Herpesviruses
Editor(s): Min-Hua Luo, PhD, Professor; Hua Zhu, PhD, Professor; Klaus Früh, PhD, Professor Wuhan Institute of Virology, China; Rutgers-New Jersey Medical School, USA; Oregon Health & Science University, USA
The special issue included perspectives, minireviews, research articles, and brief communications. All manuscripts accepted will be waived for publication charge. And all submitted manuscripts will be processed following the standard journal procedure such as being screened by editors and peer-reviewing.
The special issue included perspectives, minireviews, research articles, and brief communications. All manuscripts accepted will be waived for publication charge. And all submitted manuscripts will be processed following the standard journal procedure such as being screened by editors and peer-reviewing.
-
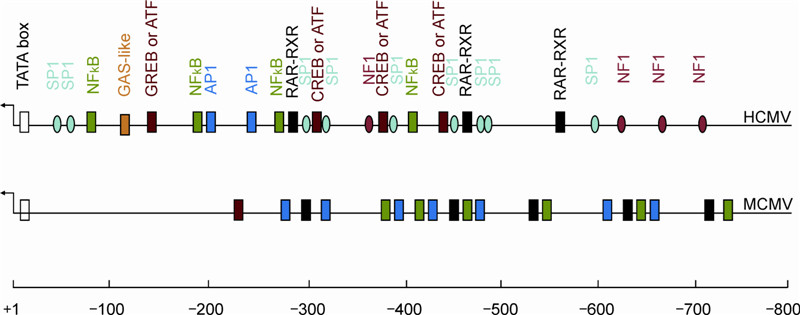
Immediate-Early (IE) gene regulation of cytomegalovirus: IE1-and pp71-mediated viral strategies against cellular defenses
2014, 29(6): 343 doi: 10.1007/s12250-014-3532-9
Received: 27 October 2014 Accepted: 11 November 2014 Published: 25 November 2014Three crucial hurdles hinder studies on human cytomegalovirus (HCMV): strict species specifi city, differences between in vivo and in vitro infection, and the complexity of gene regulation. Ever since the sequencing of the whole genome was fi rst accomplished, functional studies on individual genes have been the mainstream in the CMV fi eld. Gene regulation has therefore been elucidated in a more detailed fashion. However, viral gene regulation is largely controlled by both cellular and viral components. In other words, viral gene expression is determined by the virus-host interaction. Generally, cells respond to viral infection in a defensive pattern; at the same time, viruses try to counteract the cellular defense or else hide in the host (latency). Viruses evolve effective strategies against cellular defense in order to achieve replicative success. Whether or not they are successful, cellular defenses remain in the whole viral replication cycle: entry, immediate- early (IE) gene expression, early gene expression, DNA replication, late gene expression, and viral egress. Many viral strategies against cellular defense, and which occur in the immediate-early time of viral infection, have been documented. In this review, we will summarize the documented biological functions of IE1 and pp71 proteins, especially with regard to how they counteract cellular intrinsic defenses. -
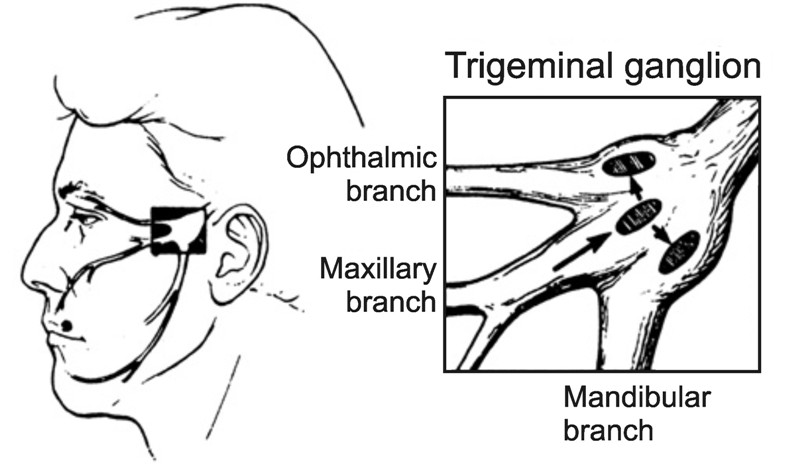
Ocular herpes: the pathophysiology, management and treatment of herpetic eye diseases
2014, 29(6): 327 doi: 10.1007/s12250-014-3539-2
Received: 06 November 2014 Accepted: 13 November 2014 Published: 15 December 2014Herpesviruses are a prominent cause of human viral disease, second only to the cold and influenza viruses. Most herpesvirus infections are mild or asymptomatic. However, when the virus invades the eye, a number of pathologies can develop and its associated sequelae have become a considerable source of ocular morbidity. The most common culprits of herpetic eye disease are the herpes simplex virus (HSV), varicella zoster virus (VZV), and cytomegalovirus (CMV). While primary infection can produce ocular disease, the most destructive manifestations tend to arise from recurrent infection. These recurrent infections can wreck devastating effects and lead to irreversible vision loss accompanied by a decreased quality of life, increased healthcare usage, and signifi cant cost burden. Unfortunately, no method currently exists to eradicate herpesviruses from the body after infection. Treatment and management of herpes-related eye conditions continue to revolve around antiviral drugs, although corticosteroids, interferons, and other newer therapies may also be appropriate depending on the disease presentation. Ultimately, the advent of effective vaccines will be crucial to preventing herpesvirus diseases altogether and cutting the incidence of ocular complications. -

