 1145 (44) ND Cx. bitaeniorhynchus 0 ND 0 ND 0 ND 0 ND 1 ND 0 ND 0 ND ND ND 18 (0.7) ND Cs. longiareolata 25 0 1 ND 118 0 12 0 0 3 1 ND 2 ND ND 0 159 (6.1) 3 (0.2) Ae. caspius 0 3 0 ND 0 ND 104 150 0 1 9 ND 0 ND ND 45 113 (4.34) 199 (10.6) Ae. vexans 0 75 0 ND 0 ND 2 1390 0 0 0 ND 0 ND ND 0 2 (0.08) 1465 (78) Ae. falvescens 0 ND 0 ND 0 ND 0 ND 0 ND 2 ND 0 ND ND ND 2 (0.08) ND Ae. caballus 0 ND 0 ND 0 ND 1 ND 0 ND 0 ND 0 ND ND ND 1 (0.04) ND Ae. unlineatus ND 0 ND ND ND ND ND 1 ND 0 ND ND ND ND ND ND ND 1 (0.1) Ae. detritus ND 12 ND ND ND ND ND 18 ND 0 ND ND ND ND ND 0 ND 30 (1.6) Ae. albopictus 0 0 3 ND 0 ND 0 6 0 0 0 ND 2 ND ND 0 5 (0.19) 6 (0.3) Total 301 128 341 ND 118 0 834 1619 677 86 230 ND 102 ND 0 45 2603 1878 Species Hormozgan Total (N/%) rRT-PCR Bandar Abbas Bashagard Sirik Rudan Bastak Bandar Khamir Bandar Lengeh L A L A L A L A L A L A L A L A Cx. perexiguus 4 ND 0 ND ND ND 0 ND ND ND 0 ND 0 ND 4 (0.3) ND Cx. arbieeni 0 ND 2 ND ND ND ND ND ND ND 0 ND 0 ND 2 (0.1) ND Cx. laticinctus ND 0 ND 2 ND 0 ND 0 ND 0 ND 0 ND 0 ND 2 (3.2) Cx. pipiens complex 3 0 215 19 ND 0 3 0 ND 0 11 0 0 0 235 (15.7) 19 (30.1) Cx. theileri 0 ND 0 ND ND ND 0 ND ND ND 0 ND 155 ND 155 (10.5) ND Cx. mimeticus 1 ND 77 ND ND ND 3 ND ND ND 0 ND 0 ND 81 (5.5) ND Cx. sinaiticus 0 0 52 3 ND 0 0 0 ND 0 0 0 0 0 52 (3.5) 3 (4.8) Cx. tritaeniorhynchus 10 8 61 1 ND 0 0 0 ND 0 34 0 37 0 142 (9.6) 9 (14.3) Cx. quinquefasciatus 0 ND 125 ND ND ND 0 ND ND ND 1 ND 9 ND 135 (9.1) ND Cs. longiareolata 0 ND 648 ND ND ND 0 ND ND ND 0 ND 0 ND 648 (43.8) ND Ae. caspius ND 0 ND 1 ND 5 ND 7 ND 0 ND 5 ND 5 ND 23 (36.5) Ae. vexans ND 0 ND 2 ND 0 ND 0 ND 1 ND 0 ND 0 ND 3 (4.8) Ae. vittatus ND 0 ND 0 ND 0 ND 0 ND 0 ND 0 ND 4 ND 4 (6.3) Ae. caballus 0 ND 1 ND ND ND 0 ND ND ND 0 ND 0 ND 1 (0.1) ND Total 18 8 1207 28 ND 5 6 7 ND 1 46 5 201 9 1478 63 Species Bushehr Total (N/%) rRT-PCR Tangestan Dashtestan Bandar Ganaveh Bushehr Bandar Kangan L A L A L A L A L A L A Cx. perexiguus 22 ND 20 ND ND ND 8 ND 1 ND 45 (4.8) ND Cx. hortensis 1 ND 0 ND ND ND 0 ND 0 ND 1 (0.1) ND Cx. pusillus ND ND 0 ND ND ND 63 ND 0 ND 63 (5.9) ND Cx. pipiens complex 106 0 0 0 ND 6 126 ND 282 28 522 (49.2) 34 (12.1) Cx. theileri 0 ND 0 ND ND ND 16 ND 0 ND 16 (1.5) ND Cx. mimeticus 22 ND 0 ND ND ND 0 ND 0 ND 22 (2.1) ND Cx. laticinctus 0 ND 2 ND ND ND 0 ND 0 ND 2 (0.2) ND Cx. sinaiticus ND 1 ND 0 ND 0 ND ND ND 0 ND 