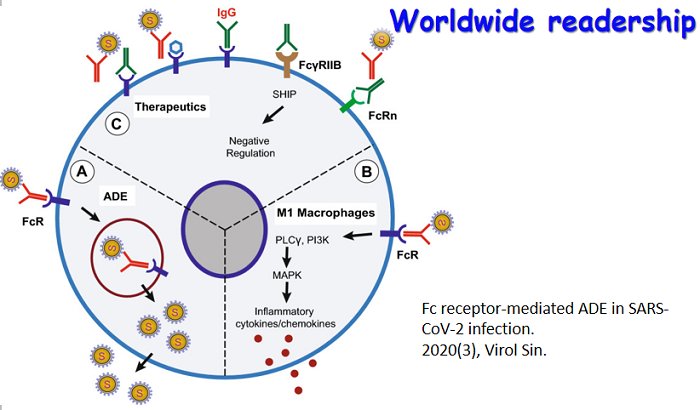
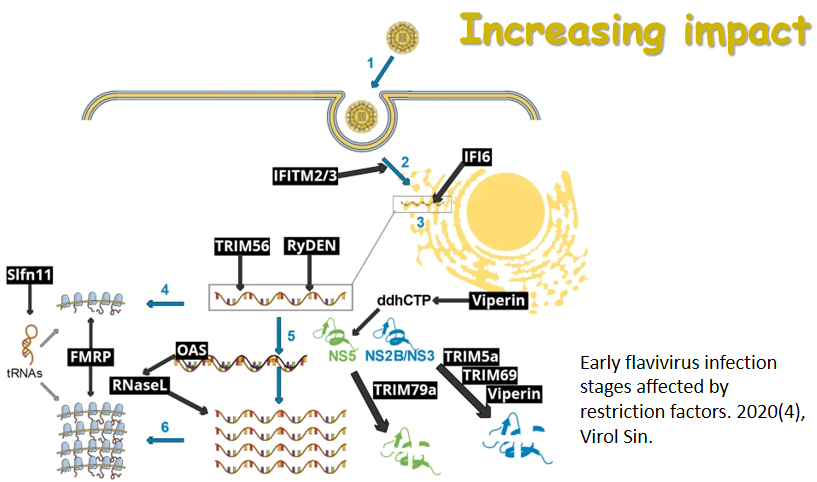
Hepatitf B virus(HBV1 genome DNA was directly extracted from human serum infected with HBV.The full length preS gene DNA was obtained by PCR using the HBV genome DNA as template.Then it w0s cloned into the pUCm—T vector and sequenced using the M 1 3 Primers.The sequencing data shows that it contains 1 203bp an d it length with 24 different nucleotides compared with the standard sequence of HBV (subtype adr)in China,it was confirmed to be the preS gene with three ATGs which corresponded to the initiation codon of PrsS1,PreS2 an d S proteins respectively.Th is DNA fragment was cloned into the SnaB 1-Avr II sites of expressing vector pPIC9K,in framed with the AOXI promotor and then the pPIC9K—PreS recombined plasmid DNA linearized by Sal 1 was introduc‘ ed to Pichia pastoris GSll5 by electroporation device.GSll5一pPIC9K—PreS was got by selecting with MD-G418 plates an d identifying with PCR.Th e GSll5-pPIC9K,PreS Can grow in the media with methan ol and can produce the PreS protein in secreted form with molecular weight 48KD as detected by SDS—PAGE.ELISA experiment proved that the protein Can react with the positive human serum against HBV
To study the association of heat-shock protein GRP94 an d HSP27 with Hantaan virus nucleocapsid protein(HTNV—NP)during viral replication,new—bom mice less than 72h of age were experimentally infected with Hantaan virus intracerebrally.Brains of mi ce at the 8th day post-infection were prepared for tissue sections an d extracts.Hantaan virus infection,GRP94,HSP27 expression an d their localization were demonstrated by immunohistochemi cal staining.The possible interaction between HTNV—NP an d GRIX)4.HSP27 was an alyzed by ELISA,immunoprecipitation.HTNV infection led to increased expression of GRIX)4 but not HSP27 at cytoplasma of neurons in brains of infected suckling mice.colocalizationof GRIX)4 with HTNV—NP,an d association of both GRP94 and HSP27 with HTNV—NP.Th e results provide the basis for further research of the roles of HSPs in the viral infection and replication.
