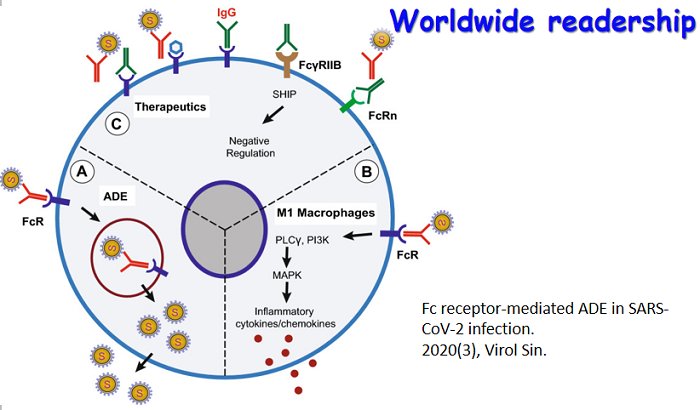
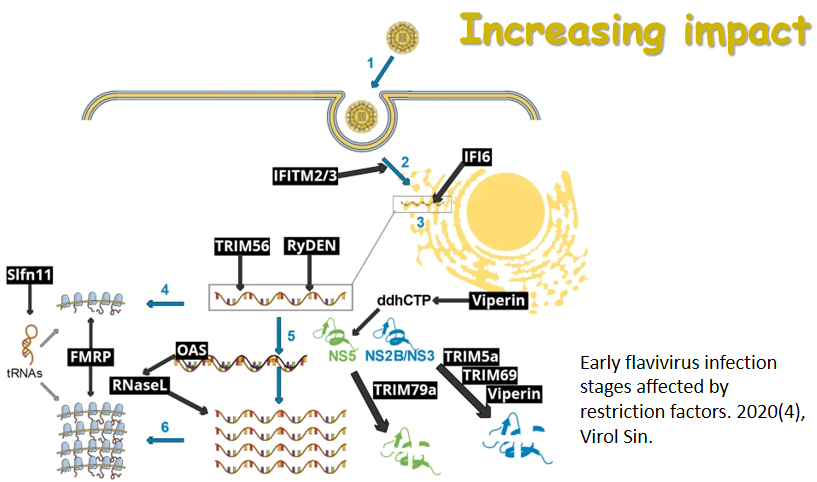
|
To develop the pathogenesis of Hantavirus infection, the differentially expressed signal trans- duction-associated genes of integrin in Hantavirus infected cells were investigated by using cDNA microarray. The primary human embryo pulmonary fibroblasts (HEPFs) were infected with Hantaan 76-118 virus. Total RNAs were extracted from infected and normal control group 6h, 24h and 96h post infection, respectively. During reverse transcription, the cDNAs were labeled by Cy5-dUTP and Cy3-dUTP. The labeled cDNAs hybridized with the Biostar-ⅡH gene chip. After hybridization, the fluorescence signal was analyzed by scanning. It showed that the expression level of most (113/150) Hantavirus receptor and signal transduction-associated genes was normal, however, 37 genes presented obvious change. Among them, expressions of 31 genes changed at 6h post infection. 13 genes were down-regulated, and 18 up-regulated. However, 5 and 4 genes expressed diversely at 24h and 96h post infection respectively. Furthermore, some integrin signal transduction pathway-associated genes such as the gene of some related protein kinases were up-regulated at 6h post infection. The gene of enhancer of filamentation 1 was down-regulated at 6h and 96h post infection. The gene of microtubule-associated protein 1A was up-regulated at 6h post infection, but down-regulated at 24h and 96h post infection. These indicate that Hantavirus infection is related to integrins.
A Vero-E6 culture system was developed to assay Hepatitis C virus (HCV) replication by plasmid (pHCV) transfection,which contains a T7 promoter at the 5’end, full-length cDNA of the HCV genome and a T7 terminator. To facilitate high-level transcription of HCV RNA, Vero-E6 cells were transfected with pHCV and then infected with recombinant vaccinia virus (vTF7-3) containing the T7 RNA polymerase gene. This transfection-based cell culture system produced high levels of HCV genome (107-108 copies/mL) detectable by real-time quantitative PCR and the production of HCV RNA transcripts was confir med by RT-PCR. Western blot analysis revealed that HCV structural and non-structural proteins were correctly processed. The transfected Vero-E6 cells assembled 40-50nm virus-like particles were analysed with transmission electron microscope. This model can be utilized for studying mechanisms of HCV replication, preparation of HCV vaccine and to test potential antiviral drugs.