1 (0.4) Cx. sitiens 0 ND 0 ND ND ND 56 ND 0 ND 56 (5.3) ND Cx. tritaeniorhynchus 0 ND 1 ND ND ND 0 ND 4 ND 5 (0.5) ND Cx. quinquefasciatus 21 ND 1 ND ND ND 33 ND 25 ND 80 (7.5) ND Cs. longiareolata 50 ND 0 ND ND ND 3 ND 9 ND 62 (5.8) ND Ur. unguiculata 0 ND 2 ND ND ND 6 ND 0 ND 8 (0.8) ND Ae. caspius 15 22 0 86 ND 0 150 117 0 2 165 (15.6) 227 (80.5) Ae. vexans 7 2 0 0 ND 0 0 ND 0 0 7 (0.7) 2 (0.7) Ae. detritus ND 0 ND 0 ND 0 ND ND ND 18 ND 18 (6.3) Total 244 25 26 86 ND 6 461 117 321 48 1060 282 Species Khuzestan Total (N/%) rRT-PCR Abadan Behbahan Shadegan Bandar Mahshahr Hendijan L A L A L A L A L A L A Cx. perexiguus 0 ND 1 ND 0 ND 0 ND 0 ND 1 (0.2) ND Cx. pusillus 0 ND 13 ND 12 ND 0 ND 0 ND 25 (5.1) ND Cx. pipiens complex 31 ND 70 ND 86 ND 0 ND 0 ND 187 (33.8) ND Cx. theileri 6 ND 7 ND 2 ND 0 ND 0 ND 15 (1.6) ND Cx. sitiens 6 ND 0 ND 0 ND 0 ND 0 ND 6 (1.1) ND Cx. tritaeniorhynchus 0 ND 112 ND 34 ND 0 ND 11 ND 157 (28.4) ND Cx. quinquefasciatus 0 ND 1 ND 57 ND 0 ND 0 ND 58 (10.5) ND Ur. ungiuculata 0 ND 6 ND 0 ND 0 ND 0 ND 6 (1.1) ND Ae. caspius 20 ND 34 ND 6 ND 41 ND 0 ND 101 (18.2) ND Total 57 ND 232 ND 200 ND 41 ND 11 ND 553 ND Species Gilan Total (N/%) rRT-PCR Langarud Bandar Anzali Lahijan L A L A L A L A Cx. perexiguus 11 1 14 4 10 2 35 (11.11) 7 (7.53) Cx. pipiens complex 37 11 57 21 75 24 169 (53.8) 57 (60.2) Cx. theileri 15 6 19 9 10 0 44 (12.42) 15 (16.12) Cx. mimeticus 0 ND 3 ND 1 ND 4 (1.3) ND Cx. sitiens 4 1 7 3 2 0 13 (4.2) 4 (4.3) Cx. tritaeniorhynchus 1 ND 5 ND 2 ND 9 (2.55) ND Cx. hortensis 15 3 20 5 5 2 40 (12.7) 10 (10.75) Cs. longiareolata 1 ND 2 ND 0 ND 3 (0.96) ND Ae. caspius 0 ND 2 ND 0 ND 2 (0.64) - Ae. vexans 0 0 0 1 1 0 1 (0.32) 1 (1.1) Total 84 22 129 43 101 28 314 93 Species Mazandaran rRT-PCR Chalus Nowshahr Total (N/%) L A L A L A Cx. perexiguus 37 11 22 6 59 (10.61) 17 (7.7) Cx. pipiens complex 115 46 158 71 273 (49.1) 117 (52.94) Cx. theileri 45 17 63 24 108 (19.42) 41 (18.55) Cx. mimeticus 2 0 5 2 7 (1.26) 2 (0.91) Cx. sitiens 27 13 71 28 98 (17.63) 41 (18.55) Cx. hortensis 1 0 2 1 3 (0.54) 1 (0.45) Cs. longiareolata 3 1 0 0 3 (0.54) 1 (0.45) Ae. caspius 5 1 0 0 5 (0.9) 1 (0.45) Total 235 89 321 132 556 221 Abbreviation: L: Larvae; A: Adults; rRT-PCR: Real-time Reverse Transcriptase-Polymerase Chain Reaction;
ND: Not determined.Table S2. Distribution of collected mosquito's species in 6 province of Iran.