Role of the virion host shutoff protein in neurovirulence of monkey B virus (Macacine herpesvirus 1)
2014, 29(5): 274 doi: 10.1007/s12250-014-3495-x
Received: 19 August 2014 Accepted: 11 October 2014 Published: 22 October 2014Monkey B virus (Macacine herpesvirus 1; BV) is noted for its extreme neurovirulence in humans. Since the vhs protein encoded by the UL41 gene has been shown to be a neurovirulence factor in the related human herpes simplex viruses, the role of the UL41 gene in BV neurovirulence was investigated. BV mutants were constructed that lacked the entire UL41 ORF (Δ41) or had the RNase active site mutated (Δ41A). Neither mutant shut off host protein synthesis, degraded β-actin mRNA, or prevented an IFN-β response, indicating that the vhs protein and its RNase activity are both necessary for these activities. Replication of both mutants in primary mouse cells was impaired and they exhibited a prolonged disease course in mice. Whereas Δ41 infected mice were euthanized for symptoms related to central nervous system (CNS) infection, Δ41A infected mice were euthanized primarily for symptoms of autonomic nervous system dysfunction. While neuroinvasiveness was not affected, lesions in the CNS were more limited in size, anatomical distribution, and severity than for wild-type virus. These results indicate that the vhs protein affects the general replicative efficiency of BV in vivo rather than being a specific neurovirulence factor critical for invasion of or preferential replication in the CNS. -

Varicella zoster virus vaccines: potential complications and possible improvements
2014, 29(5): 265 doi: 10.1007/s12250-014-3516-9
Received: 06 October 2014 Accepted: 12 October 2014 Published: 24 October 2014Varicella zoster virus (VZV) is the causative agent of varicella (chicken pox) and herpes zoster (shingles). After primary infection, the virus remains latent in sensory ganglia, and reactivates upon weakening of the cellular immune system due to various conditions, erupting from sensory neurons and infecting the corresponding skin tissue. The current varicella vaccine (v-Oka) is highly attenuated in the skin, yet retains its neurovirulence and may reactivate and damage sensory neurons. The reactivation is sometimes associated with postherpetic neuralgia (PHN), a severe pain along the affected sensory nerves that can linger for years, even after the herpetic rash resolves. In addition to the older population that develops a secondary infection resulting in herpes zoster, childhood breakthrough herpes zoster affects a small population of vaccinated children. There is a great need for a neuro-attenuated vaccine that would prevent not only the varicella manifestation, but, more importantly, any establishment of latency, and therefore herpes zoster. The development of a genetically-defined live-attenuated VZV vaccine that prevents neuronal and latent infection, in addition to primary varicella, is imperative for eventual eradication of VZV, and, if fully understood, has vast implications for many related herpesviruses and other viruses with similar pathogenic mechanisms.