HTML
-
Human immunodeficiency virus type 1 (HIV-1) is the major causative agent of acquired immunodeficiency syndrome (AIDS) (Mitsuya et al. 1985). Although treatment with highly active antiretroviral therapy (HAART) maintains viral levels below the level of detection and extends a patient's lifespan, these treatments are limited by increasingly frequent drug-resistant mutations, adverse side effects, and the high cost of long-term HAART (Gibson et al. 2019; Margolis et al. 2014). In addition, latent reservoirs of HIV-1-infected cells can persist in a patient, which prevents clearance of the virus even during HAART (Churchill et al. 2016). Therefore, efforts are under way to develop new agents that target key steps in the HIV-1 life cycle.
Numerous naturally derived products have been suggested as inhibitors of HIV-1 infection, including calanolides (reverse transcriptase (RT) inhibitors), kuwanon-L (an RT and integrase (IN) inhibitor), patentiflorin A (an RT and IN inhibitor), and betulinic acid (a viral maturation inhibitor) (Cary and Peterlin 2018). Despite the high potency of these natural products against HIV-1, their use as therapeutics could be restricted due to the difficulty of high-yield purification or large-scale synthesis. In the field of synthetic organic chemistry, unique structure-bearing purine-based compounds, such as seliciclib (also called R-roscovitine or CYC202) (Agbottah et al. 2005), and several purine derivatives bearing aryl groups (Yuan et al. 2007) are known to inhibit HIV-1 infection; therefore, to improve their anti-HIV-1 activity, derivatives of these compounds have been explored. Some derivatives bearing an indole core (which is structurally similar to purine) have been investigated as anti-HIV-1 agents (Xu and Lv 2009). The benzodiazepine derivative (Ro 5-3335) containing benzene has been intensively investigated as a potent antiHIV-1 agent (Hsu et al. 1991). Interestingly, both purine and the benzene-based compounds described above have been observed to impede HIV-1 infection by inhibiting Tat-dependent viral transcription (Mousseau and Valente 2012), which is a pivotal step in viral replication. Therefore, this step is considered to be a potential target for blocking HIV replication. As such, numerous studies have attempted to identify Tat-dependent transcriptional inhibitors such as Durhamycin A (Jayasuriya et al. 2002), betacarboline derivatives (Yu et al. 2004) and DidehydroCortistatin A (dCA) (Mousseau et al. 2012) from naturally derived compounds. Although individual structures containing either indole or benzene substituents have antiHIV-1 effects, the potential anti-HIV-1 effects of compounds containing hybrid benzene–indole cores, such as aristolactams, have not yet been investigated.
Aristolactams bearing unique dibenzo-indole cores are recognized as an important family of alkaloids due to their potent biological activities, including anti-inflammatory, antiplatelet, anti-mycobacterial, neuro-protective, and antitumor effects. Aristolactams have been isolated from several plant species and used as traditional medicines (Chia et al. 2000; Kim et al. 2004; Kumar et al. 2003; Tsai et al. 2005; Zhang et al. 2007). Several aristolactam derivatives have been successfully synthesized, and some of these have exhibited potent biological activity; Structure-activity relationship studies have explored the chemistry needed to attain specific biological activities in an effort to advance clinical applications (Bedini et al. 2005; Choi et al. 2009; Reddy and Jeganmohan 2017; Yao and Larock 2005). Despite the multi-potent activity of aristolactams, their potential anti-HIV-1 effects, including the contribution of their unique structural feature—the dibenzo-indole core—have not yet been elucidated.
For the purposes of this study, we identified an aristolactam derivative from a synthetic organic compound library that inhibited HIV-1 infection. The effect of substituting side chains on the aristolactam derivative on antiHIV-1 activity and cytotoxicity was investigated. In addition, the mode of anti-HIV-1 action of the aristolactam derivatives was identified as inhibition of Tat-mediated viral transcription rather than inhibition of viral reverse transcription or integration.
