2019年11月4日,中国科学院武汉病毒研究所张波研究员团队和军事科学院军事医学研究院微生物流行病研究所秦成峰研究员团队密切合作在Emerging Microbes & Infections期刊在线发表论文,首次利用新型的近红外荧光蛋白(infrared fluorescent protein, iRFP)成功开发了基孔肯雅报告病毒,实现了基孔肯雅病毒体内外感染的实时可视化示踪。原文题为“Visualization of chikungunya virus infection in vitro and in vivo”(基孔肯雅病毒在体外和体内感染的可视化)。
在该项研究中,研究人员利用反向遗传学技术将近红外荧光蛋白(iRFP)基因构建到基孔肯雅病毒基因组中,成功拯救得到了可以表达近红外荧光蛋白的基孔肯雅报告病毒。进一步研究表明,该报告病毒和亲本基孔肯雅病毒的蚀斑形态和生长曲线等生物学特性基本一致。与亲本病毒不同的是,该报告病毒在细胞中复制的同时可以表达近红外荧光蛋白,其荧光信号强度与病毒滴度线性相关(图1)。
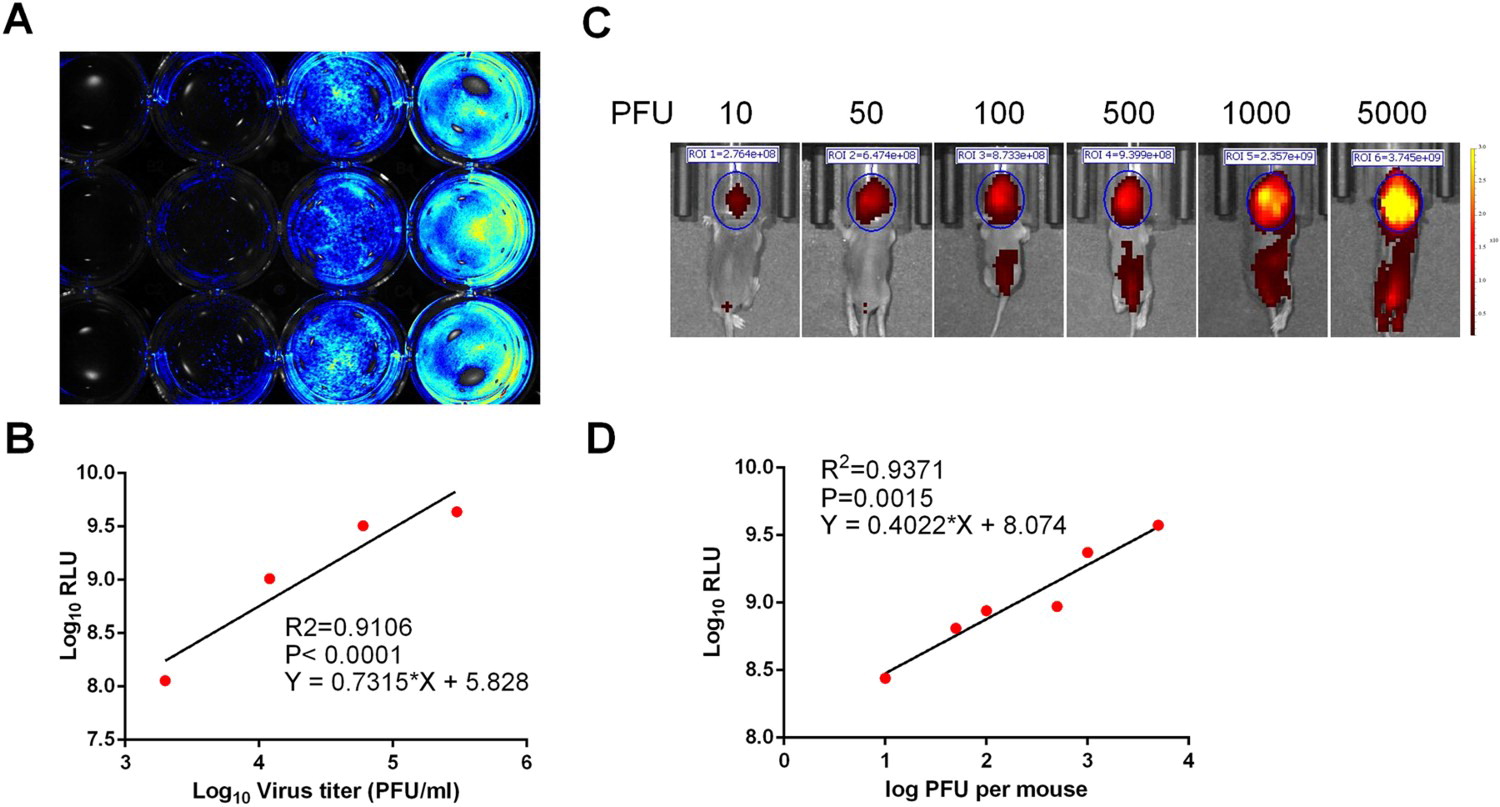
图1 CHIKV-iRFP在体外与体内感染时,荧光强度与病毒复制能力的相关性。
更为重要的是,研究人员利用该报告病毒实时再现了基孔肯雅病毒在不同小鼠模型体内的感染和扩散过程,证实了该病毒对小鼠骨骼系统的特殊嗜性(图2),与基孔肯雅热患者的关节痛症状十分吻合。上述发现不仅揭示了基孔肯雅病毒生命周期中的关键环节,也为基孔肯雅病毒疫苗和药物的评价提供了重要工具。
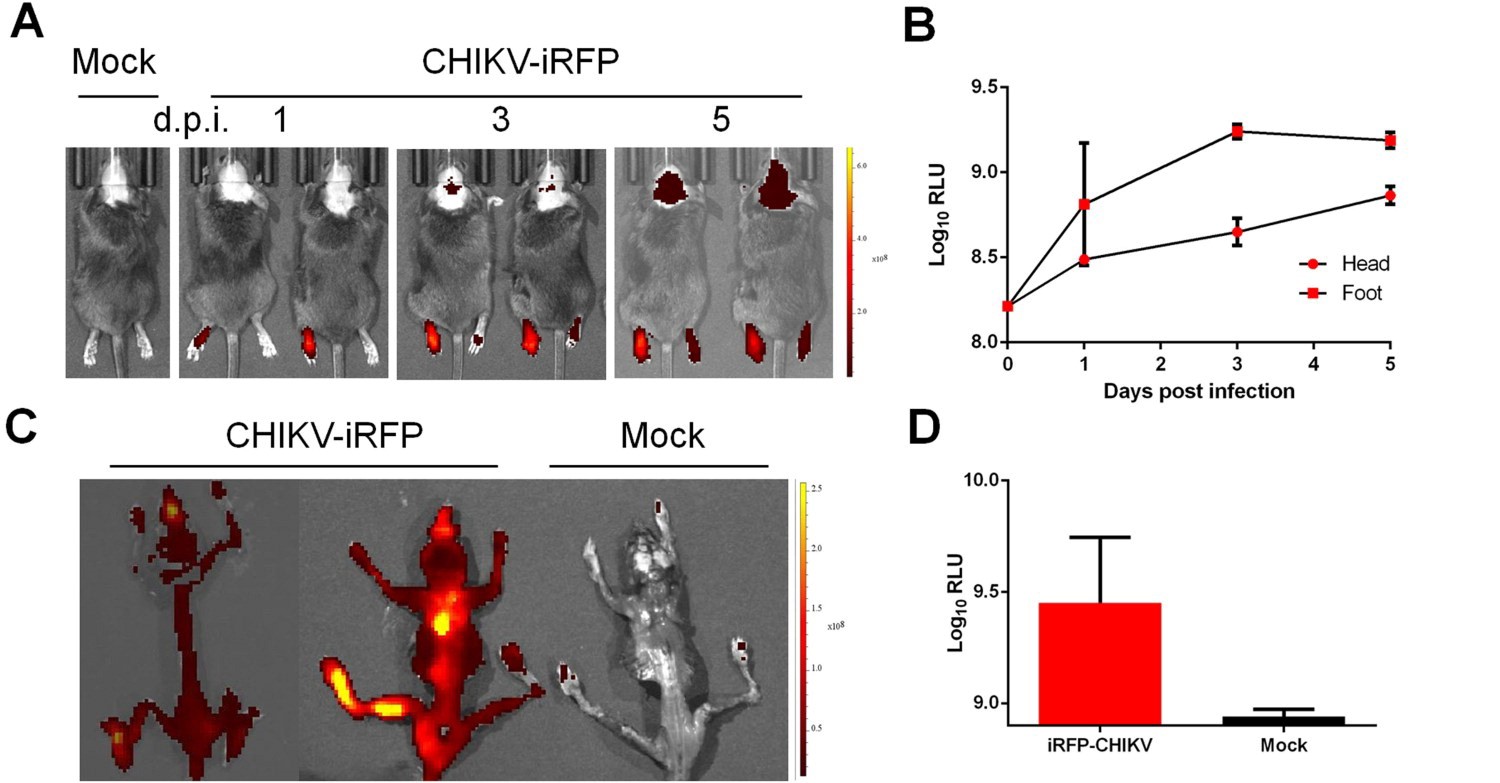
图2 CHIKV-iRFP在小鼠体内动态感染观察,和对骨骼系统的特殊嗜性。
中国科学院武汉病毒研究所的张红垒博士(现河南农业大学牧医工程学院)与军事医学研究院的董好龙硕士为论文的并列第一作者,中国科学院武汉病毒研究所张波研究员与军事科学院军事医学研究院秦成峰研究员为本论文共同通讯作者。该项目获得科技部重点研发计划和国家自然科学基金项目联合资助。
原文链接:
https://www.tandfonline.com/doi/full/10.1080/22221751.2019.1682948
ABSTRACT: Chikungunya virus (CHIKV), a mosquito-borne alphavirus, has become an important re-emerging pathogen with its rapid spread to many non-endemic areas. The lack of effective vaccines and antiviral agents is largely attributed to the elusive infection and dissemination dynamics in vivo. In this study, we designed and developed a novel, replication-competent, CHIKV reporter virus (CHIKV-iRFP) encoding a near infrared fluorescent protein (iRFP). In vitro and in vivo characterization demonstrated that CHIKV-iRFP retained similar replication and virulence phenotypes to its parental virus. Neonatal BABL/c mice and IFNAR^−/− A129 mice were highly susceptible to CHIKV-iRFP infection. Following intracranial (i.c.) inoculation, CHIKV-iRFP efficiently replicated and disseminated into whole body, resulting in rapid death in an age-dependent manner. Remarkably, upon footpad injection, CHIKV-iRFP readily disseminated from footpad to head and whole skeleton, with a specific tropism for bone marrow. Taken together, this novel reporter virus provides a powerful tool to track real time CHIKV replication and to test the in vivo efficacy of vaccines and antiviral therapeutics.