To clone and express in vitro Human papillomavirus type 16 l1 (HPV16L1) gene ,and provide a good basis for further research DNA vaccine against HPV16 infection and human cervical cancer, the HPV16l1 gene fragment was amplified from genome of p16L1BN1 by PCR and cloned into eukaryotic expression vector pcDNA3.0.Sequencing showed the plasmid pcDNA3-HPV16L1 was constructed successfully. Then the recombinant plasmid pcDNA3-HPV16L1 transfected 7721 human liver cancer cells using Lipofectamine 2000 reagent. The molecular weight of the expressed protein, was 55kDa, analyzing by SDS-PAGE, as same as the molecular weight of HPV16L1. The Western blotting result show that the expressed protein could be detected by HPV16L1 specific monoclonal antibody. This study provided a good basis for further research on HPV16L1 DNA vaccine.
A PCR—M PH—ELISA method detecting HCMV infection was established.The PCR products of HCMV DNA were binting—labeled by a 5’-biotinlabeled primer through sample am plification.After the labeled product was mixed with a 5’-digoxin—labeled probe,hybridization was carried out for 2 mi nutes at 90℃ and for 1 minute at 55℃ .then the hybridized product was captured on microplate wells coated with streptoavidin and detected with a peroxidase—labeled an tibody and TMB substrate. The sensitivity of the method was 2.5×1 04 copies/mL . and higher than that of conventiona1 PCR and ELISA method.The results did not cross—react with that of HSV1,HSV2,RuV,EBV and ADV DNA. Th e method has potentia1 use for qualitative an d quantitative detection of HCMV infection.
A filamentous virus HN021 was isolated and purified from mosaic leaves of Solanum tuberosum collected from Shimen, Hunan province. It was primarily identified as potato virus X by double-stranded RNA analysis, host reaction tests and morphological observation of virus particles and inclusions. A fragment of about 1kb size was obtained by RT-PCR, which was performed with ds-RNA as template of cDNA synthesis and respective primers designed for 3’ end sequence of PVX. Homological comparison was carried out between the nucleotide sequences of ORF4-UTR-ORF5 regions of HN021 and those of 11 PVX isolates submitted to Genbank. Our result showed that HN021 was 78.4%-79.4% identical to three isolates from South America (COAT, KPA and HB), whereas up to 96.4%-97.8% identical to other eight isolates. From the comparison of sequences of amino acids, the similarities of CP and 8kDa protein were 86.5%-89.0% and 74.3%-75.5% respectively between HN021 and the three isolates from South America, and were 97.1%-98.7% and 97.1%-1
A rapid an d reproducible method was first established for assessment of Hog cholera lapinized virus(HCLV)loads in Classical Swine Fever vaccine using Flurogenic quantitative PCR (FQ·PCR)combines LiIghtCycler sequence detection system.The method contains a pair of primers and an internal daul·labled fluorogenic probe spanning the part of 5’noncoding region(5’NCR)of HCLV, the use of such a probe combined with the 5’-3’nuclease activity of Taq polymerase allows direct quan tification of the PCR product by the detection of a fluorescent reporter released in th e course of the exponential phase of the PCR.Th e sensitivity of the assay was 10 copies per reaction.Th e assay is linear within 6-log dynamic rang.The coeficient of variation(CV)of the standard of Ct value is 2.3% 一5.1% (n=10);The CV ofvaccine sample is 0.85%一2.8% in intra—assay and 2.5%一7.3% in inter·assay(n =5),respectively;Th e CV of the same sam ple in diferent RNA isolation and reverse transcription iS 5.0 % (n=5).Nine vaccines were quantified by this method and give similar but more accurate results compared to the conventional rabbit fever reaction.Th e entire assay,including RNA isolation,reverse tran scription,an d quan tification,could be completed within 4 hour s.In conclusion,the high sensitivity, simplicity,an d reproducibility of the HCLV RNA quan tification which allows the screening of large numbers of sam ples,combined with its wide dynamic ran g,makes this method especially suitable for evaluating th e viral loads an d guiding how to confect th e vaccine,it also provides a novel an d simple research tool for CSFV
Abstract:The capsid protein(VP60)gene of Rabbit hemorrhagic disease virus(RHDV)isolate WX84 was amplified by RT.PCR and cloned into pGEM R-T vector for sequence.Th e sequence an alysis showed that the nucleotide sequence of the vp60 gene of WX84 was 1 740 bp an d encoding a protein of 580 amino acids.Sequence comparison with other published RHDV vp60 genes showed that the homology of the nucleotide was between 98.2% and 99.0% .Th e result showed that vp60 gene was a highly conserved gene.
While observing Duck viral hepatitis seed virus in chicken embryonic allantochoroin liquid. another symmetry nonenveloped icosahedral viron with a diam eter of 7O tO 80 am was found.In order to investigation this contam inating virus,we selected four random primers to am plify this unknown virus by RT—PCR method an d PCR method respectively,that resulted three DNA fragments,showing 99.5% ,99.6% an d 99.5% homologue respectively with the genome of Adenovirus type I,or Chicken embryo orphan lethal virus(CELOV)during the sequencing analysis(one cycle reaction)for them. This results suggested that Duck viral hepatitis seed virus was contam inated by CELOV
Abstract:The f gene was amplified with one—step RT—PCR,cloned and sequenced from three virus isolates(Avian paramyxovirus type I),which were isolated from diferent avian (chicken and pigeon) inYunnan province.Th e result of the nucleotide sequence an alysis indicated that the homology between the three strains studied was 88.1%-94.9% :while the studied strains with refe;rence strains LaSota and F48F-,9 only showed 85.6% homology.Th e study on the cleavage site amino acid sequence deduced from the nucleotide sequence(residues 1 1 2-1 1 7)showed that the two NDV su~ns matched to all NDV velogenic strains and the Pigeon paramyxovirus(PPMV)I strain matched to PPMV ZQ98—1 strain(one mesogenic strain),SO the former were velogenic strains and the later was mesogenic strain.Phylogenetic tree constructed with 16 reference strains of 1 662nt(ORF region)of thef gene showed that the two NDV strains should belongs to genotype VII an d the PPM V strain belongs to genotype VI.
The nucleocapsid protein gene(orf 7)of Porcine reproductive and respiratory syndrome virus(PRRSV)was isolated from PRRSV genome by RT PCR.The gene was cloned into pMD 1 8一T vector,the recombinant plasmid pMD1 8N containing orf 7 was sequenced and compared with other PRRSV isolates.The result shown that the homologies between the cloned orf 7 and America type PRRSV ATCC VR一2332 reached 100%,the orf 7 was highly conservative sequence in PRRSV genome. The orf 7 was subcloned into prokaryofic expressing vector pGEX—KG,the recombinant plasmid named pGEX—KGN was constructed.The pGEX—KGN was used to tran sfo rnl into E colf BL21.Th e results of SDS—PAGE an d W estem—blot indicated that the nucleocapsid protein gene cloned in downstream of Glutathione S—tran sferase(GST)was expressed in a high level an d the recombinan t fusion protein, which was about 41kDa.had immunologicaily reactive activity.This study lay on foundation for th e development of the diagnosis meth ods in serology fo r PRRSV
Morphological characterization of Infectious canine hepatitis virus specific inclusion bodies was carried out by both immunogold electron microscopy and electron microscopy in sire hybridization. Three morphological types of inclusion bodies were observed:loose homogeneous,paracrystalline an d dense gran ular inclusion body.The former two labelled with immunogold are viral an tigenic protein of viral particles,the latter one labelled with viral DNA probe is composed of viral DNA.Th e possible relationship among the inclusion bodies an d their be aring to virus morphogenesis were discussed
Abstract:The Foot and mouth disease virus(FMDV、non.structural protein 3ABC can be used for diferentiation of infection from vaccination.The primers of 3ABC gene were designed an d synthesized, an d the 5’end an d 3’end of primers were adding the sequence of restriction endonuclease BamH I an d Hind III respectively.The 3ABC gene coding region was obtained from the FMDV genome RNA by RT.PCR.Th e amplified fragment was cloned into T.vector.The recombinan t plasmid pT.3ABC was digested with BamH I and ,ld III an d then cloned into pET32a.Th e recombinan t plasmid pET3ABC was transformed into BL21(DE3)plysS and the target protein was induced by IP1℃ .Expression of NSP.3ABC protein was examined and identificated bV SDS.PAGE,W_estem blotting an d EU SA.Th e results showed that recombinan t plasmid pET3ABC was constructed an d the NSP.3ABC was expressed in Ecoli successfullv.A special electrophoretic ban d in SDS.PAGE (56kDa)Was noted,Westem bloting showed it Can react witIl FM DV infected an imal serum ,and ELISA result showed the expressed protein Can be used to diferentiate the infection from vaccination.
A 0.77kb UL49 gene encoding the tegument protein VP22 was amplified from cells culture infected with Bovine herpesvirus 1 (BHV—I)bv PCR.The DNA fragment was further cloned into pM D 1 8一T vector,resulting in recombinant plasmid pM D—VP22.The sequences were determined an d there is no diference with the sequences of the 【,L 9 gene of BHV—I Cooper strain .Th e 0.77kb fragment was released from plasmid pMD..VP22 an d subcloned into the vectors pET..28a an d pEGFP-C 1 respectively,to genera~ a prokaryotic expression pET一28aVP22 an d eukaryotic expression plasmid pEGFPVP22.pET一28aVP22 was consquenfly transformed into E.coli BL21(DE3).After induced with IPTG.the fusion protein was expressed an d the molecular weight was about 38l(Da. pEGFPVP22 Was tran sfected into PK一15 cells an d the clone cells were selected out wim G418.Th e autofluorescence of the clone cells line tran sfected with pEGFPVP22 could be observed under inve~ed fluorescence microscope.It is interesting that the fluorescence primarily targeted the nucleus of cells whereas the autofluorescence of cells tran sfected with control vector pEGFP—C1 localized throughout the cytoplasm.
Abstract:In this experiment the conditions for the expressing of HCLV E2 glycoprotein A and D an tigenic domains in E coli were optimized.On this basis,the expressed protein was purified by dissolving the inclusion body in Tris.HC1 with 6 mol/L guan dinine an d passed throh a His Bind affinity chromatography.The product was collected an d th e concentrated recombinan t protein was quentified. Th e recombinan t protein was then used to coat 96一well ELISA plates an d applied to detect the titers of the serums from swine immunized with HCLV vaccine.
Abstract:Grass carp reovirus(GCRV)is a disaster agent to aquatic animals,which belongs to genus Aquareovirus.family Reoviridae.Sequences an alysis revealed GCRV S2 was 3877 nucleotides long encoding a 1 38kDa protein VP2,which is deduced as virus RNA polymerase.To understan d the properties of its RN A polym erase,here we constructed 2 expression recombinan ts as pR/RRpN an d pR/RRpC,that covered the gene sequences of N terminal an d C terminal region of RN A polymerase. The 2 recombinan ts were demonstrated in frame expression by SDS—PAGE,an d their molecular weight are about 98kDa an d 103kDa,which were interest fusion proteins.It showed the fusion proteins were able to bind to rabbit serum an ti GCRV—VP2 by using W estern Blot an alysis.In addition,6XHis—tagged GCRV RN A polymerase products were purified by afinity chromatography an d got around 90% purification of the interest proteins.Th is data provided the evidence for further GCRV RN A polymerase characterization.
Abstract:Genechip technology was adopted to diferentiate the four animal vesicular diseases in order to distinguish them properly,rapidly an d largely.Thirty-nine amplified target gene fragments were puriried with Qiaquick 96 plate kit,and diluted with TE to obtain a concentration of 0.36 0.36/ag//aL.A . microarray was spotted with a GMS417 (pine.and.ring spotter)apparatus,after which the cDNA fragments were coupled with ..NH2 on a genechip using ultraviolet cross linkage technique.Afterwards the quality of chip was confirmed by aspecific hybridization with salmon sperm .Large quan tities of Foot.and.mouth disease virus(FMDV)RNA from infected tissues and Swine vesicular disease virus rSVDV)RNA from infected cell culture supematant were extracted and,fluoromarker were used to label cDNA fragments of FM DV an d SVDV when they am plified with random primers.After the labeled cDNA was randomly sheared by ultrasonic treatment,the broken pieces were SUbjected to specific hybridization,an d then photoscanned using optimal fight intensity,an d the result an alyzed with Imagene software.Comparison of the signals from the specimens while taking into consideration correspondent position of each virus on the genechip,allowed differential diagnoses of the four kinds of vesicular diseases.The diagnostic genechip method was rapid,sensitive and exact,rush through.
Abstract:A luciferase gene has been inserted into the recombinan t plasmid PIDNV—pUCl l9 which contained partly deletion of genome of Periplanete fuliginosa densovirus(PfDNV).The recombinant plasmid with luciferase gene was co··transfected with pfDNV·pUCl l9 into Periplanelefuliginosa larvae an d had a high luciferase gene expression in enteron of the tran sfected larvae.
DNA of Syngraphafalcifera nuclearpolyhedrosis virus D—clone(Sfa-D clone)was extracted and digested by three kinds of restriction endonuclease.W e calculated its molecular weight an d measure its melting temperature.G+C% .virus particle an d polyhedrin were purified.The structural polypeptides an d polyhedrin are an alysed by SDS—PAGE.
Abstract:According to the published genome seqflences of Porcine circovirus type 2(PCV2)and Porcine reproductive and respiratory syndrome virus(PRRSV),primers were designed and PCR, RT-PCR were set up for the detection of PCV2 an d PRRS respectively.W ith the established meth ods, 38 clinical samples from the respiratory disease pigs were detected for the presence of PCV2 an d PRRSV.The results demonstrated that 22 sam ples were posirive for PCV2,27 sam ples were positive for PRRSV an d am ong th e above positive sam ples,1 8 sam ples were positive fo r both viruses.The data obtained in the present study indicated that PCV2 an d PRRSV maybe play an important role in the course of the development of respiratory diseases.