-
The inhibition of HIV-1 infection by aristolactam derivatives was tested in TZM-bl cells, which contain long terminal repeat (LTR)-driven firefly luciferase and lacZ (bgalactosidase), and express human CD4, CXCR4, and CCR5, in order to facilitate HIV-1 infection, as described previously (Platt et al. 1998). bl-DTR (TZM-bl-derived dual Tat reporter) cells were generated from TZM-bl cells by transforming two doxycycline-inducible lentiviral expression cassettes encoding flag-tagged tat and Renilaluciferase genes; these cells were then used to determine Tat-mediated HIV-1 transcriptional activity, as described previously (Shin et al. 2017). TZM-bl and bl-DTR cells were cultured in Dulbecco's modified Eagle's medium supplemented with 1% penicillin–streptomycin and 10% (v/v) heat-inactivated fetal bovine serum (all obtained from Gibco-BRL, Gaithersburg, MD, USA). The bl-DTR cells were additionally supplemented with 1 μg/mL puromycin and 200 μg/mL zeocin. Peripheral blood mononuclear cells (PBMCs) were purchased from AllCells (Alameda, CA, USA) and cultured, as described previously (Yoon et al. 2015). HIV-1 clones pNL4-3 and AD8, as well as TZM-bl and A3.01 cells, were obtained from the National Institute of Health's AIDS Research and Reference Reagent Program (NIH, Bethesda, MD, USA). Organic chemical compounds were provided by the Korea Research Institute of Chemical Technology (KRICT) (Choi et al. 2009). The chemicals were renamed as follows: (1-(2-(dimethylamino)ethyl)-9-methoxybenzo[6, 7]oxepino[4, 3, 2-cd]isoindol-2(1H)-one (ID 262860): 1, 2, 8, 9-tetramethoxy-5-(2- (pyrrolidin-1-yl)ethyl)dibenzo[cd, f]indole-4(5H)-one < Compound 1 > : 1, 2, 8, 9- tetramethoxy-5-(2-(piperidin-1-yl)ethyl)- dibenzo[cd, f]indole-4(5H)-one < Compound 2 > : 5-(2-(diethylamino)ethyl)-tetramethoxydibenzo[cd, f]indole-4(5H)-one < Compound 3 > : 5-(2-(diethylamino)ethyl)-1, 2-dimethoxydibenzo[cd, f]indole-4(5H)-one < Compound 4 > : 1, 2, 9-trimethoxy-5-(2-(piperidin-1-yl)ethyl)dibenzo[cd, f]indole-4(5H) -one < Compound 5 > : 1, 2-dimethoxy-5-(2-piperidin-1- yl)ethyl)dibenzo[cd, f]indole-4(5H)-one < Compound 6 > : 2-amino-5-(2-piperidin-1-yl)ethyl)dibenzo[cd, f]indole-4(5H)- one < Compound 7 > : 8-fluoro-1, 2-dimethoxy-5-(2-piperidin-1-yl)ethyl) dibenzo[cd, f]indole-4(5H)-one < Compound 8 > : 8-cloro-1, 2-dimethoxy-5-(2-piperidin-1-yl)ethyl) dibenzo- [cd, f]indole-4(5H)-one < Compound 9 > .
Seliciclib, azidothymidine (AZT), adefovir (ADV), raltegravir (RAL), and elvitegravir (ELV) were purchased from Sigma Aldrich (St. Louis, MO, USA).
-
To determine the inhibitory effect of the compounds on HIV-1 infection, TZM-bl cells were used, as described previously (Shin et al. 2020), with some minor modifications. In brief, 2 × 104 cells were cultured in 96-well plates for 24 h and then treated with compounds at a final concentration of 3 μmol/L (single dose assay; Fig. 1 and Table 1) or with 1:2 serially diluted compounds at concentrations ranging from 0 to 25 μmol/L (dose-dependent assay; Table 2). At 1 h after treatment, the cells were infected with the HIV-1NL4-3 virus at a multiplicity of infection (MOI) of 1. After 48 h, the inhibitory effect of the compounds was determined using a Bright Glo luciferase assay kit (Promega). The infectivity data are presented as a percentage relative to the DMSO control (vehicle).
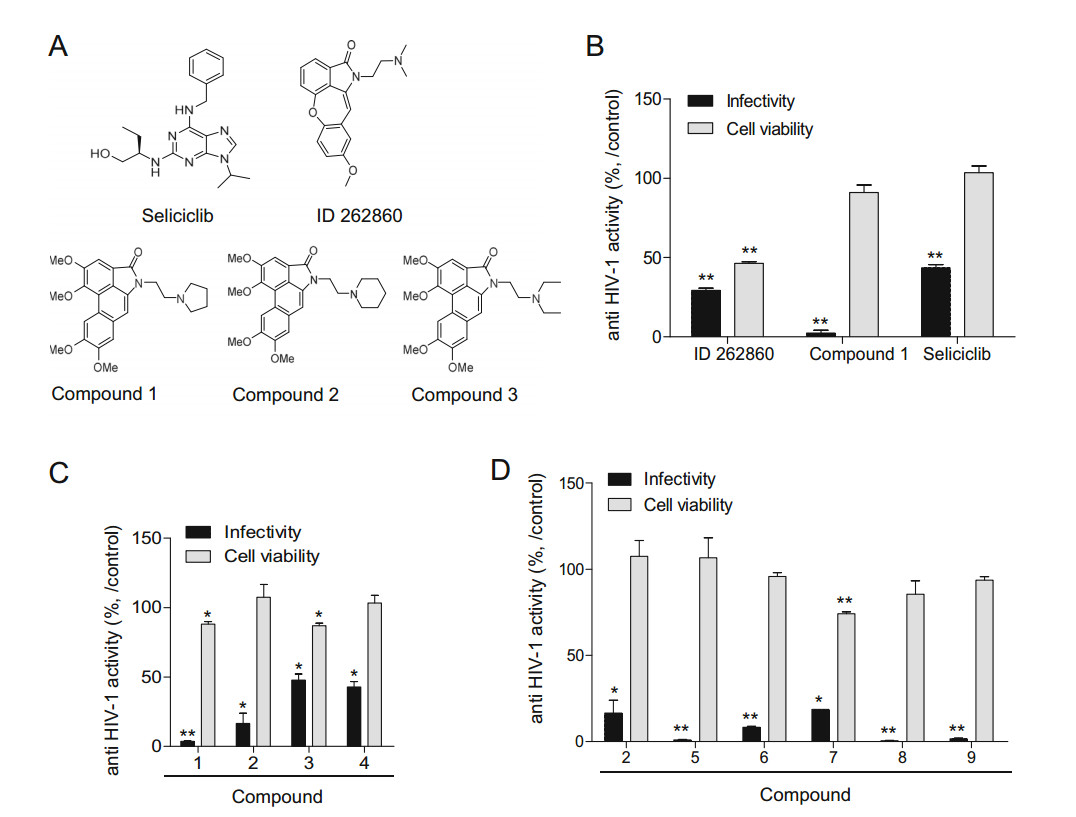
Figure 1. Comparison of the anti-HIV-1 effect of aristolactam derivatives. TZM-bl cells (2 × 104 cells) were treated with 3 μmol/L of each Compound 1 h prior to infection with the HIV-1NL4-3 strain at an MOI of 1. After 48 h, viral infectivity was determined with a firefly luciferase assay kit. Cell viability was assessed using an MTT-based cell viability reagent. The tested compounds are as follows: A Chemical structure of aristolactam derivatives and seliciclib; B dibenzo[cd, f]indol-4(5H)-one (Compound 1), similar compounds containing benzo[6, 7]oxepino[4, 3, 2-cd]isoindol-2(1H)-one (ID 262860), and purine (seliciclib); C 50N substitution of 5-(2-(pyrrolidinyl)ethyl) (Compound 1), 5-(2-(piperidinyl)ethyl) (Compound 2), 5-(2-(diethylamino)ethyl) (Compound 3), and H-substitution at R6-R7 of 5-(2-(diethylamino)ethyl) (Compound 4) on dibenzo[cd, f]indol- 4(5H)-one; D methoxy-deleted forms on 5-(2-(piperidinyl)ethyl) (Compound 2). The graphical data are presented as a value relative to the vehicle (DMSO)-treated controls, as the mean ± SD (n = 3). *P < 0.05 and **P < 0.01 compared with cells treated with the vehicle.
Compound R1 R3 R4 R6 R7 HIV-1 infectivity (%) Cell survival rate (%) 1 -CH2CH2N pyrrolidine OMe OMe OMe OMe 3.62±0.27 88.07±1.88 
2 -CH2CH2N piperidine OMe OMe OMe OMe 16.61±7.37 107.46±9.21 3 -CH2CH2N-(CH2CH3)2 OMe OMe OMe OMe 47.78±4.35 86.89±1.97 4 -CH2CH2N-(CH2CH3)2 OMe OMe H H 42.71±3.95 103.37±5.57 5 -CH2CH2N piperidine OMe OMe OMe H 0.98±0.27 106.63±11.69 6 -CH2CH2N piperidine OMe OMe H H 8.32±0.51 95.84±2.26 7 -CH2CH2N piperidine NH2 H H H 18.69±0.00 74.18±1.17 8 -CH2CH2N piperidine OMe OMe H F 0.48±024 85.50±7.76 9 -CH2CH2N piperidine OMe OMe H Cl 1.65±0.25 93.67±2.12 Seliciclib 45.01±1.99 103.57±4.30 ID 262860 29.35±1.33 46.34±0.93 The inhibitory effects on HIV-1 infection and cell viability were determined in TZM-bl cells infected with HIV-1NL4-3 at an MOI of 1. Table 1. Structure and inhibitory effect of aristolactam derivatives 1–9 on HIV-1 infection.
Compound IC50 (μmol/L)a CC50 (μmol/L)b SIc 1 0.69 ± 0.09 6.88 ± 0.31 9.94 2 1.03 ± 0.38 16.91 ± 3.22 16.45 3 3.73 ± 1.00 17.15 ± 0.34 4.59 4 3.07 ± 0.22 6.98 ± 0.09 2.27 5 1.07 ± 0.06 4.51 ± 0.96 4.23 6 1.06 ± 0.03 4.97 ± 0.62 4.70 7 2.00 ± 0.61 3.62 ± 0.15 1.80 8 0.55 ± 0.01 3.72 ± 0.01 6.74 9 0.44 ± 0.01 3.64 ± 0.08 8.31 Seliciclib 2.29 ± 0.40 25.49 ± 0.17 11.11 The inhibitory effects on HIV-1 infection and cell viability were determined in TZM-bl cells infected with HIV-1NL4-3 at an MOI of 1.
aIC50: half-maximal inhibitory concentration.
b CC50: concentration that reduces cell viability by 50%.
cSI: selectivity index, i.e. the ratio of IC50 to CC50.Table 2. Concentration–responses of aristolactam derivatives on cytotoxicity and anti-HIV activity.
The inhibitory effects of the compounds on viral replication were determined, as described previously (Shin et al. 2020). In brief, 5 × 104 cells/well of A3.01 cells, PBMCs and MOLT4-R5 cultured without activation were infected with HIV-1NL4-3 or HIV-1AD8 at an MOI of 0.1 in 96-well plates for 4 h. After infection, the compounds were added to the infected cells at a final concentration of 3 μmol/L. After 72 h of treatment, the inhibitory effect of the compounds on viral replication was determined by measuring the amount of p24, a HIV capsid protein, using an HIV-1 p24 ELISA kit. Cell viability was determined using the 3-2, 5-diphenyltetrazolium bromide (MTT)-based PrestoBlue Cell Viability Reagent (Invitrogen) according to the manufacturer's instructions.
-
To determine the inhibitory effect of the compounds on Tat-mediated transcription, a concentration–response assay was performed, as described previously (Shin et al. 2020). In brief, 1 × 104 bl-DTR cells cultured in a 100 μL medium were treated with serial dilutions of the compounds (0–25 μmol/L), after which the expression of Tat and renilla luciferase was induced by adding 50 μL doxycycline to achieve a final concentration of 50 ng/mL. After 24 h of treatment, the activities of Tat-induced firefly luciferase and doxycycline-induced renilla luciferase were determined using a dual luciferase assay kit (Dual Glo, Promega), as described previously. The data are presented as a percentage relative to the DMSO control (vehicle) in the presence of doxycycline. The experiment was performed in triplicate.
-
Reverse transcriptase activity was determined, as described previously, with certain modifications (Clouser et al. 2010). In brief, after treatment with DNase I, 2 × 105 TZM-bl cells were treated with the indicated compounds for 1 h prior to infection with HIV-1NL4-3 (at a MOI ratio of 1). Sixteen hours after infection, cytosolic DNA from the cells was isolated and the levels of reverse transcription (RT) products were determined by quantitative PCR (qPCR). The primers for RT products were 5′- GGTCCAAAATGCGAACCCAG-3′ (forward) and 5′- TCTTGCTTTATGGCCGGGTC-3′ (reverse). To determine the relative levels of the RT products, rRNA from the lysed cells were analyzed with one-step quantitative RTPCR using the following primer sets for 18S rRNA: 5′- GTAACCCGTTGAACCCCATT-3′ (forward) and 5′- CCATCCAATCGGTAGTAGGG-3′ (reverse). The relative level of each RT product was analyzed using the delta/ delta CT method, as described previously (Shin et al. 2020).
-
To determine how the compounds inhibited HIV-1 infection, the integrase activity was assessed using an XpressBio HIV-1 integrase assay kit, according to the manufacturer's protocols.
-
All data are expressed as the mean ± SD (n = 3). The data were compared using a Student's t test, and *P < 0.05, ** P < 0.01 was considered to be statistically significant. All statistical analyses were performed using Prism software (v.5.0; Graph Pad Software, San Diego, CA, USA).
Cells, Virus, and Reagents
Inhibition of HIV-1 Infection
Inhibition of Tat-mediated Transcription
Reverse Transcriptase Assay
Integrase Assay
Statistical Analysis
-
Initially, a random screen of an organic compound library identified an aristolactam derivative (Compound 1) that inhibited HIV-1 infection. To explore whether the dibenzoindole moiety in the core of the aristolactam derivative is critical for the inhibition of HIV-1 infection, the antiviral effects of benzo[6, 7]oxepino[4, 3, 2-cd]isoindol-2(1H)-one (ID 262860), a compound that is structurally similar to dibenzo[cd, f]indol-4(5H)-one (Compound 1), and seliciclib, which bears a purine moiety that is structurally similar to an indole moiety (Fig. 1A), were compared at a final concentration of 3 μmol/L. As shown in Fig. 1B, the compound bearing a benzo-oxepino-isoindol moiety (ID 262860) had a moderate inhibitory effect on HIV-1 infection and was cytotoxic. Seliciclib, a known inhibitor of HIV-1 infection, had a moderate inhibitory effect on HIV-1 infection and was not cytotoxic. Dibenzo[cd, f]indol-4(5H)- one (Compound 1) exhibited the greatest inhibitory effect on HIV-1 infection and was slightly cytotoxic at the concentration tested (Fig. 1B and Table 1). To investigate whether variation of the R1 side chain on dibenzo[cd, f]indol-4(5H)-one influences the anti-HIV-1 effect, 5-(2- (pyrrolidiny)lethyl) of Compound 1 was substituted to 5-(2-(piperidinyl)ethyl) (Compound 2) and 5-(2-(diethylamino)ethyl) (Compound 3). Although the 5-(2-(piperidinyl)ethyl) (Compound 2) substitution inhibited HIV-1 infection slightly less than the Compound 1 containing pyrrolidine, Compound 2 exhibited no cytotoxicity at the same concentration. However, 5-(2-(diethylamino)ethyl) (Compound 3) had a low inhibitory effect on HIV-1 infection and cell viability (Fig. 1C and Table 1). Since 5-(2-(piperidinyl)ethyl) (Compound 2) exhibited no cytotoxicity and had an efficient antiviral effect, we explored an extended set of derivatives substituted at the R3-R4-R6-R7 position on Compound 2 in order to find agents that could have a potent inhibitory effect on HIV-1 infection while lowering toxicity. As shown in Fig. 1D and Table 1, the substitution of R7-H (Compound 5) greatly improved the inhibitory effect of the compound on HIV-1 infection without increasing cytotoxicity. However, the additional deletion of the methoxy moiety (Compound 6 and 7) slightly reduced the inhibitory effect of the compound and increased cytotoxicity, whereas substitution with R7-F (Compound 8) or R7-Cl (Compound 9) greatly improved the inhibitory effect on HIV-1 infection and decreased cell viability (Fig. 1D and Table 1). The deletion effect of these methoxy was not detected in a compound bearing the 5-(2- (diethylamino)ethyl) moiety (Compound 4) (Fig. 1C). These results show that the aristolactam derivatives containing a dibenzo-indole core inhibited HIV-1 infection, and provided proof-of-principal that R1-substitution of (piperidinyl)ethyl and (pyrrolidinyl)ethyl moieties connected to the lactam ring increased anti-HIV-1 activity, and that substitutions at R3-R4-R6-R7 on Compound 2 influenced antiviral activity and cell viability. The inhibitory effect of the aristolactam derivatives on HIV-1 infection and cell viability (tested at a concentration of 3 μmol/L) are summarized in Table 1.
-
To accurately evaluate the potency of the aristolactam derivatives, TZM-bl cells infected with HIV-1NL4-3 were treated with 1:2 serially diluted compounds at a starting concentration of 25 μmol/L. The cytotoxicity of the compounds was also evaluated. As a control, seliciclib had a half-maximal inhibitory concentration (IC50) value of 2.29 μmol/L and a selectivity index (SI, the ratio of IC50 to CC50: a concentration which reduces cell viability by 50%) of 11.11; this value was similar to that reported in a previous study (Shin et al. 2020). As shown in Table 2, 5-(2- pyrrolidinyl)ethyl) (Compound 1) exhibited a strong inhibitory effect, with an IC50 value of 0.69 μmol/L. The IC50 values of 5-((2-piperidinyl)ethyl) (Compound 2) and 5-(2- diethylamino)ethyl) substituents (Compound 3) were 1.03 μmol/L and 3.73 μmol/L, respectively. The CC50 values of Compound 1, Compound 2, and Compound 3 were 6.88 μmol/L, 16.91 μmol/L, and 17.15 μmol/L, respectively, and the selectivity indices were 9.94, 16.45, and 4.59, respectively. From these results, the 5-(2-piperidinyl)ethyl) substitution at R1 (Compound 2) showed the highest SI with low cytotoxicity. The effect of substituting side chains at R3-R4-R6-R7 on 5-(2-(piperidinyl)ethyl)- dibenzo[cd, f]indol-4(5H)-one (Compound 2) was evaluated; two derivatives substituted to R7-H (Compound 5) and R6-R7-H (Compound 6) had IC50 values of 1.07 μmol/L and 1.06 μmol/L, respectively, which were similar to the IC50 value of tetra-methoxy (Compound 2); however, they exhibited severe cytotoxicity, as indicated by CC50 values of 4.51 μmol/L and 4.97 μmol/L, respectively, and low SIs (4.23 and 4.70, respectively). The compound with H-substitution at positions R4-R6-R7 (Compound 7) showed the highest cytotoxicity, as indicated by the lowest SI (1.80). Substitution of R7-F (Compound 8) and R7-Cl (Compound 9) increased the antiviral effect (IC50 values of 0.55 μmol/L and 0.44 μmol/L, respectively), but decreased cell viability (CC50 values of 3.72 μmol/L and 3.64 μmol/L, respectively), and resulted in low SIs of 6.74 and 8.31, respectively. An H-substituent (Compound 4) at R6-R7 on 5-(2-(diethylamino)ethyl) (Compound 3) increased cytotoxicity (CC50 value of 6.98 μmol/L), but its antiviral effect was not higher than that of Compound 3. Consequently, among the 50N-substituents (R1), 5-(2-(piperidinyl)ethyl) (Compound 2) exhibited good inhibition of HIV-1 infection and the lowest cytotoxicity. H-substitution at positions R6-R7 on Compound 2 increased cytotoxicity, and substitution to F and Cl at position R7 increased the anti-HIV-1 effect, but this was accompanied by high cytotoxicity (Table 2). These data indicate that aristolactam derivatives potently inhibited HIV-1 infection, while cytotoxicity increased greatly at concentrations of over 6.25 μmol/L (data not shown).
-
To determine the inhibitory effect of the aristolactam derivatives on the overall HIV-1 life cycle, HIV-1 replication assays were performed in A3.01 T-cell lines (Fig. 2A) and PBMCs (Fig. 2B) infected with CXCR4 tropic HIV-1NL4-3 in the presence or absence of these compounds. 5-(2-pyrrolidinyl)ethyl (Compound 1) and derivatives of 5-(2-piperidinyl)ethyl (Compounds 2 and 5–9) almost completely abrogated viral replication at a concentration of 3 μmol/L, with a cell survival rate of over 50%. 5-(2-(diethylamino)ethyl) (Compound 3) and its R6- R7-H substituted form (Compound 4) showed less inhibition of viral replication, which accorded with its reduced effect on viral infection. As a representative compound, Compound 1 suppressed replication of the CCR5 tropic HIV-1AD8 strain in the MOLT4-R5 cell line and in PBMCs; these effects were similar to the inhibitory effect of this compound on replication of the CXCR4 tropic HIV-1NL4-3 strain at the same concentration (Fig. 2C).
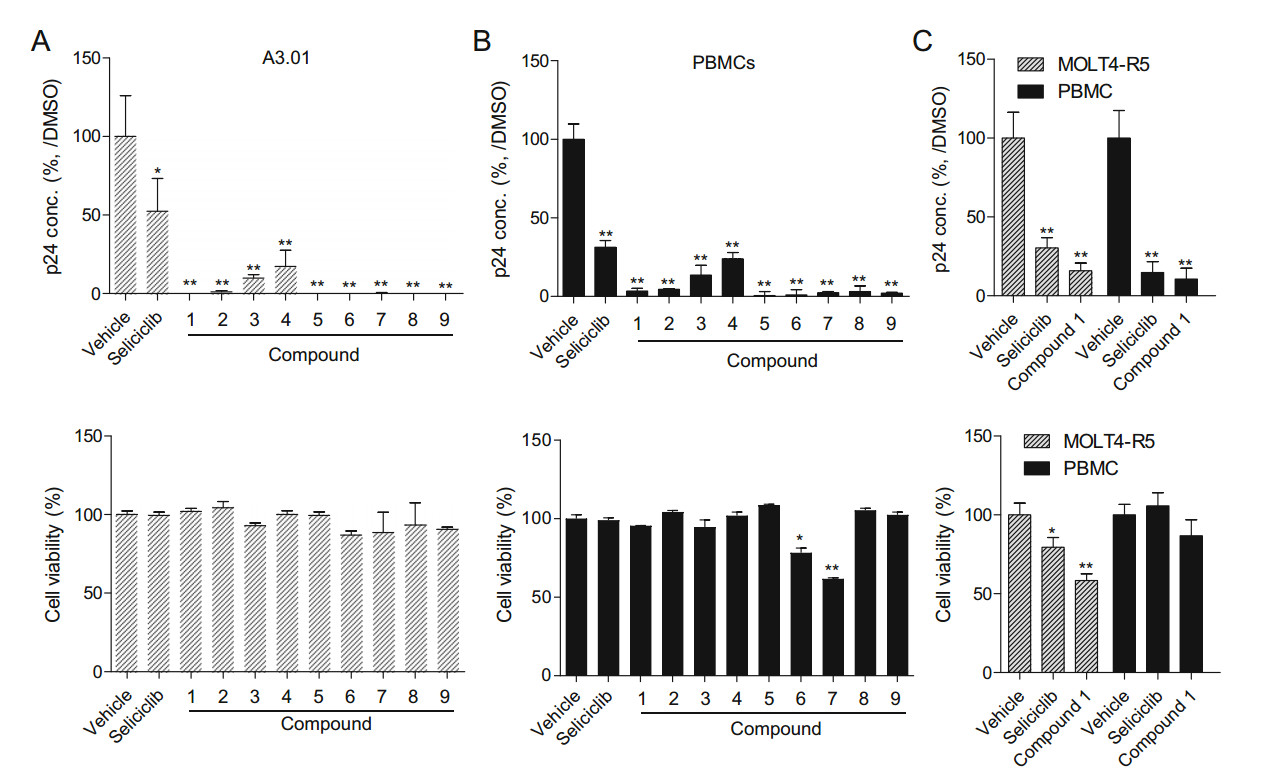
Figure 2. Inhibitory effect on viral replication. A3.01 cells (A) and PBMCs (B) (both 5 × 104) cultured without activation were infected with HIV-1NL4-3 at an MOI of 0.1 for 4 h and then treated with compounds (3 μmol/L) for 72 h. The inhibitory effect of the compounds on viral replication was determined by measuring the amount of p24 using an HIV-1 p24 ELISA kit. Cell viability was determined as described in Materials and Methods. C Cells (5 × 104 of MOLT4-R5 and PBMCs) infected with HIV-1AD8 at an MOI of 0.1 for 4 h were treated with (Compound 1) (3 μmol/L) for 72 h, and the p24 levels and cell viability were determined as shown above. Data (A–C) are presented as a value relative to the vehicle (DMSO)-treated controls, as the mean ± SD (n = 3). *P < 0.05 and **P < 0.01 compared with the cells treated with the vehicle.
-
To further define the mode of action associated with the inhibitory effects of the compounds on HIV-1 infection, a HIV-1 reverse transcriptase (RTase) assay and an integrase assay were performed. Initial studies used Compound 1 as a representative compound, since it was efficacious in inhibiting HIV-1 infection. RTase inhibitors azidothymidine (AZT) and integrase inhibitors (elvitegravir; ELG and raltegravir; RAL) inhibited the activity of RTase and integrase, respectively, but Compound 1 did not inhibit the activity of either enzyme (Fig. 3A, 3B).
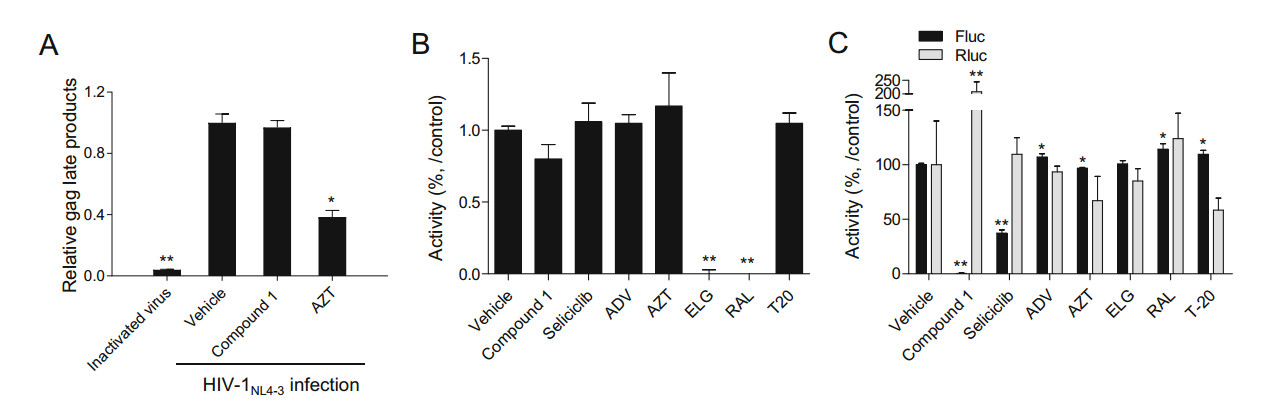
Figure 3. Determination of the anti-HIV-1 mode of action of the aristolactam derivatives. A Each compound was administered to TZM-bl cells for 1 h prior to infection with HIV-1NL4-3 at a final concentration of 10 μmol/L. After 16 h of infection, the cells were harvested and the levels of the RT product were determined as described in Materials and Methods. B An INTase assay was performed with 10 μmol/L of the indicated compounds according to the manufacturer's protocol. C bl-DTR cells (1 × 104) were treated with 3 μmol/L of the indicated compounds and then cultured in the presence of doxycycline (50 ng/mL). After 24 h of treatment, firefly luciferase and renilla luciferase activity was determined using the Dual-Glo-Luciferase assay system. The data (A–C) are presented as a value relative level to the vehicle (DMSO)-treated controls, as the mean ± SD (n = 3). *P < 0.05 and **P < 0.01 compared with the cells treated with vehicle. AZT; azidothymidine, ADV; adefovir, ELG; elvitegravir, RAL: raltegravir, T20: a fusion inhibitor.
To examine whether the aristolactam derivatives can inhibit HIV-1 transcription induced by the viral transcriptional factor Tat, a dual reporter system was employed. As this system compares the Tat-induced activity of firefly luciferase (F-Luc) or β-galactosidase (β-gal) with renilla luciferase (R-Luc) activity indicating normal cellular transcriptional activity, it can be used to assess the inhibitory effect of compounds on Tat-induced HIV-1 transcription. Compound 1 inhibited Tat-induced HIV-1 transcription without inhibiting R-Luc activity (Fig. 3C). Thus, a concentration–response assay of each of the aristolactam derivatives was conducted to determine their potency at inhibiting Tat-mediated viral transcription. All derivatives impaired HIV-1 transcriptional activity as measured by decreases in both F-Luc and β-gal activity at below 6.25 μmol/L, with no decrease in R-Luc activity (Fig. 4). Notably, 5-(2-pyrrolidinyl)ethyl (Compound 1) at a concentration of approximately 3 μmol/L completely inhibited HIV-1 viral transcription, with an IC50 value of 1.1 μmol/L (Table 3). R1 substitution of Compound 1 to 5-(2-piperidinyl)ethyl (Compound 2) and 5-(2-diethylamino)ethyl (Compound 3) resulted in IC50 values of 2.85 μmol/L and 2.58 μmol/L, respectively. Among the derivatives of Compound 2, an R7-H derivative (Compound 5) exhibited the highest inhibitory effect, with an IC50 value of 1.36 μmol/L, and the IC50 values of the other derivatives (Compound 6–9) were between * 1.78–3.38 μmol/L (Fig. 4 and Table 3). The inhibitory effect of each of the derivatives on HIV-1 transcription was generally similar to their inhibitory effect on HIV-1 inhibition (Fig. 4 and Table 2). These data revealed that aristolactam derivatives bearing a dibenzo[cd, f]-indol backbone exerted an inhibitory effect on Tat-induced viral transcription, which accounts for their inhibitory effects on HIV-1 infection. The increase in R-Luc activity in combination with a decrease in F-Luc activity might have been due to an excess of common machinery for transcription caused by the suppression of F-Luc expression (Shin et al. 2020).

Figure 4. Concentration–response of the aristolactam derivatives. blDTR cells (1 × 104) were cultured in 100 μL medium for 24 h and then two-fold serial dilutions of each compound were added prior to addition of doxycycline (final concentration, 50 ng/mL). After 24 h of treatment, the activity of firefly luciferase (F-Luc, closed circle) and renilla luciferase (R-Luc, open circle) was determined using the DualGlo-Luciferase assay system. Beta-galactosidase activity (triangle) was measured using the β-galactosidase enzyme assay system in parallel with the luciferase assay. Seliciclib was used as the experimental control. The data are presented as the mean ± SD (n = 3).
Compound IC50 (μmol/L)a 1 1.11 ± 0.02 2 2.85 ± 0.17 3 2.58 ± 0.05 4 3.40 ± 0.02 5 1.36 ± 0.01 6 2.47 ± 0.26 7 1.78 ± 0.15 8 2.25 ± 0.24 9 3.38 ± 0.05 Seliciclib 2.25 ± 0.14 aThe compounds were assessed in bl-DTR cells using a concentration–response test. Table 3. Concentration–responses of aristolactam derivatives on the inhibition of Tat-induced HIV-1 transcription.
Aristolactam Derivative Inhibits HIV-1 Infection
Concentration–Response of Aristolactam Derivatives for Inhibition of HIV-1 Infection
Inhibitory Effects of Aristolactam Derivatives on HIV-1 Replication
Mode of Action of Aristolactam Derivatives on HIV-1 Inhibition
-
Currently, four classes of HIV-1 drugs have been developed, and these target the specific viral replication steps of entry, reverse transcription, integration, and proteasemediated viral maturation (Arts and Hazuda, 2012). However, these antiretroviral treatments often lead to the emergence of drug-resistant mutations and adverse effects, as well as reservoirs of cells infected with latent HIV-1 that are not eliminated by current treatments (Finzi et al. 1997). Therefore, efforts are needed to develop a new class of HIV-1 drug to control the spread of HIV/AIDS.
Many studies have been directed at screening compounds derived from natural products to find novel and potent HIV-1 agents. Several natural products (such as calanolides (Kashman et al. 1992), betulinic acid (Kashiwada et al. 1996), kuwanon-L (Esposito et al. 2015), and patentiflorin A (Zhang et al. 2017) potently inhibit viral replication by targeting steps that are essential to the viral life cycle, but these compounds are not yet used as clinical medications. Accordingly, further screens are ongoing to discover possible candidates for ART.
The naturally occurring aristolactam family is made up of relatively simple compounds, which have druggable potency and several distinctive biological activities (Chia et al. 2000; Kim et al. 2004; Tsai et al. 2005; Zhang et al. 2007). Therefore, several research groups have synthesized diverse aristolactam derivatives and conducted SAR studies to enhance the potency of these derivatives; such compounds might have applications against several human diseases in which aristolactams are known to be therapeutically beneficial, or new biological and/or clinical applications (Choi et al. 2009; Couture et al. 2002; Hoarau et al. 2001; Reddy and Jeganmohan 2017; Yao and Larock 2005).
In this study, an aristolactam derivative (Compound 1) was found to be effective in inhibiting HIV-1 infection. A compound (ID 262860) containing an isoindole core, which is structurally similar to Compound 1, also inhibited HIV-1 infection, although it was less potent and cytotoxic. The indole feature is structurally similar to a purine ring, which is known to play an important role in anti-HIV-1 effects, as exemplified by the cyclin-dependent kinase (CDK) inhibitor (seliciclib) (Agbottah et al. 2005; Schang 2002), and some purine derivatives containing aryl groups (Pang et al. 2008). Accordingly, it is thought that the indole core of aristolactam compounds, which is similar to that of purine-based compounds, might also provide a template for the development of new compounds against HIV-1 infection, as reviewed in a previous report (Xu and Lv 2009) (Fig. 1A, 1B). In this study, an R1 substitution to 5-(2-pyrrolidinyl)ethyl (Compound 1) and 5-(2-pyrrolidinyl)ethyl (Compound 2) on the indole ring produced more potent anti-HIV-1 effects than diethyl substituents (Compounds 3 and 4) (Fig. 1 and Table 1). These data indicate that 5′N-ring-type substitution on the indole core might increase the inhibition of HIV-1 infection, similar to the case of seliciclib, which consists of a purine core connected to a benzene ring. An SAR study of the antitumor effects of aristolactam derivatives reported that compounds with methoxy substitutions on the lactam ring tended to increase cell death (Choi et al. 2009), whereas, in this study, methoxy substitution decreased cell death but did not have a significant anti-HIV-1 effect (Tables 1 and 2). These data might indicate that the dibenzo ring in aristolactam might have a greater effect on cellular toxicity than on HIV-1 infection. The difference in cytotoxicity resulting from methoxy substitution observed between our study and a previous report could be attributable to the different cell types used. R7-Cl (Compound 9) substitution showed the lowest inhibitory effect on Tat activity, but the greatest anti-HIV-1 effect. The difference might be due to the fact that the compound could affect additional certain step(s) including viral entry, reverse transcription and/or integration, but excluding viral transcription during infection. On the other hand, seliciclib and its derivatives were initially developed an anti-cancer agents based on their inhibitory effect on the kinase activity of several CDKs required for both cancer cell growth and HIV-1 transcriptional elongation (Havlicek et al. 1997; Schang 2002) (Ivanov et al. 2018; Wei et al. 1998). The purine core connected with the ring type moiety of seliciclib and its derivatives (Olomoucine and Purvalanols, etc.) is critical to compete with ATP and efficiently occupy the ATP-binding pocket of CDKs (De Azevedo et al. 1997; Schang 2002). In the present study, aristolactam derivatives efficiently and specifically inhibited Tat-induced viral transcription, and several derivatives were more potent than seliciclib (Table 2). Even though the mechanism of aristolactam inhibition of Tat-mediated transcription has not yet been elucidated, it is possible - based on the structural similarity between seliciclib and aristolactam derivatives connected with 5′-ring type moiety (Fig. 1A)-that aristolactam binds to the ATP-binding pocket of CDKs. Indeed, aristolactam derivatives connected with 5′-ring type moiety (Compound 1 and 2) have been observed to more efficiently inhibit HIV-1 infection than 5′N-diethyl substituents (Compounds 3 and 4) (Tables 1 and 2). Instead, it seems that the inhibition of CDKs by aristolactam derivatives is not the sole mode of action for their anti-HIV-1 activity because that activity was not absolutely concordant with their Tat inhibitory activity. As such, further study is required to determine whether aristolactam derivatives target the CDKs' activity directly.
Numerous investigations focused on the anti-cancer activity of aristolactams have shown that these compounds kill cancerous cells lines that are derived from the cancer cells used in our study. Although we could not reduce the cytotoxicity of the compounds to an acceptable level, it is conceivable that aristolactam derivatives could be developed to treat multifaceted diseases, such as HIV-infections in patients with cancer. Moreover, the scaffold of aristolactam could be modified to provide a novel class of HIV-1 drugs targeting viral transcription if the cytotoxicity of such drugs can be reduced. This was achieved in the case of azidothymidine, which was originally developed as an anti-cancer agent (Mitsuya and Broder 1987).
In summary, we randomly screened a synthetic organic compound library to identify a series of aristolactam derivatives that inhibit HIV-1 infection. The inhibitory effect of these compounds on HIV-1 activity was associated with the specific inhibition of Tat-mediated viral transcription. Together, our results demonstrate that aristolactams inhibit HIV-1 infection, and may provide a strategy for the development of a new class of HIV-1 drug.
-
This work was supported by grants from the Korea National Institute of Health (Grant Number: 2019-NI-066-00 and 2020-ER5106-00).
-
CHY conceived the project. YS and CMP mainly conducted the experiments and participated in the drafting of the manuscript. HGK, MSC, DEK, BSC, KK discussed and analyzed the data. CHY wrote the manuscript. All of the authors have read and approved the final manuscript.
Author Contributions
-
The authors declare that they have no conflict of interest.
-
This article does not contain any studies with human or animal subjects performed by any of the authors.















 DownLoad:
DownLoad: