HIV/AID Research in China: Chasing against Time
2007, 22(6): 417
Received: 07 November 2007
Accepted: 10 November 2007
Strategy for AIDS Prevention and Treatment
2007, 22(6): 419
Received: 29 June 2007
Accepted: 10 October 2007
HIV/AIDS has been circulating in China for over 25 year. While making progress and achievements on HIV/AIDS prevention, there still are great challenge and difficulties such as HIV epidemic controlling and vaccine research.
AIDS Research and Its Role in China's AIDS Prevention and Control Policies
2007, 22(6): 421
Received: 09 November 2007
Accepted: 12 November 2007
By the end of 2005, the estimated number of HIV infected people in China was 650,000. The seriousness of the epidemic calls for effective control measures to tackle the problems in order to avoid the tragedy in Africa from happening in China. “Prevention First” is the cornerstone of the country’s health policy. On 2003 World AIDS Day, Premier Jiabao Wen announced a new national AIDS control policy, “Four Frees and One Care”. This policy clearly shows that the Chinese government has once again taken full responsibility to solve public health problems and has profound impact far beyond the AIDS field. In early 2006, the central government put scientific and technology innovation as a national priority and set the target to build an innovative China by year 2020. Since then, the government has been increasing investment in science and technology with major emphasis on both infectious diseases control and new drug research and development. For the first time, development of 100 new drugs and control of major infectious diseases (AIDS, HBV, TB and other emerging infectious diseases) have been selected as national key scientific projects. China’s best minds in related fields will be pooled to work together in order to remove the technical barriers blocking efficient control of the major infectious disease in China. Knowledge on molecular epidemiology, immunology, pathogenesis, HAART, as well as HIVDR strains will certainly provide urgently needed scientific information for China’s AIDS control program. Only evidence-based strategy from good research will provide long-term effective control of AIDS.
HIV/AIDS in Asia: The Shape of Epidemics and Their Molecular Epidemiology
2007, 22(6): 424
Received: 09 July 2007
Accepted: 20 September 2007
The Asia-Pacific region is a home to 60% of the population in the world and to approximately one quarter of people with HIV/AIDS. Close to a million of people has been infected and a half million people died of AIDS annually in Asia, becoming the second largest epicenter of global AIDS epidemic. Molecular epidemiology has been useful tool to track a course of HIV spread. In-depth knowledge from the studies on molecular epidemiology elucidates the dynamics of HIV spread and the interrelationship of epidemics in the different regions in Asia.
Development and Challenge of HIV/AIDS Testing Laboratory Network and Quality Assurance System in China
2007, 22(6): 434
Received: 10 September 2007
Accepted: 10 October 2007
This paper describes the development and challenge of HIV/AIDS testing laboratory network and quality assurance system in China. At present, the HIV/AIDS testing laboratories includes three classes, the National AIDS Reference Laboratory, HIV/AIDS confirmatory laboratories and HIV/AIDS screening laboratories. All of them are accredited by the health authorities, and each class of laboratories take charge of their function strictly according to the “National Management of HIV/AIDS Detection (2006)”. A complete quality assurance and quality control system for HIV/AIDS testing has been developed, which includes technical training, strict laboratory monitoring and approval, examination or proficiency testing on HIV/AIDS detection, and quality evaluation and supervision of HIV/AIDS diagnostic kits. Besides conduct the routine anti-HIV antibody test, more and more laboratories began to conduct other tests, such as CD4+ T lymphocyte cell counting, HIV viral load, HIV DNA PCR, genotyping, drug resistance, and HIV-1 recent infection test. The primary challenges faced by the HIV/AIDS testing laboratory network are in the areas of laboratory management and quality control. For example, the provincial PT program is inefficient, the internal quality control is conducted perfunctorily, personnel training can not met the needs of the workplace, which need to be improved.