To study the enzymatic activity,the recombinant HCV NS3 protein with both protease and helicase activity was expressed and purified. The nonstructure gene 3 (NS3) of HCV was amplified and inserted into plasmid pPIC9 and the recombinant plasmid pPIC-NS3 was transformed into Pichia pastoris strain GS115. Recombinant protein was produced by induction of the AOX1 promoter with methanol. The recombinant protein was first purified by Hitrap SP cation exchange and then by Mono S HR column. In vitro cleavage assay showed that the recombinant protein has protease and helicase activity.
The pregenomic RNA, serves as a template for reverse transcription and encodes the reverse transcriptase for Hepatitis B virus (HBV) replication, has the energetically flexible secondary and tertiary structures, which may reduce the accessibility of ribozyme. To identify accessible target sites for hammerhead ribozyme, the secondary structure analysis of the full-length HBV pregenomic RNA was analyzed using the newest bioinformatic tools. Screening the accessible sites for ribozyme based on the structures show that the accessible sites located on the encode region of S, C, X genes and almost not on the regulation region or the encapsidation signal(ε). Thereby three hammerhead ribozymes targeted different sites were designed and transfected into cells to evaluate the catalytically inactivation of HBV gene expression. Experimental results validated that the encoding regions of S, C, X genes were the effective cleavage sites for hammerhead ribozyme and the ribozyme targeted X gene was most potential to inhibit HBV RNA accu- mulation.
Recombinant plasmid pCNS2 was constructed by inserting Hepatitis C Virus full-length NS2 gene between Xho I and Hind III sites of an eukaryotic expression vector pCDNA3.1(-). The recombin- ant plasmid pCNS2 was transfected to Huh-7 cells with LipoVecTM and HCV NS2 mRNA and protein produced in transient expression system was detected at the transcriptional and translational levels,respe- ctively. This indicated that pCNS2 could be expressed successfully in Huh-7 cells. Cotransfected pCNS2 of different doses to Huh-7 cells with reporter plasmid pNF-κB-Luc,the activity of luciferase controlled by NF-κB promoter/enhancer elements was detected by luciferase assay kit 48h after transfection. The results showed that the expression of luciferase in Huh-7 cells transfected by the pCNS2 decreased about two to four folds compared to control plasmid. It is suggested that the HCV NS2 protein distinctly suppressed the activities of NF-κB in a dose-dependent manner in Huh-7 cells and this will enable to investigate on the role of NS2 in the pathogenesis of chronic HCV infection.
Full-length NS5A gene of Hepatitis C virus(HCV) was amplified by PCR, using the plasmid pBRTM/HCV 1-3011 containing HCV full-length open reading frame(ORF)as template, and cloned into the eukaryotic expressing plasmid pcDNA3.1(-)by DNA recombination technique. The recombin- ant vector was identified by digestion with restriction enzymes and polymerase chain reaction and by directly sequencing. Then both the recombinant vector pcDNA3.1(-)-NS5A and the control vector pcDNA3.1(-)were transfected Hela cells using LipoVecTM. The cells expressing NS5A stablely were selected by G-418 and further proved by RT-PCR and Western blot analysis. We found the growth of Hela cells expressing NS5A was slower than the cells transfected by pcDNA3.1(-)in the same culture condition, and the population doubling time of Hela cells expressing NS5A gene is increased about 50%(about 35-35 hours). There was no significant difference between the control cells and the cells transfected with pcDNA3.1(-) (about 23-24 hours). The results indicate that NS5A can inhibit the proliferation of Hela cells.
Entire ORF of Hepsin was isolated from adjacent live tissue of HCC with RT-PCR. Hepsin gene was inserted into expressing vector pCMV-tag-HS. The recombinant vector pCMV-tag-HS was co-transfected into liver tumor line Hep G2.2.1.5 with plasmid encoding HBx protein. The results indicated that co-expression of HBx and Hepsin increased the expressing level of viral proteins and the copy number of HBV, which implicated that the association of HBx and Hepsin promoted the replication of HBV.
Using BTV-HbC3 to infect tumor cells including human lung cancer SPC-A-1 cells, human cervical carcinoma HeLa cells, human astrocytoma U251 cells, mouse-derived astrocytoma C6 cells and human embryonic lung cells and observe the cytopathic effects(CPE); then using transmission electron microscope and double immunodiffusing test to compare the infectivity of BTV-HbC3 to these cells. Reverse transcription-PCR was used to detect the propagations of BTV-HbC3 in these cells. The results indicated that BTV-HbC3 does not infect normal human embryonic lung cells, but it can selectively replicate in tumor cells and produces different degree of cytopathic effects and can induce apoptosis. Tumor cells were killed in the end. Among all the cells, human lung cancer SPC-A-1 cells have the highest sensitivity to BTV-HbC3. BTV-HbC3 can targetedly kill some kinds of tumor cells. These data provide the laboratory basis for approaching the possib-ility of BTV-HbC3 as an oncolytic virus.