In our study, there were two groups of cases: the ones who had not travelled to dengue-endemic areas and those who had travelled to east of Asia, Saudi Arabia. That was in parallel with previous studies in Iran (Chinikar et al. 2013; Aghaie et al. 2014). The previous study reported that there was no evidence of DENV seroprevalence in Iran before 2000 (Saidi 1974), but positive cases in this decade have been reported (Aghaie et al. 2014; Heydari et al. 2018; Tavakoli et al. 2020). Our results showed a number of DENV seropositve cases from southern regions, Khuzestan and Bushehr. Those regions are in close proximity to Saudi Arabia and Pakistan, and thousands of Iranian travel there as pilgrims annually, which may increase the probability of DNEV infection. In our report, DNEV seroprevalence was not correlated with patients who had any travel history. It is possible that these cases might be infected through contact with imported cases. Another plausible explanation is that the seropositivity of these cases might be caused by infected vectors. Pakistan country, which is near Sistan and Baluchestan province of Iran, has the largest number of confirmed cases among countries in the Middle East and North Africa (MENA) during all DENV outbreaks (Chinikar et al. 2013; Humphrey et al. 2016). And our report has shown the potential for the presence of DENV vector, Ae. albopictus and Ae. unilineatus, in Iran (Doosti et al. 2016; Yaghoobi-Ershadi et al. 2017).
An earlier report showed that the prevalence of WNV in humans in West Azerbaijan and Khuzestan was 0% (Saidi et al. 1976). However, several studies have shown the prevalence of WNV in humans and in mosquitoes (Culex and Aedes) in recent years (Chinikar et al. 2012; Shahhosseini et al. 2017). In the studies reported in 2010 and 2016, the prevalence of WNV in Khuzestan and Sistan and Baluchestan provinces were 5% and 17.96%, respectively (Sharifi et al. 2010; Aghaie et al. 2016). In a recent study, the seroprevalence of WNV in Khuzestan Province is 23.8% (Kalantari et al. 2019). Another study in the northwest of Iran showed the presence of WNV RNA in Ae. Caspius, a vector of WNV (Bagheri et al. 2015). Also, the evidence of WNV infection in mosquitos, such as Cx. pipienss.l, was found in Gilan, Mazandaran, Golestan and East Azerbaijan (Eybpoosh et al. 2019). Despite the evidence for existence and circulation of WNV, no clinical cases have been described in Iran until now. Our data showed that WNV IgG was positive in patients, but WNV RNA was not detected in vectors.
As the first study of Iran, our results showed that CHIKV seroprevalence was about 1.8% for humans, but there was no RNA detected in mosquitoes, which demonstrated that individuals might have likely been only exposed to CHIKV. Serologic evidence of CHIKV transmission has been identified in the countries surrounding the Red Sea, such as Pakistan (Ali and Dasti 2018) and Saudi Arabia (Hussain et al. 2013). Also, a newly CHIKV imported case from Sistan and Baluchistan Province of Iran was reported (Pouriayevali et al. 2019). In this report, the patient had a recent travel history to Pakistan, where a widespread epidemic of the disease was ongoing at the time of the study.