HIV and HCV: from Co-infection to Epidemiology, Transmission, Pathogenesis, and Treatment
2007, 22(6): 443
Received: 24 September 2007
Accepted: 08 November 2007
Human immunodeficiency virus (HIV) is the infectious agent causing acquired immu- nodeficiency syndrome (AIDS), a deadliest scourge of human society. Hepatitis C virus (HCV) is a major causative agent of chronic liver disease and infects an estimated 170 million people worldwide, resulting in a serious public health burden. Due to shared routes of transmission, co-infection with HIV and HCV has become common among individuals who had high risks of blood exposures. Among hemophiliacs the co-infection rate accounts for 85%; while among injection drug users (IDU) the rate can be as high as 90%. HIV can accelerate the progression of HCV-related liver disease, particularly when immunodeficiency has developed. Although the effect of HCV on HIV infection is controversial, most studies showed an increase in mortality due to liver disease. HCV may act as a direct cofactor to fasten the progression of AIDS and decrease the tolerance of highly active antiretroviral therapy (HARRT). Conversely, HAART-related hepatotoxicity may enhance the progression of liver fibrosis. Due to above complications, co-infection with HCV and HIV-1 has imposed a critical challenge in the management of these patients. In this review, we focus on the epidemiology and transmission of HIV and HCV, the impact of the two viruses on each other, and their treatment.
Novel Antiviral Agents Targeting HIV Entry and Transmission
2007, 22(6): 451
Received: 13 September 2007
Accepted: 09 October 2007
Studies of the mechanism of HIV entry and transmission have identified multiple new targets for drug development. A range of inhibitors have demonstrated potent antiretroviral activity by interfering with CD4-gp120 interaction, coreceptor binding or viral-cell fusion in preclinical and clinical studies. One of these agents, fusion inhibitor enfuvirtide, is already in clinical use. Here we review the progress in the development of specific entry inhibitors as novel therapeutics. The potential of entry inhibitors as topical microbicides to block HIV transmission is also discussed.
"Unconventional" Neutralizing Activity of Antibodies Against HIV
2007, 22(6): 462
Received: 03 July 2007
Accepted: 25 September 2007
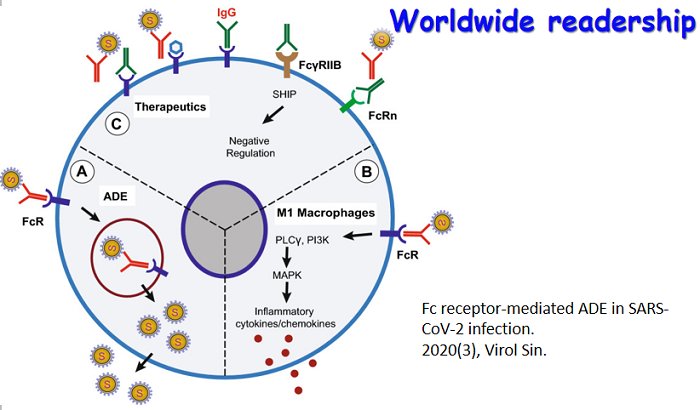
Neutralizing antibodies are recognized to be one of the essential elements of the adaptive immune response that must be induced by an effective vaccine against HIV. However, only a limited number of antibodies have been identified to neutralize a broad range of primary isolates of HIV-1 and attempts to induce such antibodies by immunization were unsuccessful. The difficulties to generate such antibodies are mainly due to intrinsic properties of HIV-1 envelope spikes, such as high sequence diversity, heavy glycosylation, and inducible and transient nature of certain epitopes. In vitro neutralizing antibodies are identified using “conventional” neutralization assay which uses phytohe- magglutinin (PHA)-stimulated human PBMCs as target cells. Thus, in essence the assay evaluates HIV-1 replication in CD4+ T cells. Recently, several laboratories including us demonstrated that some monoclonal antibodies and HIV-1-specific polyclonal IgG purified from patient sera, although they do not have neutralizing activity when tested by the “conventional” neutralization assay, do exhibit potent and broad neutralizing activity in “unconventional” ways. The neutralizing activity of these antibodies and IgG fractions is acquired through post-translational modifications, through opsonization of virus particles into macrophages and immature dendritic cells (iDCs), or through expression of antibodies on the surface of HIV-1-susceptible cells. This review will focus on recent findings of this area and point out their potential applications in the development of preventive strategies against HIV.
A General Introduction of HIV/AIDS Treatment with Traditional Chinese Medicine in China
2007, 22(6): 471
Received: 09 June 2007
Accepted: 16 October 2007
This paper gives a general introduction of HIV/AIDS treatment with Traditional Chinese Medicine (TCM) in China during the past 20 years. Although the role of TCM in treatment of HIV/AIDS is promising, there is still a long way to go.
Current Status of Targets and Assays for Anti-HIV Drug Screening
2007, 22(6): 476
Received: 18 September 2007
Accepted: 08 October 2007
HIV/AIDS is one of the most serious public health challenges globally. Despite the great efforts that are being devoted to prevent, treat and to better understand the disease, it is one of the main causes of morbidity and mortality worldwide. Currently, there are 30 drugs or combinations of drugs approved by FDA. Because of the side-effects, price and drug resistance, it is essential to discover new targets, to develop new technology and to find new anti-HIV drugs. This review summarizes the major targets and assays currently used in anti-HIV drug screening.
HIV Vaccine-Challenges and Opportunities
2007, 22(6): 486
Received: 18 July 2007
Accepted: 16 October 2007
The need for an efficacious HIV/AIDS vaccine remains the highest priority of the world HIV/AIDS agenda. The generation of an efficacious HIV/AIDS vaccine proves an enormous scientific challenge. This article reviews the neutralizing antibody problem, elusive immune protection, im- munogen design, pre-existing anti-vector immunity and design of phase 3 vaccine trials and the challenges and opportunities in development of HIV/AIDS vaccine are discussed.
Challenges and Opportunities: the Expanded Government-led HIV/AIDS Programs in China
2007, 22(6): 493
Received: 24 July 2007
Accepted: 10 October 2007
HIV/AIDS has posed an increasingly serious issue in China. In recent years, Chinese government has taken further intensified efforts to combat HIV/AIDS with high-level political commitment, supportive policy development, increased financial allocation, large-scale of government- led initiatives, expanded international cooperation and great involvement of non-governmental organizations. Meanwhile gaps and challenges coexist impacting the implementation and the results of national HIV/AIDS programs. Thus, further government efforts are needed to improve and tailor the actions to meet the requirement of HIV/AIDS control in China.
Frequency and Absolute Number of FoxP3+ Regulatory T Cells Correlate with Disease Progression of Chronic HIV-1 Infection
2007, 22(6): 501
Received: 14 August 2007
Accepted: 11 October 2007
CD4+CD25+ Regulatory T cells (Treg) have been found to down-regulate immune activation in HIV-1 infection. However, whether the depletion of Treg benefits to the disease status of HIV infection remains undefined. To address this issue, we enumerated the Treg absolute counts and frequency in 75 antiviral-na?ve HIV-1-infected individuals in this study. It was found that HIV-infected patients displayed a significant decline in Treg absolute counts but a significant increase in Treg frequency. In addition, with disease progression indicated by CD4 T-cell absolute counts, circulating Treg frequency gradually increased; while Treg absolute counts were gradually decreased, suggesting that the alteration of Treg number closely correlated with disease progression in HIV infection. Functional analysis further showed that Treg efficiently inhibit both CD4 and CD8 T cell proliferation in vitro. Thus, our findings indicates that Treg actively participate in pathogenesis of chronic HIV infection, influencing the disease progression.
Establishment of AIDS Animal Model with SIVmac239 Infected Chinese Rhesus Monkey
2007, 22(6): 509
Received: 03 July 2007
Accepted: 16 October 2007
In the present research, two Chinese rhesus monkeys were inoculated intravenously with 5000 TCID50 of SIVmac239. The changes in the numbers of CD4+ T lymphocyte in peripheral blood, plasma viral loads, proviral DNA and humoral antibodies against virus were periodically monitored during 121 days. At the early stage of infection, proviral DNA had been detected in PBMCs, and infectious SIVmac239 virus had been isolated from PBMCs. At the same period, the numbers of CD4+ T lymphocytes were significantly decreased, and maintained at low level during the 121-day period of infection. Plasma viral loads reached the peak at week 2 post-inoculation and kept at a steady state subsequently. Moreover, antibodies against viral proteins were detected from plasma. All the results showed that the two Chinese rhesus monkeys had been infected with SIVmac239 successfully. This animal model can be applied for further AIDS researches.