NS1 gene of Porcine parvovirus SD-68 strain was cloned and sequenced. Based on the sequence we can conclude that NS1 gene of SD-68 strain is composed of 1989 nucleotides, encodes a polypiptide of 662 amino acids and shares 99.9%, 99.9%, 99.7%, 98.1% identity of nucleotides and 99.7%, 99.5%, 99.5%, 96.7% identity of amino acids with that of SY-99 strain, Kresse strain, NADL-2(5075) strain and NADL-2(4973) strain, respectively. The distances of NS1 gene of all strains analyzed by phylogenetic tree, we can conclude that SD-68 strain is near to MVM(i) strain and far to BPV3 strains. The residues of Thr and Ser at the position 435 and 473 in the NS1 gene of SD-68 strain are phosphorylated sites because they are also found at the same position in the NS1 gene of MVM(i) strain.
With the reverse transcription polymerase chain reaction (RT-PCR), the DNA sequence encoding the cattle’s interferon-gamma (BovIFN-γ) and the signal peptide was amplified, from the total RNA of the lymphocytes stimulated with concanavalin A in the peripheral blood of bovine, which was then cloned into vector pMD18-T and sequenced. The sequencing result showed that there is 100% homology among the documented sequences and sequence reported here, which was successfully inserted into the expressing plasmid pRLC and was highly expressed in E.coli. SDS-PAGE result showed that the cloned recombinant protein was expressed in the form of inclusion bodies in the E.coli cell with molecular weight of 16kDa and was amount to 42% of the whole protein in the E.coli cell, which was subsequently dissolved in 7mol/L guanidine chloride and renatured with dilution in refolding buffer containing 0.5mol/L guanidine chloride. In order to obtain pure protein, the renatured boIFN-γ was desalting by Hiprep 26/10 and purified by Hiprep Sephacryl S-200 chromatography. The purified product could inhibit the cytopathic effect, which verified it has the high cytokine activation.
To investigate Hepatitis E virus,(HEV) infection in swine in Xinjiang and the extent of genetic variation among Chinese swine HEV strains, seventy pig fecal samples from an anti-HEV positive farm in Xinjiang were tested for the presence of HEV RNA by reverse transcription nested polymerase chain reaction (RT-nPCR). Thirteen of 70 (18.57%) pigs were positive for HEV RNA. The nucleotide sequence of 348bp region within open reading frame 2(ORF2) of the 13 swine HEV isolates was determined. The DNAstar software was used for nucleotide sequences analysis. Sequence comparison showed that 13 swine HEV isolates shared 97.1%~100% nucleotide sequence identities and 74.1%~77.6%, 71.6%~74.1%, 73.3%~78.2%, and82.8%~91.4% homogeneity to human HEV genotypes Ⅰ,Ⅱ,Ⅲ and Ⅳ, especially had a 89.4%~91.4% identity to Chinese human HEV subgenotype ⅣA. Phylogenetic analyses further revealed that these swine HEV isolates were closely related to HEV ⅣA isolated from patients with acute hepatitis. Meanwhile these swine HEV isolates had 82.6%~91.3% homogeneity to other Chinese swine HEV and were grouped into genotype Ⅳ. Our findings further support the hypothesis that swine reservoirs for HEV infection.
The porcine IFN-βgene was amplified by polymerase chain reaction. The amplified fragment was cloned into pGEM-T-easy vector and then sequenced. The result indicated that the cloned gene was a complete porcine IFN-βgene with the codes for signal peptide ,which had the identities of 100% with the poIFN-βgene published in the GenBank. Another pair of primers was designed to sub-clone the gene coding porcine IFN-βmature protein and one rare codon near 5` terminus was modified to the biased codons of E.coli. After sequencing, the sub-cloned IFN-βgene was successfully inserted to expression vector pRLC and expressed in E.coli, the expressed protein was about 17.3% of the total cell protein. Recombinant porcine IFN-βexpressed as inclusion body, which was dissolved in 6 mol/L guanidine chloride and subsequently re-natured by diluting with refolding buffer containing GSH and GSSG. In order to obtain pure protein, the re-natured poIFN-βwas purified by Sepha- crylS-200 chromatography. As a result, the purified product was verified to be of high cytokine activity by inhibiting the cyto-pathogenic effect, which is about 5.6x105U/mg. In addition, the inhibition effect of recombinant poIFN-βon PEDV was determined using CPE50 method. The results indicated that high concentration of recombinant poIFN-βcould completely inhibit PEDV on PK-15 cell line.
pp24 gene was amplified from genomic DNA of Marek’s disease viruses (MDV) Md11 strain by polymerase chain reaction(PCR) and then cloned into pGEX-6P-1 according to the right open reading frame(ORF). The recombinant plasmid was transformed into E.coli BL21 strain for expression with the induction of IPTG. SDS-PAGE and Westen-blotting with goat-anti-GST antibody validated the expression of GST-pp24 fusion protein. The expressed specific band was excised from the gel and injected into four-week-old mice 3 times, then the antiserum was collected from the mice and used for IFA with Chicken Embryonic Fibroblasts(CEF) infected by MDV Md11, CVI988 and GA strains respectively. The positive staining was found in the MDV plaques, which shows that the fusion protein of pp24 in vitro expressed has some epitopes of natural pp24.
To establish a technique for preparing rabies vaccine by perfusion culture on microcarriers in bioreactor, a 5 litre bioreactor was filled with 199 cell culture medium which contains 10g/L of microcarriers. Then Vero cells were inoculated with a final cell density of 1×105/mL . After 7 days of culture, cell density can reach 6~7×106/mL, then rabies virus VaG strain was inoculated with a MOI of 0.01. After 24h inoculation, the cultured viruses were harvested continuously for 12 days. The titer of collected virus in the culture medium was about 6.0 to 8.5 logLD50/mL. From these collected viruses, vaccines were prepared through the steps of concentration, inactivation and purification, The quality of these vaccines meets “The Chinese Requirements for Biologics Year 2000”. This lab-scale method for preparing rabies vaccine by perfusion culture on microcarries is viable.
To improve the safety of recombinant vaccinia virus,a non-replicating vaccinia recombi- nant co-expressing rabies virus glycoprotein(RG) and nucleoprotein(RN) without a selectable marker was reconstructed. LacZ between fragment C and fragment K in non-replicating recombinant vaccinia virus VTKRG△CKlacZ was deleted and was substituted with rabies virus nucleoprotein by homologous recombination resulting in VTKRGΔCKRN. The recombinant vaccinia virus was screened by a two-step selection under, G418 and with White/Blue plaque color change as well as immunofluorescence. By Dot Blot analysis, RG gene and RN gene were shown to integrate stably into the non-replicating vaccinia recombinants even after 20 passages in CEF cells. The virulence of VTKRGΔCKRN was obviously less than that of VTKRG in naked mouse. The non-replicating characterization of VTKRGΔCKRN was also stable. It was proved that VTKRGΔCKRN could express RG and RN at the same time. VTKRGΔCKRN could elicit viral neutralizing antibody(VNA) similar to that of VTKRG by inoculated into the mice,and thus protect the animals from lethal challenge of rabies virus. It could elicit efficient viral neutralizing antibody(VNA) and protect the dogs from lethal challenge of rabies street strain SBD by intramuscular inoculation at dose of only 1.6×106PFU. This study confirmed the efficacy and safety of non-replicating recombinant vaccinia virus VTKRGΔCKRN.
The PCR product of Helicoverpa armigera single nucleocapsid nucleopolyhedrovirus HaSNPV fp25k gene was cloned into a prokaryotic expression vector pProEXHTb. After inducing with IPTG, the fp25k gene was successfully expressed in E.coli DH5α. The expression product was 32 kDa. This protein was purified and injected into rabbit to raise polyclonal antiserum. Western blot analysis using this antiserum showed the specific reaction to FP25K protein expressed in both prokaryotic with GST fusion and viral infected insect cells. The antibody made it possible to analysis the function this protein in infected insect cells.
It is very important to understand the laws of Tobacco mosaic virus(TMV) inactivation under different water bodies and temperatures. Choosing the typical plant virus TMV as a model virus, the inactivation dynamics of it in Min River water, tap water, super-pure water, life sewage and sterile sewage under different temperatures were studied in this paper. The results showed that the temperature was a very important factor to influence the survival of TMV. The higher temperature of the water body, the faster the inactivation rate of TMV. Besides the environmental temperature, some water quality factors also influence the inactivation rate of TMV. The influences of the dissolved salts on the inactivation of TMV were different from the various environmental conditions. Some microbes or their metabolites were harmful to the survival of TMV. The biodegradable organic substances lead to TMV inactivation may be throught to promote the bacteria multiplying.
5 strains of verocytotoxigenic (VT) bacteriophage were isolated from E.coli O157 strains, cow feces, chicken feces, sewage and purified by two-layer agar assay. Gene vt2 was amplified by PCR in these phage isolates. The plaques of these phages were 0.5-2mm in diameter and the titre of phages with lucidity plaques against host strain MC1061 was more than 109 PFU/mL. A lysogen with vt2 gene was obtained after transfecting MC1061 by VT2 phage SHФW1 isolated from sewage. The cytopathic effect on Vero cells was produced after inoculating the filtrate from the above lysogen, while no cytopathic effect was appeared inoculating the filtrate from MC1061 cultured in the same condition. All these showed that VT2 phages were common in environment and VT2 virulent factor could transfer horizontally through the lysogenic infection by VT2 phage. VT2 phage is related to the virulence of bacteria.
With a pair of primers and the E. coli chromosome DNA as the template, an intact recA gene was obtained by PCR. The recombinant plasmid was constructed by ligating the PCR product and pUC18 in vitro and then transformed into E. coli DH5α and E. coli k12(λ+), respectively. The plasmid pUR4 containing the recA gene has been screened. The biological function of recA gene in different strains was determined with or without UV induction. The results indicated that the recA gene showed a remarkable physiological function in the λ lysogenic bacteria. It could induce the λ prophage to enter the lysis cycle from lysogenic state. This recombinant plasmid will be a useful tool in the studies of the inductive mechanism of λ prophage and the repair of cells damaged by UV-irradiation.
Porcine reproductive and respiratory syndrome virus(PRRSV) isolates identified in samples from 50 field cases originated from eastern China herds were studied. ORF5 gene of PRRSV isolates was amplified by reverse transcript polymerase chain reaction(RT-PCR) and restriction fragment length polymorphism (RFLP) patterns for enzymes MluⅠ, HincⅡand SacⅡwere determined on these ORF5 genes. The RFLP pattern obtained from 39 isolates was 2-2-1(78%), 3 isolates was 1-1-1(6%) and 8 isolates identified in 2003 was 1-3-1 (16%). The results showed PRRSV strains have variations and existed no less than 3 gene subtypes in the area.
The purified recombinant nuclocapsid protein of PRRSV was used to conjoined with latex, and the conjoined concentration,temperature and time were optimized, a detection method for serum antibodies against Porcine reproductive and respiratory syndrome virus was established. Some tests for the method’s sensitivity, specificity and stability were confirmed. About 200 serum samples were detected by using the method, and these samples were also detected by recombinant N protein based ELISA and IDEXX ELISA kit. The result showed that the agreement ratio of the recombinant N protein based LAT with other two methods arrived at 93%, 78%, respectively.
Enzyme-linked immunosorbent assay(ELISA), direct florescent antibody staining, and RT-PCT were used to detect growth characteristics of Cassical swine fever virus C-strain (Derived from Spleen) in SK6 cell. The results indicated than C-strain (Derived from Spleen) can grow in SK6 cell at a lower level. Direct florescent antibody staining method was not suitable for the detection of attenuated lapinized C-strain. The study provided a primary guide for the detection of attenuated classical swine fever virus. It also supplies an elementary foundation for the study of its growth characteristic in SK6.
A novel procedure was used for cloning large adenovirus genome fragment by the homologous recombination in E.coli strain BJ5183. The 11.2Kb downstream fragment of the CAV-2 strain YCA18 genome was cloned by homologous recombination, the 1029bp left end and the 970bp right end of this fragment were separately amplified by PCR. They were then cloned into plasmid pPoly2 with direction from left fragment to right fragment, obtaining a“rescue”plasmid pT615. The pT615 was liberalized by Hind Ⅲ and PstI digestion and was cotransformed with the purified CAV-2 genome which was cut by BstBI into competent E.coli strain BJ5183. Recombinant plasmids harboring the 11.2Kb downstream fragment of CAV-2 genome were obtained after bacterial intermolecular homologous recombination. The recombinant efficiency of all E.coli strains tested was 78.3%. One of the recombinant plasmids, pT618, was further identified by enzyme digestion analysis and PCR amplification. The results showed the plasmids contained the 11.2kb fragment downstream the genome of CAV-2.
In order to study the feasibility of gene chips technology in the detection of HBV mutation associated with lamivudine, we detected the mutation of HBV in peripheral blood of 30 patients treated with lamivudine for at least half a year by gene chips. The result was compared with that from direct sequencing. Both results are highly coincident. The rate reaches 100% while detecting single strain of virus infection, and 85% in multi-strains virus infection. Gene chip technology is quite valuable and practical in future clinic.