In recent study, age showed independently association with WNV and CHIKV seropositivity, and a significant association of WNV seroreactivity with the increase of age was found (Mease et al. 2011; Ang et al. 2017; Shaibi et al. 2017; Humphrey et al. 2019). The observed rate was higher in the people with 45 or older compared to those who are below 45, which may be related to a higher probability of exposure to WNV among older people in life period. These findings are consistent with another study (Gómez-Dantés and Willoquet 2009). A significant relationship was found between the residential area and WNV/CHIKV seroreactivity. People residing in Gilan Province had the lowest seroprevalence of WNV, but Gilan and Hormozgan had the highest seroprevalence of CHIKV antibodies compared with other regions. This is in agreement with the studies elsewhere (Ang et al. 2017; Vongpunsawad et al. 2017).
In conclusion, our results revealed the seroprevalence of WNV, CHIKV and DENV in human population in Iran and no proof of viral RNAs was presence in vectors. Gilan and Hormozgan areas were high risk regions and the elderly persons were at higher risk of getting infected by WNV and CHIKV. These results help us to better understand the epidemiology of the infection and the ecology of the vectors in Iran. Therefore, considering the risk factors identified by this study, we recommend that the prevention and control strategies should be designed in the country.
HTML
-
The authors thank all colleagues for the help in the study design. This research received a specific grant from National Institute for Medical Research Development (Grand No. 940947).
-
The authors declare that they have no conflict of interest.
-
The informed consent have been obtained from all participants and the studies have been approved by the National Institute for Medical Research Development ethics committee (IR.NIMAD.REC.1394.940947).
Conflict of Interest
Animal and Human Rights Statement
-
The online version of this article contains supplementary material, which is available to authorized users.
-
This cross-sectional study was conducted in 6 provinces and 29 counties located in southern, eastern south and north of Iran that as depicted in Fig. 1. All of these data were extracted based on the 2016 general census data. Mazandaran and Gilan as north provinces, Sistan and Baluchestan as the eastern south province, Hormozgan, Bushehr and Khuzestan as southern and south-western provinces were included. Mazandaran and Gilan are among the most densely populated provinces in Iran, have a moderate climate that their maximum temperature is 40.2 ℃ and the minimum is −19 ℃. Sistan and Baluchestan Province has a tropical climate with the maximum and minimum temperatures 43 ℃ and 22 ℃. Hormozgan is south of the country facing Oman and The UAE. This province has a tropical climate with the maximum and minimum temperatures 51 ℃ and 22 ℃. Bushehr Province a Persian Gulf coast of south-western of Iran with a sub- and mild tropical climate, almost Mediterranean. Its maximum and minimum temperature is 50 ℃ and 31 ℃. Khuzestan Province is southwest of the country, bordering Iraq and the Persian Gulf. It is very hot and occasionally humid and summertime temperatures routinely exceed 45 ℃. Regarding the fact that Iran has different types of climate, definitely, sampling time was different between sites in the north and south. In the north, the preferred seasons for sampling are just spring and summer, while in the south, sampling is done in all seasons. Therefore, we selected various provinces with different weather conditions.
-
From September 2017 to June 2018, a total of 1257 serum samples were collected after an agreement with the private and governmental public laboratories at six provinces including; Mazandaran, Gilan, Bushehr, Hormozgan, Sistan-Baluchestan and Khuzestan. Patients consulting private laboratories had heterogeneous conditions and mainly previous history of occasional fever, headache, body ache, arthralgia or rash illness. The eligibility criteria were acceptance to participate in the study and age over 15 years. To calculate the sample size, we estimated a prevalence of 20% with a variation of ± 10 and 80% power and finally cluster sampling was performed. At the time of initial sample collection, a verbal consent was obtained from the participants. For each serum sample, basic demographic information including age, sex, residential area and traveling history was obtained. Sera were stored at -20 ℃ until testing.
-
Commercially available Euroimmune ELISA kits (EUROIMMUN AG, Lübeck, Germany) were used to detect IgG antibodies against WNV, DENV and CHIKV (Andayi et al. 2014). Positive and negative control sera were provided by the National Reference Centre for Arbovirus or by the kits' manufacturers. For each serologic assay, a minimum of three positive controls was included, alongside three negative controls and three blank controls (normal saline), in accordance with the established standard protocols (Jacobson 1998). The assays were carried out at the Virology Department of Tehran University of Medical Science. For each sample, a ratio of the extinction value of the control or patient sample over the extinction value of the calibrator was calculated according to the manufacturer's instructions. Specimens with an optical density (OD) value of ≥ 1.1 were considered positive for WNV, DENV and CHIKV IgG antibodies. An OD value of ≤ 0.8 and < 1.1 was considered as an equivocal result and an OD value < 0.8 was determined to be negative. All samples with borderline results were tested twice.
-
Morphological identification of mosquitoes was carried out using the keys of Becker et al. (Schaffner et al. 2001; Becker et al. 2010). Specimens were identified in Medical Entomology, Department of Medical Entomology and Vector Control, Tehran University of Medical Sciences. Adult females and larvae mosquitoes were collected in six provinces from 232 sites based on previous studies (Doosti et al. 2016; Yaghoobi-Ershadi et al. 2017). The mosquitoes were classified into different species and then pooled according to the collection site, species and day of collection (all of the stages were performed on ice). The samples were then placed into cryovials, immersed in RNase blocking solution, and transported in a liquid nitrogen gaseous phase. Totally, 6212 mosquito larvae and 2668 adults were collected and pooled by sampling site, date and taxon comprising between 1 to 20 specimens per pool. Finally, 290 pools were kept at a -70 ℃ freezer for further analysis.
-
Mosquito pools were placed in chilled 15-mL falcons with 1 mL of cooled PBS then were homogenized using glass beads and vortexing for about 1 min. Mosquito homogenates were centrifuged for 5 min at 2500 × g at 4 ℃ and supernatants were collected. RNA was extracted from 200 μL of each mosquitoes' homogenate using the NucleoSpin® RNA Kit (MACHEREY-NAGEL GmbH & Co. KG, Germany) according to the manufacturer's instructions.
-
Commercially Altona kits (RealStar® Dengue RT-PCR Kit, cat no: 282013, RealStar® WNV RT-PCR Kit, cat no: 321013 and RealStar® Chikungunya RT-PCR Kit, cat no: 012013, Altona Diagnostics GmbH, Germany) were used to detect and amplify DENV, CHIKV and WNV RNAs. The tests were performed using an Applied Biosystem step one plus real-time PCR machine (Applied Biosystem, CA, USA). Amplification of WNV RNA took place in a 50 μL single-tube, 21 μL master mix and 9 μL of extracted sample RNA or serially diluted positive control copy number that was provided by the kits. The cycling conditions consisted of one cycle at 55 ℃ for 10 min, one cycle at 95 ℃ for 2 min, and 40 cycles at 95 ℃ for 15 s, 55 ℃ for 1 min and 72 ℃ for 15 s. The test condition for detection of DENV and CHIKV RNAs was the same as above except for the amount of master mix.
-
All statistical analyses were conducted using IBM SPSS Statistics version 22 (IBM Corp, Armonk, NY). Logistic regression analysis using single and multiple univariate analysis was used to determine the relationship between the variables and seroreactivity for anti-WNV, DENV and CHIKV. Adjusted odds ratios (OR) and 95% confidence intervals (CI) for multiple univariate analysis was used to determine independent factors associated with WNV, DENV and CHIKV seroprevalence. A P-value of less than 0.05 was considered to be statistically significant.












 DownLoad:
DownLoad: